Mariam Rabaa; Amani Aridi; Ghassan Younes; Ramadan Awad
Abstract
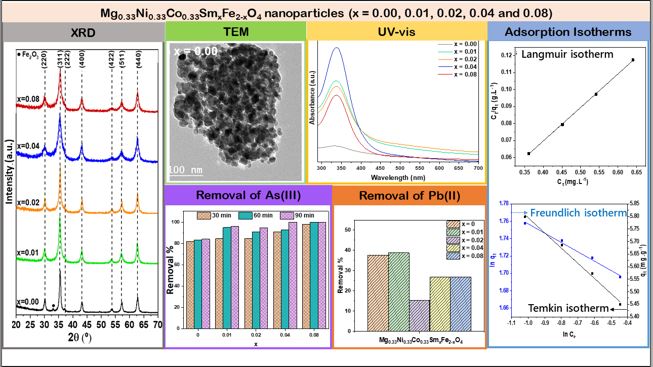
Heavy metal ions, such as As (III) and Pb (II), are harmful even at trace levels and have caused series health effects on living beings. Therefore, removing these heavy metal ions from the aqueous environment is highly desirable. In this study, Mg0.33Ni0.33Co0.33SmxFe2-xO4 nanoparticles, where x = 0.00, ...
Read More
Heavy metal ions, such as As (III) and Pb (II), are harmful even at trace levels and have caused series health effects on living beings. Therefore, removing these heavy metal ions from the aqueous environment is highly desirable. In this study, Mg0.33Ni0.33Co0.33SmxFe2-xO4 nanoparticles, where x = 0.00, 0.01, 0.02, 0.04 and 0.08, were synthesized by the co-precipitation method and characterized using X-Ray Diffraction (XRD), Transmission Electron Microscopy (TEM) and UV-Vis spectroscopy techniques in order to study the structural and optical properties. The prepared nanoparticles were applied as adsorbents for the removal of As (III) and Pb (II) from wastewater. Among the prepared samples, Mg0.33Ni0.33Co0.33SmxFe2-xO4 nanoparticles with x = 0.04 and 0.08 exhibited improved adsorption performance where As (III) was totally removed after 90 min. The experimental adsorption data of As (III) was well fitted with a second-order kinetics model and Langmuir isotherm. Furthermore, the highest removal % of Pb (II) was revealed by Mg0.33Ni0.33Co0.33SmxFe2-xO4 nanoparticles with x = 0.01. Thus, doping Mg0.33Ni0.33Co0.33Fe2O4 with Sm improved the adsorption performance of nanoparticles for the removal of As (III) more than that of Pb (II).