Document Type : Original Research Paper
Authors
PG Department of Chemistry, JSS College of Arts, Commerce and Science, A Research Centre Recognized by University of Mysore, Mysuru-570 25, Karnataka, India
Abstract
Novel results in this study showcase the utilization of sunlight-dried, ground Lablab purpureus husk (LLPh), treated with water and alkali, as a highly efficient bio-adsorbent for the removal of cationic dyes from aqueous solutions. Methylene blue (MB), malachite green (MG), and crystal violet (CV) were effectively adsorbed onto NaOH activated LLPh (NaOH-LLPh) as bio-adsorbent. Employing the Chromatrap method within a column, successfully removed these dyes, while the surface morphology of the bio-adsorbent was elucidated through scanning electron microscopy (SEM) analysis. FTIR spectrometric data revealed valuable insights into the extent of adsorption. The impact of factors including adsorbate concentration, adsorbent dose, pH, contact time, and flow rate on the adsorption process was systematically studied and optimized. Up to 1000 µg/mL of MB and MG, 50 µg/mL of CV were found to be effectively removed by adsorption at pH 4-5, 3 and 2, respectively, at the flow rate of 1 mL/min. The results of kinetic studies and adsorption isotherms of above-mentioned dyes indicates that, all the three dyes follow the pseudo-second order kinetics. The adsorption of MB and MG are well fitted with the Langmuir isotherm model. The other dye CV suits with the Freundlich isotherm model. Based on the results, NaOH-LLPh, as an inexpensive and eco-friendly adsorbent, is suitable for the removal of cationic organic dyes from aqueous samples.
Graphical Abstract
Highlights
Keywords
INTRODUCTION
Dyes are widely used to colorize the products in textile, leather, paper, wood, wool, pharmaceutical, cosmetic as well as in food industries [1, 2]. The effluents discharged straight into the water bodies from these industries are largely responsible for endangering aquatic life and creating carcinogenic effects. Long-term contact with dyes causes itching and irritation in humans; however, children are affected more quickly [3]. Thus, it is inevitable to make sure that industrial effluent is rendered less contaminated by removing harmful compounds by utilizing removal processes. Various techniques, including physical, chemical, and biological methods, can be employed to address significant contaminants such as organic compounds or dyes. The utilization of nanocatalysts [4-6], Fenton catalysts [7], and artificial neural networks [8] for eliminating these pollutants has gained increasing attention in research. Conversely, adsorption is on the rise in popularity due to its efficiency, environmental friendliness, and straightforward interpretability [9].
Methylene blue (MB), malachite green (MG) and crystal violet (CV) are familiar with the IUPC names: [7-(dimethyl- amino)phenothiazin-3-ylidene]-dimethyl-azanium, chloride (Fig. 1a), [4-[[4-(dimethylamino)phenyl]-phenylmethylidene]cyclohexa-2,5-dien-1-ylidene]- dimethyl-azanium, chloride (Fig. 1b) and tris(4-(dimethylamino)phenyl)methylium chloride (Fig. 1c), respectively.
Among the many synthetic dyes, MB, MG, and CV are the most widely used organic cationics in the textile and pharmaceutical industries. The effluents containing trace levels of these organic cations must be eliminated to reduce the nocuous properties of water [10, 11].
Bio-adsorbents have garnered considerable attention in recent years for their effectiveness in removing various contaminants from wastewater [12]. These materials, derived from natural sources like plants, agricultural waste, and microorganisms, offer distinct advantages such as affordability, abundant availability, biodegradability, and environmental friendliness [12]. Researchers have explored a broad spectrum of bio-adsorbents and devised innovative strategies for pollutant removal. Some of the adsorbents investigated for MB removal include activated carbon from jatropha husk [13], magnetic peanut husk [14], phosphoric acid-treated balanites aegyptiaca seed husk powder [15], sodium hydroxide-treated rice husk [16], mentha plant waste [17], rice husk ash [18], nanoparticles synthesized from rice husk [19], oxalic acid-modified rice husk [20], rice husk biochar [21], porous carbon material from rice husk [22], and neem (Azadirachta indica) leaves [23]. Furthermore, activated carbon from coffee husk [24], zinc chloride-modified hazelnut husk [25], and lemongrass husk [26] were explored for MB removal. Additionally, Hydnocarpus pentandra fruit peel charcoal [27], coffee husk biochar [28], dried biomass of haloxylon recurvum plant stems [29], lathyrus sativus husk [30], magnetic rice husk ash [31], and biochars from rice husk, cow dung, and domestic sludge were effectively employed, achieving approximately 90% MB removal. Corn plant husks [33], sawdust of Gmelina arborea roxb [34], and palm oil mill effluent waste-activated sludge were also utilized for MB removal [35].
Numerous adsorbents for MG removal, as reported by different researchers, include highly porous quinoa husk [36], sodium carbonate-treated rice husk [37], raw and activated forms of Gibto (Lupinus albus) seed peel powder [38], activated carbon from groundnut shell powder [39], typha australis leaves [40], durian seed-based activated carbon [41], rice husk, Thespesia Populnea bark, Calatropis gigantic bark, corn cob, neem bark, mango bark, root powder of water hyacinth (Eichhornia crassipes), Araucaria cookii bark, and seaweed Enteromorpha [42], hydrochloric acid-treated sphagnum peat moss [43], jackfruit leaf powder [44], root powder of chemically modified activated walnut shell powder [45], garlic root powder [46], luffa aegyptica peel powder [47], Cocos nucifera husk [48], potato peel, and neem bark [49]. Additionally, durian peel-activated carbon [50], date-stone activated carbons [51], xanthate wheat bran [52], activated carbon from teak wood waste [53], clove leaves powder [54], sawdust waste from Gmelina arborea [34], EDTA-modified groundnut husk [55], Rhizophora mucronata stem-barks [56], and lemongrass leaf powder [57] were explored for MG removal.
A few reports described the procedures for the removal of CV includes, including methods using lignocellulosic agricultural waste [58], Eichhornia charcoal [59], NaOH-modified rice husk [60, 61], the activated carbon of African star apples [62], red algae Gelidium pusillum [63], phyllanthus emblica fruit powder [64], olive pomace [65], activated carbon of the coconut shell [66], tartaric acid modified Holarrhena antidysenterica and Citrullus colocynthis husks [67] and Urtica dioica leaf powder [68].
Furthermore, adsorbents derived from Carnauba straw [69], Eucalyptus leaves [70], and amine-functionalized chitosan [71] were identified as effective mediums for both CV and MB adsorption.
In the review of literature, methods to remove MB, MG, and CV were found using a variety of husks as adsorbents. The details about the husk, adsorbent dose, and experimental parameters are given in Table 1. Most of the methods follow a batch adsorption process to trap the dye. Almost in each case, a longer equilibration time for adsorption was noticed.
Although numerous researchers employed different materials to remove MB, MG, and CV, none of them documented the utilization and effectiveness of Lablab Purpureus husk (LLPh). Lablab Purpureus (Haycinth beans) is utilized as a food in South Asia, East Asia, South East Asia, and East Africa. Since LLPh is the most commonly available and discarded trash, its usage for removing dyes from aqueous samples will elevate the interest of researchers. Out of all the described adsorbents, the LLPh material provides a better adsorption surface for the effective binding of dyes, according to careful examination of the performance. The application of the treatment is neither difficult nor complicated.
Hypothetically, LLPh, when processed by sunlight drying, grinding, and alkali washing, can effectively serve as a bio-adsorbent for the removal of cationic organic dyes, namely, MB, MG, and CV from water. It is expected that NaOH-activated LLPh (NaOH-LLPh) will exhibit significant dye removal capabilities, making it a viable and eco-friendly option for wastewater treatment.
The principle of ‘chromatrap’, i.e., trapping of dyes in the column using adsorption onto the adsorbent is streamlined in this work for the very first time. This study earnestly endeavors to assess the potency of NaOH-LLPh as a bio-adsorbent for the effective removal of MB, MG, and CV. It also aims to ascertain the optimal operational conditions, encompassing crucial parameters like adsorbate concentration, adsorbent dosage, pH, contact time, and flow rate. Furthermore, it seeks to delve into the kinetic characteristics of dye adsorption and to scrutinize adsorption isotherms, thus unraveling the intricacies of the adsorption mechanism. Notably, this work represents a pioneering effort in this field.
The pioneering application of the Chromatrap principle through column adsorption, employing the cost-effective NaOH-LLPh, marks the novelty of this research. This breakthrough eliminates the requirement for intricate equipment or specialized techniques, thereby making this methodology accessible in a broader sense. This advancement has the potential to elevate its utilization to standard practice in wastewater treatment processes.
Therefore, a smart attempt was made to remove MB, MG, and CV from aqueous solutions using NaOH-LLPh as a harmless material by column elution approach.
EXPERIMENTAL
Apparatus
The Agilent Cary 60 UV-visible spectrophotometer (Agilent Technologies Ltd, Mumbai, India) was used for all spectrophotometric measurements. Fourier Transform Infrared Spectrometer (FTIR) (Perkin Elmer Instrument, Version 10.7.2, Mumbai, India) was used for all infrared spectral recordings. Scanning Electron Microscope Zeiss (Model EVO LS 15, Germany) with smartSEMTM software was used for the study of the surface morphology of adsorbent before and after adsorption.
Materials
All chemicals utilized in the experiment were of analytical chemical grade. Local distributors provided methylene blue (MB), malachite green (MG), crystal violet (CV), and sodium hydroxide (NaOH) pellets (all from SD Fine Chem Ltd., Mumbai, India). Three water samples were procured from the textile industries of Mysuru city and used for analysis.
Preparation of adsorbent
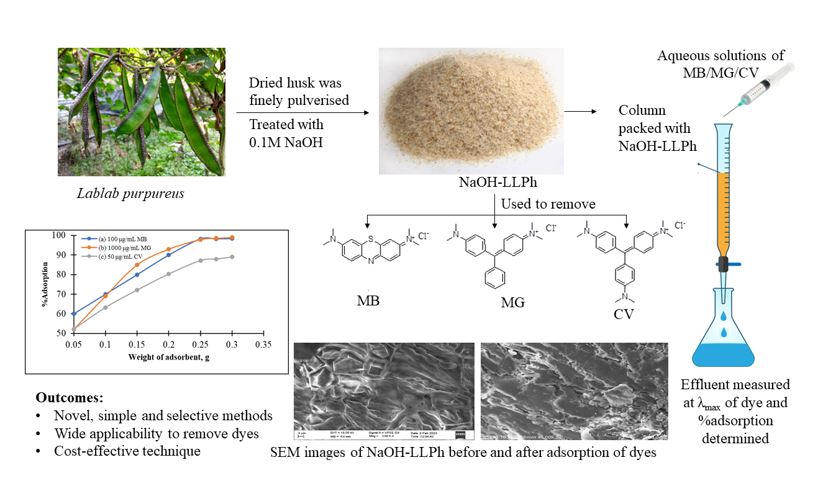
Lablab purpureus was grabbed from the vegetable market in Mysuru and the husk was meticulously separated and dried under sunshine to ensure total moisture removal. To make raw husk powder, the dried husk was finely pulverized. To remove any dust or undesired elements, the raw husk powder was washed many times with distilled water and then dried under sunlight to produce beneficiated husk powder. It was then treated with a 0.1M NaOH solution. This treatment not only aids in the removal of pigments but also improves the adsorption sites on the adsorbent’s surface. The resulting NaOH-activated Lablab pupureus husk (NaOH-LLPh) powder was carefully preserved in a glass jar for later use.
Standard solutions of dyes
By dissolving appropriate weights of chemicals in bi-distilled water, stock standard solutions of MB (3000 µg/mL), MG (3000 µg/mL), and CV (1000 µg/mL) were created. These solutions were employed for elution through the column after adequate dilutions with water.
General procedures
Procedure for column preparation
As adsorbent, 0.25 g of carefully weighed NaOH-LLPh powder was packed in a 25 mL borosilicate glass column. Water was allowed to pass through the column to ensure the absence of any break in the column. Furthermore, a small amount of excess water was left in the column right above the packed adsorbent to prevent the column from splitting.
General Procedure for Dye Removal
With the help of a syringe, a 5 mL of stock solution of each dye (MB and MG: 1000 mg/L; CV: 50 mg/L,) was introduced at a flow rate of 1 mL/min along the sides of the column. The effluent was collected into a small beaker and the absorbance was measured at the corresponding absorption maximum of each dye using a visible spectrophotometer. The concentrations and thus the proportions of dye adsorbed were estimated using the following formula:
where Di and Df are the concentrations of dyes before and after adsorption, respectively.
Application to removal of dyes from industrial effluent
A 10 mL portion of industrial effluent was mixed with 10 mL of separate 2000 mg/L standard aqueous solutions of MB, MG, and CV. From this, precisely 5 mL of the solution was drawn into a syringe and then introduced into a column filled with 0.25 g of NaOH-LLPh adsorbent. All within the confines of carefully optimized experimental conditions, the effluent from the column was collected in a beaker, and its absorbance at the respective wavelength of maximum was measured. The procedure has been repeated two more times. The mean percent adsorption of each dye was calculated by using the formula described above.
Characterization of Adsorbent
Recording of FTIR Spectra
The FTIR analysis of adsorbent before and after adsorption was carried out using KBr pellets and recorded in the range from 500 – 4000 cm−1. Different peaks of vibrations were identified and correlated with the functional groups present in the subjected individual samples.
Scanning electron microscopic (SEM) study of adsorbent
The SEM experiment was performed using the instrument with a thermionic emission electron source under variable pressure mode. The optimal images were captured at an accelerating voltage of 10000 V, a working distance of 6500 µm, and an emission current of 99000 nA. The fast scan speed was maintained during the recording of images.
Physicochemical analysis
Moisture content
To determine the moisture content of the NaOH-LLPh, the temperature of the hot air oven was set to a temperature of 105 oC. A thoroughly cleaned crucible containing 1g of the NaOH-LLPh adsorbent was placed in the oven for 4 hours. The crucible was carefully removed and brought to room temperature. The adsorbent was weighed and the moisture content was then calculated using the formula:
where, M0 and Mf are the weight of adsorbent before and after heating, respectively.
Ash content
A 1g of NaOH-LLPh adsorbent was weighed carefully and placed in a clean, dry crucible and resided in a muffle furnace maintained at 500°C for 4 hours. It was then taken out of the furnace and placed in a desiccator to cool to room temperature. It was then reweighed. The adsorbent’s ash content (AC) was then estimated using the formula:
Ash Content (AC) = (3)
where Ma and Ms are the recorded weights of adsorbent before and after furnacing, respectively.
Volatile Matter (VM)
An accurately weighed 1g of the NaOH-LLPh was taken in a crucible and heated in a muffle furnace set to 500°C for 8 minutes. Then, the adsorbent was removed and the Volatile Matter (VM) was calculated by using the formula:
Volatile Matter (VM)= (4)
where M1 and M2 are the initial and final weights of the adsorbent, respectively, in the experiment.
Fixed carbon content (FCC)
It was determined by subtracting the percentage of MC, AC, and VM from 100%, i.e.,
Fixed carbon content (FCC) =100% - (MC+AC+VM) ---------(5)
The experiments were also performed to determine the MC, AC, VM, and FCC of dyes by following the procedure described above for adsorbent.
RESULTS AND DISCUSSION
The LLPh proves to be a promising adsorbent thanks to its anticipated composition, much like other plant materials. It comprises a diverse array of organic compounds, encompassing cellulose, hemicellulose, lignin, and numerous other constituents. Following thorough washing with both alkali and water, all soluble species are effectively eliminated. Consequently, the activated husk, NaOH-LLPh, endowed with a variety of adsorption sites, is expected to exhibit excellent performance in adsorbing dyes, namely, MB, MG, and CV, as demonstrated in this pioneering study.
The adsorbent has been subjected to FTIR and Scanning Electron Microscopic (SEM) studies before and after adsorption to ensure the expected process.
FTIR spectroscopic study
The FTIR spectra of NaOH-LLPh were recorded before and after adsorption. The adsorbent exhibited three distinct absorption peaks at 1023.80, 1609.92, and 3338.67 cm-1 (Fig. 2a). These peaks are likely attributed to the C-O and C=C stretching vibrations within the adsorbent. The peak at 3338.67 cm-1 is concerned with the stretching vibration of O-H bonds.
The spectrum of the adsorbent after the adsorption of MB is presented in Fig. 2b. Among the total of eight peaks observed, five are significant: around 1096 cm-1 for C-S stretching, 1147 cm-1 and 1252 cm-1 for C-N stretching, and 1337 and 1394 cm-1 for C-H bending vibrations of MB.
In Fig. 2c and 2d, absorption peaks at around 1300 and 1400 cm-1 are evident, which can be attributed to the presence of C-N stretching and C-H bending vibrations in amine and alkyl groups of MG and CV. Additionally, there are additional peaks in Fig. 2d and they are at 800 cm-1 (out-of-plane C-H bending vibrations), approximately 1200-1300 cm-1 (C-N stretching), 1550 cm-1 (C=C stretching), and around 2800-2900 cm-1 (C-H stretching). These peaks unmistakably indicate the characteristic presence of CV on the adsorbent.
The identification of specific functional groups on the surface of the NaOH-LLPh provides valuable insights into the molecular interactions and adsorption mechanisms involved in the adsorption of MB, MG, and CV [37, 73].
SEM Analysis of adsorbent
The Scanning Electron Microscopic (SEM) analysis was performed using an acceleration voltage of 10 kV to examine the surface properties and structural attributes of the adsorbent both before and following the adsorption process. The SEM produced crisp images of the NaOH-LLPh at the nanoscale. At high magnification, the photos revealed convolutions, small pores, and up and down surfaces on the adsorbent’s surface. The emergence of an exposed surface in Fig. 3a and the subsequent concealment of certain spots, as depicted in Fig. 3b, provided the confirmation of adsorption occurring on the material’s sites.
Physicochemical values of adsorbent
The experimental results of MC, AC, VM, and FCC of adsorbent and the dyes are summarised in Tables 2 and 3. As shown in Table 2, it is clear that the NaOH-LLPh exhibited perfect behavior in attracting colors via surface phenomena without enduring its destructive pathway after treatment.
Optimization of experimental parameters
The experimental parameters: adsorbent dose, dye concentration, contact time, flow rate, and pH were investigated. The optimization was carried out by altering the parameter of interest while holding the others constant.
Adsorbent dose
For each dye, different amounts of the NaOH-LLPh adsorbent material were used. To determine the adsorbent load, the effects of flow rate, adsorption capacity, volume of introduction, and speed of removal were considered. The amount of adsorbent in the column was specifically designed to improve removal efficiency. Thus, of the 0.05 to 0.30 g of material load in the column, the feasible and maximum percentage of dye removal was recorded at 0.25g. As a result, the column was filled with 0.25 g of adsorbent to remove the dyes. The effect of adsorbent dosage on percent adsorption is presented in Fig. 4.
Effect of concentration of dye
To accurately determine the adsorption capacity of the NaOH-LLPh adsorbent the amount of dye that can be adsorbed per unit mass needs to be fixed. Therefore, in evaluating the efficiency of the adsorbent material, in a series of separate experiments, 5 mL aliquots of varied concentrations of MB, MG, and CV dyes were passed through the column filled with NaOH-LLPh to cap the concentrations. The resultant effluent of each case was spectrophotometrically measured at the wavelength maxima of 590, 668, and 624 nm for CV, MB, and MG, respectively. It was discovered that utilizing 0.25 g of NaOH-LLPh, 50 mg/L CV was efficiently removed up to 87.98% as shown in Fig. 5(a-c). Up to 1000 mg/L of both MB and MG were effectively removed to an extent of more than 98% by using the same dose of adsorbent. Concentrations of MB exceeding 1000 mg/L necessitated extended contact times to achieve removal rates exceeding 98%. As a result, the threshold concentration for effective MB removal was established at 1000 mg/L.
Effect of pH on adsorption of dyes
The pH influence on the adsorption of MB, MG, and CV dyes was studied. The experimental results revealed that each dye has a different pH dependence. The pH was adjusted between 2 and 11 for each dye to investigate the effect. Following elution via the column, the corresponding percentage elimination of CV, MB, and MG dyes was computed and the effect was depicted in Fig. 6. According to the experimental data, the highest removal of CV was seen at pH 2, resulting in a removal percentage of 87.98. This may be attributed as at pH 2, CV is likely to be positively charged due to its basic nature. This positive charge facilitates its adsorption onto adsorbents, leading to effective removal. For MB, pH between 4 and 5 resulted in the highest removal rates, reaching >98%. The dye MB exhibits optimal removal between pH 4 and 5 using NaOH-LLPh. The charge and solubility characteristics of MB are conducive to adsorption under slightly acidic conditions. It forms a stable complex with the NaOH-LLPh at this pH range. When it comes to MG dye, pH 3 was shown to be the best level, with a removal rate of 98.64%. This is probably due to similar pH-dependent charge and solubility effects. It is positively charged at lower pH levels, enhancing its adsorption to surfaces of NaOH-LLPh.
Effect of contact time
Experiments were carried out to optimize the contact time at room temperature to obtain maximum dye removal. A 5 mL solution containing a specified concentration of CV (50 mg/L), MB, and MG (both 1000 mg/L) was eluted using a column filled with 0.25 g of adsorbent. The elution process was carried out after a residence time ranging from 2 to 20 minutes, and the removal rate was evaluated. The removal rate was found to be essentially stable throughout the various contact times examined. This shows that the contact period had a negligible effect on the dye removal.
Effect of Flow Rate
The effect of the flow rate of dye on removal efficiency was examined while keeping the adsorbent weight constant at 0.25 g. Flow rates ranging from 0.5 mL/min to 5 mL/min were evaluated. At the flow rates between 0.5 and 1.0 mL/min, an almost consistent percentage of adsorption of CV, MB, and MG was observed. According to the findings, as the flow rate increased, the proportion of dye elimination reduced. At greater flow rates, the contact period between the dye molecules and the adsorbent is shortened. We established that the optimized flow rate for the dye was 1 mL/min, as it was observed after careful consideration of the experimental findings. However, higher flow rates of 5 mL/min and above were found to make liquid elution through the column challenging. Fig. 7 depicts the fluctuations observed during the adsorption of CV, MB, and MG at various flow rates.
Chemical stability of adsorbent
The chemical stability of NaOH-LLPh was assessed across a range of organic and acidic solvents. This evaluation involved the use of solvent-washed and sun-dried material. The solvents employed in the treatment included methanol, ethanol, acetone, diethyl ether, methyl acetate, methyl benzoate, 0.1 M solutions of HCl, HNO3, HClO4, acetic acid, oxalic acid, and tartaric acid. The experimental investigation demonstrated that the adsorption capacity of NaOH-LLPh for MB, MG, and CV remained unaffected when utilizing the adsorbent treated with methanol, ethanol, acetone, diethyl ether, methyl acetate, methyl benzoate. The average percentage adsorption values for MB, MG, and CV on NaOH-LLPh post-treatment with the solvents were found to be 96.23, 97.52, and 87%, respectively, with corresponding relative standard deviations of 2.13, 1.89, and 1.88%. However, the adsorption capacity of NaOH-LLPh adsorbent treated with 0.1 M solutions of HCl, HNO3, HClO4, acetic acid, oxalic acid, and tartaric acid declined very marginally. The mean percent adsorption of MB, MG, and CV observed were 91.45, 86.45 and 81.26%, respectively. This provided conclusive evidence of the exceptional chemical stability of the adsorbent in these solvents.
Adsorption isotherm studies
Adsorption isotherm studies provide information on the interaction between adsorbent and adsorbate [73], as well as the underlying process. Attempts were made to investigate the adsorption of MB, MG, and CV on the adsorbent’s surface. Adsorption isotherm analysis determines the relationship between the concentration and amount of dye accumulated on the surface of a certain amount of adsorbent under equilibrium conditions. To explain the adsorption process mathematically, the Langmuir and Freundlich adsorption isotherm models were used. According to the Langmuir isotherm model, the adsorbent surface is created with a single layer on the adsorbent’s outer surface [43]. It confirms that the adsorbent surface has uniform adsorption sites, i.e., a homogeneous surface. Hence, the energy required for dye adsorption is uniform over the adsorbent’s surface [73]. The Freundlich isotherm model, on the other hand, explains the non-uniform structure of the adsorbent’s heterogeneous outer surface [67]. As a result, the energy required to adsorb the dye on the adsorbent’s surface varies across the surface. The following is the mathematical expression for the Langmuir isotherm model [73]:
where Ce is the equilibrium concentration of dye (mmol/L), qe is the amount of dye adsorbed per unit weight of adsorbent, qm is the adsorption of dye on unit weight of adsorbent at a time (t) or adsorption capacity (mmol/g) or monolayer capacity. The terms b and qm were determined from the intercept and slope of the Ce/qe v/s Ce plot, respectively.
The mathematical equation for Freundlich isotherm model is given below [68]:
log (qe) = log (kf) + log (Ce) / n (7)
where kf and n are empirical parameters that reflect sorption capacity and sorption intensity, respectively. These parameters are computed by plotting log(qe) versus log(Ce). If 1/n is less than one, it implies normal adsorption; n = 1 indicates that the partition between the two phases is independent of concentration; and 1/n more than one shows cooperative adsorption.
The Langmuir isotherm was described in terms of a dimensionless constant called the separation factor (RL), as indicated in the equation below [73]:
where C0 is the initial dye concentration and b is the Langmuir constant. The separation factors (RL) are shown in Table 4. It is well understood that if the RL value is more than one, adsorption is very low or negligible, RL=1: adsorption is linear, and RL<1: adsorption is outstanding. Tables 4 and 5 present the experimental outcomes of the study. Figs. 8 and 9 (provided as supplementary file: SF) show the experimental Langmuir and Freundlich adsorption isotherm graphs, respectively.
By analyzing the R2 and RL data in Tables 4 and 5, it is clear that MB and MG are well suited to the Langmuir isotherm model, but CV is well fitted with the Freundlich isotherm model.
Kinetics studies
The kinetic studies were carried out under optimal conditions, which included passing 1000 mg/L MB and MG dyes, and 50 mg/L CV through a 0.25g packed adsorbent column with a contact time ranging from 20 to 100 min, eluted at a flow rate of 1 ml/min, and taking absorbance measurements of the eluate. These investigations sought to study the dye’s interaction with the adsorbent over time as well as its efficiency [44]. To undertake kinetic analyses on experimental data, mathematical equations were employed to determine the mechanism of adsorption and its rate-controlling or rate-determining steps, which can differentiate between mass transport and chemical reaction. Using experimental data, pseudo-first order and pseudo-second order models were used to determine the kinetics of adsorption and this provided insights into the rate constants and mechanism. The regression coefficient or correlation coefficient (R2) and experimental qe values were used to determine the applicability of the kinetic models.
The following is the mathematical expression for the pseudo-first-order model [73]:
where qe is the amount of dye adsorbed (mg/L) at equilibrium, qt is the amount of solute adsorbed per unit weight of adsorbent at time t (mg/g), and k1 is the rate constant for the pseudo first-order model. The graph of log(qe-qt) against t can be used to find the values of k1 and qe using slope and intercept, respectively. Table 6 displays the findings of these studies.
The following is a mathematical representation of the pseudo-second-order model [73]:
where k2 is the pseudo-second-order model’s rate constant. The slope and intercept of t/qt versus t graph were used to derive the values of k2 and qe, respectively. Fig. 10 and 11 (in SF) depict the corresponding kinetic models. Table 7 summarises the acquired results of this study.
The experimental data reported in Tables 6 and 7 for k1, k2, and R2 indicate that the adsorption of MB, MG, and CV dyes followed the pseudo-second-order kinetics model.
Application to removal of dyes from spiked industrial effluent samples
The proposed adsorbent was employed to extract CV, MB, and MG from industrial effluent water samples spiked with known amounts of standard solutions. Table 8 shows the percentage of dye estimated after adsorption. The results show that utilizing NaOH-LLPh adsorbent, the dyes CV, MB, and MG can be removed up to 87.98%, 97.63%, and 99.10%, respectively.
Thermodynamic studies
The effect of temperature on the adsorption of MB, MG, and CV was studied. The MB and MG were taken at 600 mg/L and CV at 40 mg/L. The dye adsorption and elution procedure was performed for each dye separately at 298, 303, 308, and 313 K. The changes in Gibbs free energy (ΔG0), entropy (ΔS0), and enthalpy (ΔH0) were determined [74] using the Van’t Hoff equation. The results of thermodynamic studies are presented in Table 9. The results of negative ΔG0 values and positive ΔH0 values indicated the spontaneous endothermic process of adsorption.
Adsorption mechanism
The primary challenge in a study on adsorption lies in comprehending the adsorption mechanism. Nevertheless, before grasping this mechanism, it’s essential to take into account two factors: firstly, the structure of the adsorbate, and secondly, the surface properties of the adsorbent. It’s worth noting that MB, MG, and CV are cationic dyes containing amine groups within their composition. In aqueous solutions, the dyes dissociate into MB+, MG+, CV+ ions, and Cl− ions [37, 61]. Treatment with NaOH alters the morphology of the adsorbent, eliminates hemicelluloses and lignin components, reduces silica content, and likely increases the crystallinity of the cellulose fraction. This may promote chemical interactions between the more exposed hydroxyl groups of the adsorbent and the dye ions. The mechanical bonding takes place more readily as the dye ions penetrate the adsorbent microstructure [37, 61].
Therefore, the adsorption of the studied dye onto the surface of the adsorbent, which is the NaOH-activated Lablab Pupureus husk (NaOH-LLPH), likely occurs because of the creation of surface hydrogen bonds between the hydroxyl groups of the sorbent and the nitrogen atoms within the dyes. The mechanism for a dye-hydrogen ion exchange process is outlined as follows [37, 61]:
NaOH-LLPH – OH = NaOH-LLPH – O− + H+
NaOH-LLPH – O− + Dye+ = NaOH-LLPH – O− - Dye+ + H+.
CONCLUSION
The chroma trap procedure was used to remove three ionic organic dyes, CV, MB, and MG using a novel bio adsorbent, NaOH-activated Lablab purpureus husk, which is one of the most commonly available natural materials. Adsorption capacity was found to be increased in the alkali-treated adsorbent. The pictures of SEM analysis inferred the probable adsorption of dyes onto the surface of the natural biodegradable adsorbent. The adsorption surface and numerous parameters, including dye concentration, adsorbent dose, dye pH, contact time, and flow rate, were evaluated, and optimized, and were found free from complexities. The findings of Langmuir and Freundlich’s adsorption isotherm experiments are described, and they show that CV, MB, and MG adsorb well on the adsorbent. Kinetic studies revealed the pseudo-second-order adsorption model on the material. This first-ever and novel column chromatographic adsorption approach is found to be the most cost-effective way to remove CV, MB, and MG. The proposed adsorption method for spiked water samples produced outstanding findings that correlated with those that evolved to a greater degree of agreement. A comparative evaluation of the effectiveness of previously documented techniques for eliminating MB, MG, and CV using various husks as adsorbents has been carried out. As outlined in Table 1, it is clear that the use of NaOH-activated Lablab purpureus husk in this investigation surpasses other studies that employed different husks as adsorbents for the elimination of MB, MG, and CV. This superior performance can be attributed to its exceptional adsorption capacity, straightforward elution process, and uncomplicated removal mechanism. Despite recent advancements in the field of nanocomposites for sono-sorption [75, 76], this straightforward adsorption method for dye removal should be prioritized. This novel adsorbent holds promise for a wide range of industries seeking to remove harmful organic dyes from their wastewater.
CONFLICT OF INTEREST
The authors hereby declare that there is no conflict of interest.
- Celia, M.P and S. Suruthi, 2016. Textile Dye Degradation Using Bacterial Strains Isolated from Textile Mill Effluent. International Journal of Applied Research, 2 337-341.
- Shindhal, T., P. Rakholiya, S. Varjani, A. Pandey, H. H. Ngo, W. Guo, H. Y. Ng and M. J. Taherzadeh, 2021. A Critical Review on Advances in the Practices and Perspectives for the Treatment of Dye Industry Wastewater. Bioengineered, 12: 70-87. https://doi.org/10.1080/21655979.2020.1863034
- Adane, T., A. T. Adugna and E. Alemayehu, 2021. Textile Industry Effluent Treatment Techniques. Journal of Chemistry, 2021: 5314404.
https://doi.org/10.1155/2021/5314404 - Shokri, A., 2021. Using NiFe2O4 as a Nano Photocatalyst for Degradation of Polyvinyl Alcohol in Synthetic Wastewater. Environmental Challenges, 5: 100332.
https://doi.org/10.1016/j.envc.2021.100332 - Shokri, A., 2020. Using Mn Based on Lightweight Expanded Clay Aggregate (Leca) as an Original Catalyst for the Removal of NO2 Pollutant in Aqueous Environment. Surfaces and Interfaces, 21: 100705.
https://doi.org/10.1016/j.surfin.2020.100705 - Saghi, M., A. Shokri, A. Arastehnodeh, M. Khazaeinejad and A. Nozari, 2018. The Photo Degradation of Methyl Red in Aqueous Solutions by Α-Fe2O3/SiO2 Nano Photocatalyst. Journal of Nanoanalysis, 5(3): 163-70.
https://doi.10.22034/JNA.2018.543608.
- Shokri, A., and M. S. Fard, 2023. Using α-Fe2O3/SiO2 as a Heterogeneous Fenton Catalyst for the Removal of Chlorophenol in Aqueous Environment: Thermodynamic and Kinetic Studies. International Journal of Environmental Science and Technology, 20: 383-396.
https://doi.org/10.1007/s13762-022-04498-w - A. Shokri, 2022. Degradation of 4-Chloro Phenol in Aqueous Media Thru UV/Persulfate Method by Artificial Neural Network and Full Factorial Design Method. International Journal of Environmental Analytical Chemistry, 102(17): 5077-5091.
https://doi.org/10.1080/03067319.2020.1791328 - Adegoke, K. A., and O. S. Bello, 2015. Dye Sequestration using Agricultural Wastes as Adsorbents. Water Resources and Industry, 12: 8-24. https://doi.org/10.1016/j.wri.2015.09.002
- Dassanayake, R. S., S. Acharya and N. Abidi, 2021. Recent Advances in Biopolymer-Based Dye Removal Technologies. Molecules, 26(15): 4697. https://doi.org/10.3390/molecules26154697
- Al-Degs, S. Y., and A. J. Sweileh, 2012. Simultaneous Determination of Five Commercial Cationic Dyes in Stream Waters Using Diatomite Solid-Phase Extractant and Multivariate Calibration. Arabian Journal of Chemistry, 5(2): 219-224. https://doi.org/10.1016/j.arabjc.2010.08.016
- Karić, N., A. S. Maia, A. Teodorović, N. Atanasova, G. Langergraber, G. Crini, A. R. L. Ribeiro and M. Đolić, 2022. Bio-Waste Valorisation: Agricultural Wastes as Biosorbents for Removal of (In)Organic Pollutants in Wastewater Treatment. Chemical Engineering Journal Advances, 9: 100239.
https://doi.org/10.1016/j.ceja.2021.100239 - Namasivayam, C., D. Sangeetha and R. Gunasekaran, Removal of Anions, 2007. Heavy Metals, Organics and Dyes from Water by Adsorption onto a New Activated Carbon from Jatropha Husk, An Agro-Industrial Solid Waste. Process Safety and Environmental Protection, 85: 181-184.
https://doi.org/10.1205/psep05002 - Aryee, A.A., R. Zhang, H. Liu, R. Han, Z. Li and L. Qu, 2020. Application of Magnetic Peanut Husk for Methylene Blue Adsorption in Batch Mode. Desalination and Water Treatment, 194: 269-279.
https://doi.org/10.5004/dwt.2020.25862 - Ibrahim, A. A., E. A. Zayid, S. A. Dhikeel and M. Y. Najem, 2022. Biosorption Removal of Methylene Blue Dye from Aqueous Solutions using Phosphoric Acid-Treated Balanites Aegyptiaca Seed Husks Powder. Biointerface Research in Applied Chemistry, 12(6): 7845-7862.
https://doi.org/10.33263/BRIAC126.78457862 - Ashrafi, S. D., H. Kamani and A. H. Mahvi, 2016. The Optimization Study of Direct Red 81 and Methylene Blue Adsorption on NaOH-Modified Rice Husk. Desalination and Water Treatment, 57(2): 738-746.
https://doi.org/10.1080/19443994.2014.979329 - Abhay, P. R., V. Kumar and D. P. Singh, 2020. A Combined Effect of Adsorption and Reduction Potential of Biochar Derived from Mentha Plant Waste on Removal of Methylene Blue Dye From Aqueous Solution. Separation Science and Technology, 55(5): 907-921.
https://doi.org/10.1080/01496395.2019.1580732 - Pankaj, S., R. Kaur, C. Baskar and W-J. Chung, 2010. Removal of Methylene Blue from Aqueous Waste using Rice Husk and Rice Husk Ash. Desalination, 259(1-3): 249-257. https://doi.org/10.1016/j.desal.2010.03.044
- Leshan, U., C. Thambiliyagodage, R. Wijesekera and M. G. Bakker, 2021. Synthesis of Mesoporous Silica Nanoparticles Derived from Rice Husk and Surface-Controlled Amine Functionalization for Efficient Adsorption of Methylene Blue from Aqueous Solution. Current Research in Green and Sustainable Chemistry, 4: 100116.
https://doi.org/10.1016/j.crgsc.2021.100116 - Weihua, Z., K. Li, H. Bai, X. Shi and R. Han, 2011. Enhanced Cationic Dyes Removal from Aqueous Solution by Oxalic Acid Modified Rice Husk. Journal of Chemical Engineering Data 56(5): 1882.
https://doi.org/10.1021/je100893h - Praveen, S., J. Josephraj, B. P. Thillainayagam and G. Ravindiran, 2023. Evaluation of the Adsorptive Removal of Cationic Dyes by Greening Biochar Derived from Agricultural Bio-Waste of Rice Husk. Biomass Conversion Biorefinery, 13: 4047-4060.
https://doi.org/10.1007/s13399-021-01415-y - Sharma, Y. C., Uma and S. N. Upadhyay, 2011. An Economically Viable Removal of Methylene Blue by Adsorption on Activated Carbon Prepared from Rice Husk. The Canadian Journal of Chemical Engineering, 89(2): 377-383.
https://doi.org/10.1002/cjce.20393 - Stevens, A. O., U. N. Emeh and N. O. Eddy, 2018. Experimental and Computational Chemistry Studies on the Removal of Methylene Blue and Malachite Green Dyes from Aqueous Solution by Neem (Azadirachta Indica) Leaves. Journal of Taibah University for Science, 12(3): 255-265.
https://doi.org/10.1080/16583655.2018.1465725 - Thi, H. T., H.H. Le, T. H. Pham, D. T. Nguyen, D.D. La, S.W. Chang, S.M. Lee, W.J. Chung and D.D. Nguyen, 2021. Comparative Study on Methylene Blue Adsorption Behavior of Coffee Husk-Derived Activated Carbon Materials Prepared using Hydrothermal and Soaking Methods. Journal of Environmental Chemical Engineering. 9(4): 105362.
https://doi.org/10.1016/j.jece.2021.105362 - Karaçetin, G., S. Sivrikaya and M. Imamotlu, 2014. Adsorption of Methylene Blue from Aqueous Solutions by Activated Carbon Prepared from Hazelnut Husk Using Zinc Chloride. Journal of Analytical and Applied Pyrolysis, 110(1): 270-276.
https://doi.org/10.1016/j.jaap.2014.09.006 - Mohd, A. A., B. A. Nur'Adilah, A. A. Kayode and S. B. Olugbenga, 2019. Sorption Studies of Methyl Red Dye Removal Using Lemon Grass (Cymbopogon Citratus). Chemical Data Collections, 22: 100249.
https://doi.org/10.1016/j.cdc.2019.100249 - Shubhada, S. N., N. A. Mirgane, V. S. Shivankar, K.B. Pathade and G.C. Wadhawa, 2020. Adsorption of Methylene Blue Dye Over Activated Charcoal from the Fruit Peel of Plant Hydnocarpus Pentandra. Materials Today Proceedings, 37: 2302-2305. https://doi.org/10.1016/j.matpr.2020.07.728
- Amanda, R., O. Pezoti, L. S. Souza, I.P.A.F. Souza, K. C. Bedin, P. S. C. Souza, T. L. Silva, S. A. R. Melo, A. L. Cazetta and V. C. Almeida, 2017. Hydrothermal Carbonization of Coffee Husk: Optimization of Experimental Parameters and Adsorption of Methylene Blue Dye. Journal of Environmental Chemical Engineering, 5(5): 4841-4849.
https://doi.org/10.1016/j.jece.2017.08.035 - Warda, H., U. Farooq, M. Ahmad, M. Athar and M.A. Khan, 2017. Potential Biosorbent, Haloxylon Recurvum Plant Stems, for the Removal of Methylene Blue Dye. Arabian Journal of Chemistry, 10: S1512.
https://doi.org/10.1016/j.arabjc.2013.05.002 - Indrajit, G., S. Kar, T. Chatterjee, N. Bar, S.K. Das, 2021. Removal of Methylene Blue from Aqueous Solution using Lathyrus Sativus Husk: Adsorption Study, MPR and ANN Modelling. Process Safety and Environmental Protection, 149: 345-361.
https://doi.org/10.1016/j.psep.2020.11.003 - Chosel, P. L and R. E. C. Amon, 2020. Magnetic Rice Husk Ash "Cleanser" as Efficient Methylene Blue Adsorbent. Environmental Engineering Research, 25(5): 685-692.
https://doi.org/10.4491/eer.2019.287 - Anees, A., N. Khan, B.S. Giri, P. Chowdhary and P. Chaturvedi, 2020. Removal of Methylene Blue Dye Using Rice Husk, Cow Dung and Sludge Biochar: Characterization, Application, and Kinetic Studies, Bioresource and Technology, 306: 123202.
https://doi.org/10.1016/j.biortech.2020.123202 - Oana, M. P., C. Pecurariu and S. G. Muntean, 2014. Kinetic and Thermodynamic Studies on Methylene Blue Biosorption using Corn-Husk, Royal Society of Chemistry, 4(107): 62621-62630.
https://doi.org/10.1039/C4RA10504D - Kruti, V and D. Dharajiya, 2019. Adsorptive Removal of Six Dyes from Aqueous Solution using Normal and Modified Sawdust of Gmelina Arborea Roxb. International Journal of Pharmacy and Biological Sciences, 9(2): 179-186. https://doi:10.21276/ijpbs.2019.9.2.25
- Gobi, K., M. D Mashitah and V. M. Vadivelu, 2011. Adsorptive Removal of Methylene Blue using a Novel Adsorbent from Palm Oil Mill Effluent Waste Activated Sludge: Equilibrium, Thermodynamics, and Kinetic Studies. Chemical Engineering Journal, 171(3): 1246-1252.
https://doi.org/10.1016/j.cej.2011.05.036 - Siji, C., S. Tang, Y. Sun, G. Wang, H. Chen, X. Yu, Y. Su and G. Chen, 2018. Preparation of a Highly Porous Carbon Material Based on Quinoa Husk And its Application for Removal of Dyes by Adsorption. Materials (Basel), 11(8): 1407.
https://doi.org/10.3390/ma11081407 - Binod, K and U. Kumar, 2015. Adsorption of Malachite Green in an Aqueous Solution Onto Sodium Carbonate Treated Rice Husk. Korean Journal of Chemical Engineering, 32(8): 1655-1666. https://doi.org/10.1007/s11814-014-0351-5
- Adugna, N. A., G. Y. Abate and A. T. Habte, 2020. Bioadsorption of Basic Blue Dye from Aqueous Solution onto Raw and Modified Waste Ash as Economical Alternative Bioadsorbent, Journal of Chemistry, 2020: Article ID: 8746035.
https://doi.org/10.1155/2020/8746035 - Malik, R., D.S. Ramteke and S.R. Wate, 2007. Adsorption of Malachite Green on Groundnut Shell Waste Based Powdered Activated Carbon, Waste Management, 27(9): 1129-1138.
https://doi.org/10.1016/j.wasman.2006.06.009 - Abdoulaye, D. N., Y.A.E.H. Ali, O.E.M. Abdallahi, M.A. Bollahi, M. Stitou, M. Kankou and D. Fahmi, 2020. Sorption of Malachite Green from Aqueous Solution using Typha Australis Leaves as a Low-Cost Sorbent. Journal of Environmental Treatment Techniques, 8(3): 1023-1028.
- Mohd, A. A., N. Ahmad and O. S. Bello, 2014. Adsorptive Removal of Malachite Green Dye using Durian Seed-Based Activated Carbon. Water Air Soil Pollution, 225(8): 2057.
https://doi.org/10.1007/s11270-014-2057-z - Ravikumar, M and E. G. Aklilu, 2017. A Comparative Study on Removal of Malachite Green Dye from Waste Water using Different Low Cost Adsorbents, International Journal of Engineering Research and Technology, 6(04): 262.
https://doi.org/10.17577/IJERTV6IS040338 - Farnaz, H., R. Norouzbeigi, F. Sarbisheh and H. Shayesteh, 2016. Malachite Green Removal using Modified Sphagnum Peat Moss as A Low-Cost Biosorbent: Kinetic, Equilibrium, and Thermodynamic Studies, Journal of Taiwan Institute of Chemical Engineers, 58: 482-489.
https://doi.org/10.1016/j.jtice.2015.07.004 - Manoj, D and C. Mishra, 2019. Jackfruit Leaf as an Adsorbent of Malachite Green: Recovery and Reuse of the Dye, SN Applied Science. 1(5): 483.
https://doi.org/10.1007/s42452-019-0459-7 - Arora, V and D. P. Tiwari, 2020. Remediation of Colour Pollutant Malachite Green Dye with Mesoporous Walnut Based Particles as Efficient Adsorbent: Isotherms, Kinetics and Batch Adsorption Equilibrium Study. Indian Journal of Environmental Protection, 40(3): 227-234.
- Hejun, R., R. Zhang, Q. Wang, H. Pan and Y. Wang, 2016. Garlic Root Biomass as Novel Biosorbents for Malachite Green Removal: Parameter Optimization, Process Kinetics and Toxicity Test. Chemical Research in Chinese Universities, 32(4): 647-654.
https://doi.org/10.1007/s40242-016-6095-5 - Fouzia, M and A. Nasar, 2019. Preparation, Characterization, and Adsorption Studies of the Chemically Modified Luffa Aegyptica Peel as a Potential Adsorbent for the Removal of Malachite Green from Aqueous Solution. Journal of Molecular Liquids, 274: 315-327.
https://doi.org/10.1016/j.molliq.2018.10.119 - Arpita, G., 2021. Application of Cocos Nucifera's Husk to Remove Malachite Green Dye and Response Surface Modeling. Bulgarian Chemical Communications, 53: 44. https://doi:10.34049/bcc.53.D.28
- Neetu, S., D. P. Tiwari and S. K. Singh, 2020. Sorption Potential of Treated Plant Residues viz. Potato Peel and Neem Bark for Removal of Synthetic Dyes from Aqueous Solution. Rasayan Journal of Chemistry, 13(2): 1063-1073.
https://doi.org/10.31788/RJC.2020.1325405 - Mohamad, F. M. Y., M. A. Ahmad, N. A. Rosli, F. N. Gonawan and S. J. Abdullah, 2021. Scavenging Malachite Green Dye from Aqueous Solution using Durian Peel Based Activated Carbon. Malaysian Journal of Fundamental and Applied Sciences, 17(1): 95.
https://doi.org/10.11113/mjfas.v17n1.2173 - Mouhammad, H., P. Parthasarathy, H. R. Mackey, T. Al-Ansari and G. McKay, 2021. Minimizing Adsorbent Requirements using Multi-Stage Batch Adsorption for Malachite Green Removal using Microwave Date-Stone Activated Carbons. Chemical Engineering and Processing - Process Intensification, 167: 108318.
https://doi.org/10.1016/j.cep.2021.108318 - Dilli, D and P. L. Homagai, 2020. Adsorptive Removal of Malachite Green Dye from Aqueous Solution using Chemically Modified Charred and Xanthated Wheat Bran. Journal of Nepal Chemical Society, 41(1): 103-109.
https://doi.org/10.3126/jncs.v41i1.30495 - Vijayalakshmi, G and S. Sivajiganesan, 2020. Removal of Malachite Green Dye by Teak Wood Waste Biomass Activated Carbon and its Antimicrobial Activity. European Journal of Medicinal Plants, 31(6): 18-25.
https://doi.org/10.9734/ejmp/2020/v31i630244 - Sudarni, D. H. A., U. O. Aigbe, K. E. Ukhurebor, R. B. Onyancha, H. S. Kusuma, H. Darmokoesoemo, O. A. Osibote, V. A. Balogun and B. A. Widyaningrum, 2021. Malachite Green Removal by Activated Potassium Hydroxide Clove Leaf Agrowaste Biosorbent: Characterization, Kinetic, Isotherm, and Thermodynamic Studies. Adsorption Science and Technology, 2021: Article 1145312
https://doi.org/10.1155/2021/1145312 - Bamgbose, J. T., B. A. Adesina, N. M. Olubunmi, A. S. Akinyeye and I. A. Ayodeji, 2012. Kinetics and Thermodynamic Studies of Adsorption of Malachite Green onto Unmodified and EDTA-Modified Groundnut Husk. African Journal of Pure and Applied Chemistry, 6(14): 141.
https://doi:10.5897/AJPAC12.047
- Chrispine, M. O., J. M. Onyari, W. C. Wanyonyi, J. N. Wabomba and V. M. Muinde, 2020. Adsorptive Removal of Hazardous Crystal Violet Dye from Aqueous Solution using Rhizophora Mucronata Stem-Barks: Equilibrium and Kinetics Studies. Environmental Chemistry and Ecotoxicology, 2: 64-72.
https://doi.org/10.1016/j.enceco.2020.05.001 - Khoiria, N. A. P., S. Kaewpichai, A. Keereerak and W. Chinpa, 2021. Facile Green Preparation of Lignocellulosic Biosorbent from Lemongrass Leaf for Cationic Dye Adsorption. J Pol Env. 29(6): 1681-1693.
https://doi.org/10.1007/s10924-020-02001-5 - Ashish, K. N and A. Pal, 2021. Enhanced Adsorption of Gentian Violet Dye from Water using Lignocellulosic Agricultural Waste Modified With Di- and Tri-Carboxylic Acids: Artificial Intelligence Modeling, Practical Comprehension, Mechanistic And Regeneration Analyses. Journal of Environmental Chemical Engineering, 9(4): 105578. https://doi.org/10.1016/j.jece.2021.105578
- Sumanjit, K., S. Rani and R. K. Mahajan, 2015. Adsorptive Removal of Dye Crystal Violet onto Low-Cost Carbon Produced from Eichhornia Plant: Kinetic, Equilibrium, and Thermodynamic Studies. Desalination and Water Treatment, 53(2): 543-556. https://doi.org/10.1080/19443994.2013.841109
- Sagnik, C., S. Chowdhury and P. D. Saha, 2013. Response Surface Optimization of a Dynamic Dye Adsorption Process: A Case Study of Crystal Violet Adsorption onto NaOH-Modified Rice Husk. Environmental Science and Pollution Research, 20(3): 1698-1705.
https://doi.org/10.1007/s11356-012-0989-7 - Sagnik, C., S. Chowdhury and P. D. Saha, 2011. Adsorption of Crystal Violet from Aqueous Solution onto NaOH-Modified Rice Husk. Carbohydrate Polymers, 86(4): 1533-1541.
https://doi.org/10.1016/j.carbpol.2011.06.058 - Babalola, A. O., O. C. Olawole, A. O. Ayeni and S. E. Sanni, 2020. African Star Apples Whole Seed Activated Carbon Powder as a Bio-Adsorbent of Crystal Violet Dye Removal from Aqueous Solution. Water Conservation Science and Engineering, 5(1-2): 97-114.
https://doi.org/10.1007/s41101-020-00088-4 - Satyaprasad, Y., A. I. Raju Ch and R. P. Jaganada, 2020. Isothermal and Kinetics of Biosportion Capacity of Crystal Violet Dye by using Red Algae Gelidium Pusillum, Journal of Critical Reviews, 7(05): 2477-2493.
- Hira, U., T. Javed, M. B. Taj and M. N. Haider, 2023. Adsorption of Crystal Violet Dye from Wastewater on Phyllanthus Emblica Fruit (PEF) Powder: Kinetic and Thermodynamic. International Journal of Environmental Analytical Chemistry, 1: In Press.
https://doi.org/10.1080/03067319.2023.2176229 - Youssef, H. M., S. M. El-Dafrawy, E. A. Badran and M. M. El-Defrawy, 2022. Adsorptive Performance of Chemically Treated Olive Pomace for the Removal of Crystal Violet from Aqueous Solutions: Characterisation, Optimisation, Regeneration and Isotherm Studies. International Journal of Environmental Analytical Chemistry 102(19): 8019-8036.
https://doi.org/10.1080/03067319.2020.1843647 - Aljeboree, A. M and A. F. Alkaim, 2019. Role of Plant Wastes as an Ecofriendly for Pollutants (Crystal Violet Dye) Removal from Aqueous Solutions. Plant Achieves, 19: 902-905.
- Sumaira, B., R. Rehman, T. Mahmud, S. Basharat and L. Mitu, 2020. Tartaric Acid-Modified Holarrhena Antidysenterica and Citrullus Colocynthis Biowaste for Efficient Eradication of Crystal Violet Dye from Water. Journal of Chemistry, 2020: Article ID 8862167.
https://doi.org/10.1155/2020/8862167 - Garmia, D and H. Zaghouane-Boudiaf, 2019. Urtica Dioica Leaves-Calcium Alginate as a Natural, Low-Cost, and Very Effective Bioadsorbent Beads in the Elimination of Dyes from an Aqueous Medium: Equilibrium Isotherms and Thermodynamic Studies, International Journal of Biological Macromolecules, 124: 915-921.
https://doi.org/10.1016/j.ijbiomac.2018.11.253 - Mauro C de, C. G., C. V. A. Carla, J. F. C. Jaciene, A. A. S. Sirlane, S. S. Hildo A dos, M. M. Suzyeth, F. Robson and W. B. B. Cícero, 2019. Expressive Removal Of Cationic Dyes (Methylene Blue and Crystal Violet) by Carnauba Straw (Copernicia Cerifera), The Pharmaceutical and Chemical Journal, 6(3): 37-52.
- Koushik, G., N. Bar, A. B. Biswas and S. K. Das, 2021. Elimination of Crystal Violet from Synthetic Medium by Adsorption using Unmodified and Acid-Modified Eucalyptus Leaves with MPR and GA Application. Sustainable Chemistry and Pharmacy, 19: 100370.
https://doi.org/10.1016/j.scp.2020.100370 - Hassanien, G., E. M. A. El-Monaem, A. S. Eltaweil and A. M. Omer, 2022. Efficient Removal of Noxious Methylene Blue and Crystal Violet Dyes at Neutral Conditions by Reusable Montmorillonite/NiFe2O4@Amine-Functionalized Chitosan Composite. Scientific Reports, 12(1): 15499.
https://doi.org/10.1038/s41598-022-19570-1 - Brini, L., A. Hsini, Y. Naciri, A. Bouziani, Z. Ajmal, K. H'Maida, A. Boulahya, M. Arahou, B. Bakiz, A. Albourine and M. Fekhaoui, 2021. Synthesis and Characterization of Arginine-Doped Heliotrope Leaves with High Clean-Up Capacity for Crystal Violet Dye from Aqueous Media. Water Science and Technology. 84: 2265-2277. https://doi.org/10.2166/wst.2021.446
- Alene, A. G., G. Y. Abate and A. T. Habte, 2020. Bioadsorption of Basic Blue Dye from Aqueous Solution onto Raw and Modified Waste Ash as Economical Alternative Bioadsorbent. Journal of Chemistry, 2020: Article ID 8746035. https://doi.org/10.1155/2020/8746035
- Gavioli, E., N. M. Maier, K. Haupt, K. Mosbach and W. Lindner, 2005. Analyte Templating: Enhancing the Enantioselectivity of Chiral Selectors upon Incorporation Into Organic Polymer Environments. Analytical Chemistry, 77: 5009-5018.
https://doi.org/10.1021/ac050407s - Eizi, R., T. R. Bastami, V. Mahmoudi, A. Ayati and H. Babaei, 2023. Facile Ultrasound-Assisted Synthesis of CuFe-Layered Double Hydroxides/g-C3N4 Nanocomposite for Alizarin Red S Sono-Sorption. Journal of the Taiwan Institute of Chemical Engineers, 145: 104844.
https://doi.org/10.1016/j.jtice.2023.104844 - Najafi, M., T. R. Bastami, N. Binesh, A. Ayati and S. Emamverdi, 2022. Sono-sorption versus Adsorption for the Removal of Congo Red from Aqueous Solution using NiFeLDH/Au Nanocomposite: Kinetics, Thermodynamics, Isotherm Studies, and Optimization of Process Parameters. Journal of Industrial and Engineering Chemistry, 116: 489-503.
https://doi.org/10.1016/j.jiec.2022.09.039