Document Type : Review Paper
Authors
1 Department of Chemistry College of Natural and Computational Sciences, Kebridehar University, Ethiopia
2 Department of Chemistry College of Natural and Computational Sciences, Mekdela amba University, Ethiopia
3 Department of Chemistry College of Natural and Computational Sciences, samara University, Ethiopia
4 Department of Chemistry College of Natural and Computational Sciences, University of gondar, Ethiopia
Abstract
A group of supramolecular solid materials known as "metal organic frameworks"(MOFs) are a type of hybrid networks made up of a variety of inorganic and organic linkers that are all tightly bonded to metal ions. These classes of compounds have a larger surface area with the benefit of variable pore sizes, a diverse structure, and a lovely appearance. They are promising materials for a range of applications because they are easy to develop and have consistent, fine-tunable pore structures. Controlled mixing of MOFs with functional materials is resulting in the development of new multifunctional composites and hybrids that display unique properties that outperform those of their component parts as a whole. The structural characteristics, classification, The most widely used and successful strategies for MOFs composite synthesis are presented like (Encapsulation method, Solvothermal method, Solution impregnation, and Click chemistry (reaction) method), numerous characterization techniques and their applications MOFs composite have all been covered in this review. Crystals with extremely high porosity and good thermal and chemical stability can be produced by carefully choosing the MOF ingredients. Because of these properties, MOFs composites can be used for a wide range of applications, including, sensing toxic chemicals, drugs, gases, and trace metals, components of foods and many more and also for detection of different materials. This is a rapidly developing interdisciplinary research area therefore to present the current situation of the field; this article has covered recent achievements as well as new avenues to investigate the future scope and uses of MOFs composites/hybride.
Graphical Abstract
Highlights
Keywords
INTRODUCTION
Metal-organic frameworks, often known as MOFs, have become a diverse family of crystalline materials with extremely high porosity (up to 90% free volume) and massive inner surface areas, exceeding 6 000 m2/g [1]. Since its initial definition in the 1990s, Metal-Organic Frameworks (MOFs), which consist of coupling units (metal ions or metal-oxo clusters) coordinated by organic ligands, have drawn a lot of interest [2, 3, 4]. Due to its potential for separation [5], adsorption [6], catalysis [7], chemical sensors [8], miniature electronics [9], optical materials [10], drug delivery [11], electrical and optoelectronic devices [12], and other applications, MOFs have gained significant attention by researchers. The three most commonly used orders of 3D porous covalent polymers are zeolites [13], metal-organic frameworks (MOFs) [14], and covalent organic frameworks (COFs) [15, 16]. Metal-organic frameworks are crystalline porous materials having regular network topologies made of metal ions (or metal clusters) and organic linkers. They are also known as porous coordination polymers (PCPs) [17]. MOFs may be produced utilizing a variety of unique synthetic techniques, including slow diffusion, hydrothermal (solvo-thermal) [18], electrochemical [19], mechanochemical [20], microwave-assisted heating [21], and ultrasound [22], depending on the final structures and features. The composite of MOF can be made in a variety of ways, including, encapsulation [23], impregnation [24], infiltration [25], solid grinding [26], coprecipitation [27], click reaction [28] , and more. Diverse preparation techniques can result in different MOF composite characteristics, which can increase the usefulness of composite materials [29, 30]. Numerous MOFs have been produced, and they have many beneficial properties, including (a) adjustable pore size; (b) ordered structure; (c) tunable diameter; (d) resistance to changes in their morphologies; (e) convenient and affordable processing conditions; (f) easy sample collection; (g) resistance to metal aggregation; (h) resistance to architecture disntegrat-ion; and (i) attachment of other substances inside the pores or on the surface [31, 32, 33]. Even though MOFs have a lot of benefits, they also have significant drawbacks that restrict their use, including low mechanical strength, poor chemical and hydrothermal stability, and a single function [34, 35]. As a result, MOF composites were designed to address these shortcomings. In comparison to pure MOFs, MOF composites display novel chemical and physical properties or improved performance because of the synergistic interaction between MOFs and functional materials [36, 37]. When compared to pure MOFs and other conventional materials, the use of MOF composites provides significant advantages and development potential examples according to Olorunyomi et al. report MOF composites are identified as advanced materials with potential for deployment in analytical devices for chemical and biochemical sensing applications [38], Ahmed and Jhung also reported the MOF composites adsorption applications [39] and Li et al. state that because of MOF-based membranes’ greater performance, they have garnered a lot of interest in separation applications [40].
CLASSIFICATION OF MOF AND MOF COMPOSITE
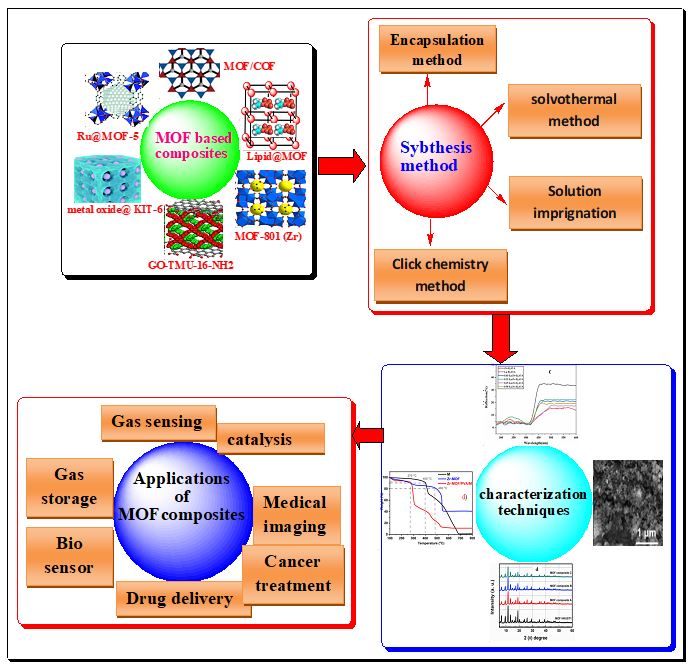
There are various kinds of MOFs, and these MOFs can integrate with other functional resources to develop MOF composites with a variety of functions (Fig. 1). Examples of these materials include metal NPs [41], metal oxides [42], quantum dots, carbon materials [43], molecules, polyoxometalates [44], polymers [45], and enzymes [46]. Depending on their constituent parts, the MOFs can be divided into a variety of categories. According to the functional materials that have been doped, the MOF composites are categorized.
Classification of MOFs
Iso-reticular MOFs (IRMOFs)
Reticular chemistry, a fundamental technique used to produce these kinds of materials, involves the connecting of molecular-scale building blocks into predetermined shapes that are recognized and ordered by strong bonding forces [47]. The primary building blocks of IRMOFs are secondary inorganic structural units [Zn4O]6+ and various aromatic carboxylic acid ligands [48]. Isoreticular MOFs (IRMOF), a subclass of MOFs, have a similar cubic topology to MOF-5 and are connected by linear organic linkages [49]. Due to its easy synthesis, effective gas adsorption, and useful storage properties, IRMOF-n (n = 1–16) has been the subject of significant research [50, 51].
Zeolitic imidazolate frameworks (ZIFs)
Zeolitic imidazolate frameworks (ZIFs) are a distinct family of metal-organic frameworks (MOFs) that maintain the basic chemical connection over wide variations in the network topology and related attributes. Because of their huge potential for hydrogen storage and carbon dioxide capture, ZIFs currently draw a great deal of attention [52]. There are different produced ZIFS including ZIF-5, ZIF-7, ZIF-8 [51, 52], ZIF-9 [53], ZIF-11, ZIF-71, ZIF-67 [54], ZIF-90 [55], ZIF-L [56], ZIF-95 [57], ZIF-100 [58], etc. Tetrahedral Si(Al)O4 units are chemically bonded by bridging O atoms to form more than 150 various forms of framework that makeup zeolite structures. In water, aqueous alkaline solution, and refluxing organic solvents, ZIFs exhibit exceptional chemical stability [59].
Coordination polymers (PCPs)
PCPs also known as Metal-organic frameworks (MOFs), are porous, polymeric (1D, 2D, or 3-D) materials linked together by coordination bonds between metallic ions and organic linkers [60]. In comparison to zeolite or activated carbon, PCPs have a better application potential as a result of their uniformity, permeability, large surface area, and performance factor. In addition to coordination bonds, PCPs also contain other weak contacts or electrostatic interaction bonds (H-bonds, -electron stacking, or van der Waals interaction), which gives them the potential to change their structural composition which is advantageous in separation applications. The use of Zn4O-based MOF compounds for fuel gas storage is widely recognized due to their high porosity and huge surface area [61].
Porous coordination networks (PCNs)
These are a novel class of nanoporous materials that possess good chemical and mechanical durability, huge surface areas, high porosities, and variable pore sizes [62]. Some examples of PCN-10, PCN-11 [62], PCN-12, PCN-14, PCN-15 [63], PCN-333 [64], PCN-224 [65], and PCN-222 [66], materials widely used as a sensor and for H2 storage due to their nanoscopic cages. It has been determined that several PCNs, including PCN-5, PCN-6 [67], PCN-610, PCN-46, PCN-68, PCN123, and PCN-124, are promising for CO2 removal [68, 69, 70]. Results show that PCN-59 exhibits the highest CO2/N2 selectivity among the various PCN frameworks taken into consideration [71].
Materials Institute Lavoisier (MILs)
MILs are porous metals of carboxylate salts with large pores and permanent porosity made up of various trivalent metal cations and carboxylic acid ligands [72]. MILs can serve as vehicles for drug delivery. Busulfan, azidothymidine triphosphate, doxorubicin, and cidofovir are examples of anti-tumor and anti-AIDS active compounds that could be collected by MIL series frameworks and returned in human organs such as the liver [73]. MIL materials include MIL-53(Fe) [74], MIL-88B (Fe) [75], MIL-68(Ga) [76], MIL-88A (H2O2) [77], MIL-125(Ti) [78], MIL-101(Cr) [79], Fe-MIL, Al-MIL [80], Cr-MIL [81], etc. Due to its permeable or flexible structure, MIL-53(Fe) is frequently used to remove pigments and hazardous chemicals from aqueous solutions [82]. An electrochemical sensor that can accurately detect the presence of PQ in a food sample was built using Fe2O3-MIL-100 [83].
Classification of MOF composites
MOF-metal nanoparticle composites
Due to their critical role in catalysis, metal nanoparticles (MNPs), especially small-size MNPs, are increasingly gaining significant attention. Unfortunately, Small MNPs are thermodynamically unstable and more likely to agglomerate during catalytic reactions because of their high surface energy, which results in a loss of efficiency [85]. MOFs are applied to modify these conditions. There are two alternative methods for producing MNPs inside MOFs. The primary and most popular technique involves using MOFs as stabilizing substrate components, which provide restricted areas that prevent agglomeration and control particle size. The process requires precursor infiltration in stages, followed by decomposition or reduction [86, 87]. The other technique involves encapsulating organic material, surfactants, and polymers that have stable prefabricated NPs in solution, followed by the addition of appropriate precursors to produce the framework around the prepared NPs [88]. The MNPs- MOF compounds formed using those methods including Pd NPs/MOFs [89], Au NPs/MOFs [90], Ru NPs/MOFs [91], Cu NPs/MOFs, Pt NPs/MOFs [92], Ni NPs/MOFs [93], Ag NPs/MOFs [94], etc are some examples (Fig. 2). High-performance anodes for lithium-ion batteries can be made using Si-MOFs [95]. Cu-MOF-GN/GCE shows effective anti-interference characteristics for the detection of H2O2 and AA in the presence of different carbohydrates [96].
MOF-carbon composites
Their large surface area, electrical properties, and high stability of carbon-based materials (CMs) make them excellent candidates for use as catalysts, energy storage systems, waste disposal, and drug delivery systems [97]. Because of their drawback in the field of sensing, separation, and adsorption MOF-carbon composite materials have received attention in recent times. As an example, smaller molecules like ammonia are challenging to remove from airstreams using activated carbon. Many findings confirmed that MOF-carbon composites are interesting materials for use in air purification [98]. The Co-MOF-MPC composite may be very promising for the designing of electrochemical environmental sensors because it shows excellent catalytic performance for the oxidation of hydrazine and reduction of nitrobenzene [99]. CeCu-MOF, synthesized by a hydrothermal process, is utilized as an electrode material in lithium-ion batteries and supercapacitors [100]. By using an in-situ growth technique, Ni-MOF@CNT material was produced on the GN substrate and used as a unique self-supporting composite for all-solid-state supercapacitors with a high energy density [101].
MOF-metallic compound composites
Metal oxide nanoparticles serve a crucial function, in the field of materials chemistry, medicine, agriculture, information technology, biomedical, optical, electronics, catalysis, environment, energy, and sensing [102]. A new technique (MOF-metallic compound composite) was developed to examine the uses and limitations of metal oxide nanoparticles. To produce composites with the best attributes, metallic compounds such as SiO2 [103], Fe3O4 [104], ZnO [105], CuO [106], CdS [107], and ZnS [108] have been doped into MOF materials. The in-situ solvothermal approach was used to produce Zr-based UiO-66-NH2 MOF for improved activity towards Cr (VI) adsorption and photocatalytic H2 evolution [109]. Metal-organic frameworks derived In2O3/ZnO synthesized using a straightforward co-precipitation process and thermal annealing in air can also be used as a highly sensitive H2S gas sensor [110]. The solvothermal approach is applied to synthesize bimetallic FeO/NiO MOF and rGO-based composites, which can also be used in the methanol oxidation reaction [111]. The adsorbent MgFe2O4@MOF was synthesized using a simple solvothermal approach to eliminate the organic dyes Rhodamine B (RB) and Rhodamine 6G (Rh6G) from water samples [112].
MOF-enzyme composites
Enzymes are flexible, environmentally friendly biological catalysts that have been widely used in the chemical, food, cosmetics, and pharmaceutical industries. Due to Low stability at higher temperatures and extreme pH values, slow recovery, and the challenge of reusing enzymes have all restricted its use [113]. The development of immobilization as a new technique for the stabilization, convenience of restoration, and persistent usage of enzymes. According to their method of synthesis, MOF-enzyme composites can be divided into four major categories: surface attachment, covalent coupling, pore entrapment, and co-precipitation [114]. The encapsulation of lipid into ZIF-67 MOF forms a lipase@ZIF-67 composite and this composite is used as a catalyst for the production of biodiesel. Lipid encapsulation in ZIF-67 greatly improved the enzyme’s heat stability, storage stability, and reusability [115]. GOx-ZIF-8 biocomposites are an excellent material for electrochemical biosensing applications for the detection of glucose and the composite was synthesized using a biomineralization-assisted in-situ encapsulation approach [116]. In general, the enzyme-MOF composites have greater application potential and have superior catalytic activity than free enzymes.
SYNTHESIS OF MOF COMPOSITE
Most of the problems related to everyday activities are solved by MOF, a fascinating and emerging science that is also making significant contributions to the advancement of technology. As technology develops, new synthetic techniques for MOF composites are continuously being developed. Some of the synthetic methods including mechanochemical, electrochemical, microwave, and sono-chemical methods, encapsulation, solution impregnation, solvothermal, one‑pot, and diffusion techniques.
Encapsulation method
The most popular and established method for producing MOF composites is encapsulation as shown in Fig. 3. The “build-bottle-around-ship” or “encapsulation method” is based on two key processes. (I) NPs are synthesized that are homogeneous in size, structure, and morphology. (II) Building MOF shells on previously produced NPs [120, 121]. Encapsulation allows for the incorporation of different morphologies and sizes of NPs, carbon compounds, molecular catalysts, enzymes, and other substances within MOF shells [122]. Pd@ZIF‑8 is a MOF‑composite material synthesized by the encapsulation method in a simple, rapid, and sustainable manner [123]. EY@Zr‑MOF composite was also synthesized by encapsulation of EY molecule and Pd on the surface of the ZIF-8 MOF [124].
Solvothermal method
A reaction mechanism in an enclosed system in the presence of a solvent at a temperature greater than that of such a solvent’s boiling point. This technique demonstrates the advantages of low cost and effective diffusion [125, 126]. An easy solvothermal technique was used to synthesize Fe-MOF/RGO composites with various RGO ratios. The precursor used for synthesis Fe-MOF/RGO was RGO, N, N-dimethylformamide (DMF), 1, 4- dicarboxybenzene, iron (III) chloride hexahydrate, and acetic acid glacial at a temperature of 150 oC for 2 h in Teflon-lined stainless autoclave (Fig. 4) [127]. The 6% weight of the CdS/MIL-101 catalyst was synthesized using the solvothermal method. In a Teflon-lined autoclave, MIL-101, DMSO, and C10H14CdO4 were combined and stirred for two hours [128].
Solution impregnation
The most popular and simple way to produce assisted catalysts for a range of heterogeneous reactions is the impregnation method [134, 135]. There are three fundamental steps. A substantial surface area support is first impregnated with a metal precursor solution, which is then condensed at a high temperature. Finally, the metal precursor solution is reduced in a suitable environment to make the catalysts [37, 136]. An Ag@MOF-5@chitosan composite was synthesized using the solution impregnation method (Fig. 5).
chitosan and silver into MOF-5 using a solution of 1 M sodium chloroacetate with 5% sodium hydroxide, DMF, and silver nitrate solution [137]. Pd@ MOF-5 was also synthesized by solution impregnation techniques. This composite was prepared by impregnating Pd into MOF-5 using palladium acetate or palladium acetylacetonate in absolute chloroform [138]. The solution impregnation is mostly used for preparing metal@MOF composite using the salt solution of the metal. The metal- MOF composite is prepared as follows in Fig. 6.
Click chemistry (reaction) methods
Sharpless and his friends first introduced click chemistry in 2001, which grew out of a desire to use molecular assembly’s capabilities for as many different types of applications as feasible (Fig. 7) [140,141]. Click chemistry is defined as a class of reactions that are quick, easy to perform, simple to refine, flexible, regiospecific, and produce large amounts of output [142,143,144]. Using click chemistry synthesis method Cu based MOF-composite was synthesized (Fig. 8) [145]. Zr-based MOF (UiO-68-triazole-A) was also synthesized by using a click reaction. The azide functional group in UiO-66-N3 was modified by phenylacetylene using the standard azide-alkyne click reaction. Triazole units produced as a result serve as effective fluorescence assays for metal ions [146, 147].
Characterization of MOF composite materials
The maximum amount of energy that the material absorbed (lamda max), electronic transition, band gap, functional group, morphology, topology, size and shape, stability, and the surface charge of the material was analyzed using different characterization techniques. The most commonly used instrument for characterizing MOF-composite was ICP, Solution NMR, UV-visible, FT-IR, XRD, SEM, TEM, Zeta‑potential, TGA, EDS, N2 adsorption, Cyclic voltammetry, etc. if further analysis is needed some other characterizing technique like XPS, XANES, EXAFS, SAXS spectroscopies can be applied.
UV-visible analysis of MOF-composites
From the Fig. 9 (a) below, the maximum wave length that PW11V@MIL-101 absorbed with the concentration of the composite 5 mg/L was 680 nm [148]. (b) Shows the maximum wave length absorbed for the composite (1) 1% TiO2/polymer composite, (2) 0.5%, (3) 1%, (4) 2% MIL-53(Cr)/polymer composites was 396, 515, 520, 572 nm respectively [149]. (c) The maximum absorption wavelengths of Zr‑H2BDC, La‑H2BDC, La/Zr‑H2BDC, La/Zr‑H2BDC, La/Zr-H2BDC, and La/Zr-H2BDC were 355, 334, 361, 443, 373, and 365 nm, respectively. La/Zr-H2BDC has a higher maximum wavelength relative to the other in the UV-visible region [150]. (d) the UV-visible spectra of PABA have a broad absorption band between 300 and 600 nm which can be attributed to the π-π ⃰ transition of the benzenoid rings and quinoid rings, respectively, for the polymer and composite [151].
SEM analysis of MOF-composites
The morphology of some of the selected MOF-composite materials is shown in Fig. 10, (a). The crystals of the original [Cu3(BTC)2(H2O)3]n sample are octahedral with a smooth surface and have an average size of 10 mm. (b) The surface of the magnetic MOF tends to be rougher after immobilization by Fe3O4‑Py [152]. (c) MIL‑125(Ti) composite with morphology in between ovoidal or flat cubes with round corners (d) MIL-53(Al) has flat cube morphology with very small particle size [153]. (e) and (f) Cu3(BTC)2.H2O, Fe3O4/Cu3(BTC)2–H2Dz has crystal losing structure [154].
XRD analysis of MOF-composites
The XRD pattern of different MOF-composites shown in Fig. 11 was analyzed as,
- a) due to a highly ordered structure of Ce-MOF@TEOS material a sharp peak was shown at 28.2°. Three other diffraction peaks at 32.3°, 47.8°, and 56.6°were displayed, respectively. The pattern of Ce‑MOF@TEOS and Ce-MOF are the same but the intensity of the peak in MOF@TEOS was strong indicating that the crystallite structure changed due to the presence of TEOS [155].
- b) There are no identifiable CsPbBr3 peaks in the XRD pattern of the CsPbBr3/MOF-5 composite. Because of the weak crystalline structure of CsPbBr3 PeQDs than MOF-5 and the small amount of PeQDs in the composite, the XRD pattern of CsPbBr3 PeQDs was covered by MOF-5. Due to this there, the XRD pattern of CsPbBr3/MOF-5 moves slightly to the small angle direction [156].
- c) In this the XRD peak of In2O3, MoS2, and In2O3/MoS2 composite was clearly seen. The characteristic peaks of In2O3 located at 2θ of 21.42°, 30.63°, 35.46°, 41.79°, 45.76°, 51.05° and 60.64°, which assigned to the (211), (222), (400), (332), (431), (440) and (622) planes of the cubic structured In2O3, respective. The peak for MoS2 occurred at 2θ of 14.31°, 32.98°, 39.59°, and 58.56° which were assigned to the s (002), (100), (103), and (110) planes respectively. The XRD pattern of the composite In2O3/MoS2 was all the peaks in In2O3 and MoS2 There is no additional peak shown indicating the purity of the material [157]. d) In this the peak of all the composite compounds (a, b, c) and the MOF (black) was the same. The intensity of MOF HKUST1 was strong compared to the composite this is due to the doped material (APTES NW s) in the composite decreasing the crystallinity of the material [158].
Thermogravimetric analysis (TGA) of MOF-composite
From the TGA curve shown in Fig. 12:
- a) the two DTG peaks below zero indicate the reaction that occurred was exothermic. The composite is thermally stable until the temperature reaches 300 °C. When the temperature reaches 300 °C the composite starts to loss its weight until the temperature becomes 440 °C due to the removal of water molecules. After that, the composite is thermally stable indicating pure Cu-MOF [159]. b) This composite loss it’s mass between the temperature range between 20-100 °C and 430-580 °C due to the removal of water and volatile matter. The thermal stability indicates the purity of the composite [160].
- c) Almost the material is thermally stable in all temperature ranges indicating the material is very pure [161].
- d) All the composites show weight loss but in comparison, Zr-MOF-PVA-M was less pure due to more conta mination during material doping [162].
Application of MOF-composite
Because of the properties of MOF-composite like crystalline porosity, reusability, selectivity, perform-ance, and stability, they are highly applicable for absorption separation [163]. They are mainly used for gas separation [164], oil-water separation [165], separation of linear and branched hydrocarbon [166], adsorption separation of heavy metals in water, dyes [167], and so on. MOF-composite has a wide application in different fields some of which are shown in Fig. 13.
Gas separation application
MOF-composite can be used for gas separation including separating CO2, purifying C2H4, purifying C3H8, separating noble gases, and separating isotopes. Mainly CO2 is separated from N2, CH4, and C2H2. Flue gas formed during combustion can cause the greenhouse effect. So highly selective MOF-composites like IL/MOF/POLYMER [168], cellulose@UiO-68-NH2 [169], ZIF-8/BMIM/SCN [170], etc. can be used. CO2 can be separated from methane. Methane is the most widely used chemical fuel and is the hydrocarbon with the lowest carbon content. After combustion, it contains the gas CO, CO2, NO, and SO2. Because of this MOF-composite can be used for separating those gases from methane [171]. For the manufacture of 1,4-butanediol, polymers, and polyurethanes as well as for welding and metal cutting, acetylene (C2H2) is a necessary precursor. During the production of acetylene main gases including CO2 exist as a byproduct. Removing CO2 is challenging because of its kinetic diameter. So MOF-composite is the best method to separate C2H2 and CO2 [172]. Those materials are also used to purify C2H4 by adsorption separation of contaminants from it. The primary steps in the purification of C2H4 are the separation of C2H2/C2H4, C2H6/C2H4, and C2H2/ C2H6/CO2/C2H4 [173]. Removing those impurities is the primary step in using C2H4 as a raw material in different industries, especially for the fabrication of polyethylene. Separating C2H6/C2H4 is challenging because some physical are similar like their boiling point and kinetic diameter. At this time, MOF is the best method to purify C2H4 [174]. The other gases purified and separated by MOF composite including C3H4/C3H6 [175], C3H4/C3H4/C3H6, and C3H6/C3H8 [176]. Purified forms of krypton (Kr) and xenon (Xe) have found extensive use in a variety of industries, including medical imaging, industrial lighting [177], insulation, lasers [178], illumination, and spaceship propellant [179]. Those noble gases are dangerous for the environment if they are released into the atmosphere. MOF-composite is applicable for Xe/Kr separation [180]. The materials are also important to separate gases with different separations called isotopic separation. The property and usage of those gases depends on their isotope. For example, isotopes of hydrogen have their property and application. MOF is important to separate isotopic gases like D2/H2 mixtures [181]. Separation of linear/branched alkane hydrocarbons (C5‑C6‑C7), cyclic C6 isomers (benzene/cyclohexane), cyclic C8 isomers (styrene/ethyl benzene, and xylene isomers), Xylene isomers’, and Styrene/ethyl benzene separation was done by MOF-composite. There is a persistent risk of leakages due to the transportation and storage of oil and petroleum products as a major source of energy around the world [182]. Because of this, the water surface is polluted by that oil. To remove the oil from the water it is costly. Due to this MOF-composite is the best method to remove oil from the water surface and it is also environmentally friendly during separation [183].
Wastewater treatment application
Water is the most vital source of all life activities but the growing number of contaminants that can pose intensified effects on both humans and aquatic life makes water pollution a more serious environmental issue [184,185]. Now adays dyes are used in a variety of industrial sectors, such as tanning leather, paper production, textiles, and printing. For instance, azo dyes are widely used in the textile industry to generate large volumes of colored organic and inorganic wastewater. Consequently, their treatment is essential [186,187]. In 2021, Shokri reported that the synthetic polymer Polyvinyl alcohol (PVA), which is used as a binder, shaping agent, and raw material in industries such as paper, textile, pharmaceutical, and membrane industries, may cause pollution issues if improperly handled upon release into the aqueous environment [188]. Waste water typically contains not only dyes it also contains heavy metals, which makes it difficult to remove both at once. Therefore, before wastewater is released into the environment, it is ideal if the materials employed for treatment can remove both dyes and heavy metals[189].
So, in an attempt to address this environmental issue, many researchers have focused their efforts on enhancing current technologies or offering a substitute plan. Therefore their superior large surface area, high porosity, and customizable properties of their structures and functions make metal-organic frameworks (MOFs) one of the appealing materials for this purpose [190].For example Shi et al. synthesized Cu-MOFs/Fe3O4 as adsorbent for removal of lead (Pb(II)) and malachite green (MG) in wastewater. The synthesized Cu-MOFs/Fe3O4 composite was found to be the potential adsorbent for waste water treatment [191]. In the same year, Ramezanalizadeh et al. also reported the Immobilization of BaWO4 nanostructures on a MOF-199-NH2 an efficient photocatalyst for the degradation of methyl orange (MO) and 4-Nitrophenol (4-NP) water contaminants and Fig. 14 depicted the mechanism of methyl orange and 4-nitrophenol degradation using the synthesezed composite. It’s noteworthy to observe that the absorption maximum of the composites with worse optical properties than the pure MOF has redshifted as a result of the immobilization of BaWO4 into the MOF-199-NH2. Furthermore, the MOF-199-NH2 has a bandgap of 3.2 eV, but the composite’s predicted bandgap is lower at 3.0 eV (Fig. 14). Thus, for methyl orange and 4-nitrophenol, the MOF-199-NH2/BaWO4 compound allowed for full degradation in 50 and 80 minutes, respectively [192].
Sensing application
Different chemo-sensory materials are developed for sensing application but MOF- the composite sensor solves the drawback that is not solved by chemo-sensory materials due to the properties of MOF-composite [193]. The property of MOF-composite includes a high porosity, a wide surface area, structural variety, flexibility, an exceptional capacity for adsorption, and chemical tenability [194]. The world’s population increases from time to time because of environmental pollution and related health risks increase. Due to this materials for sensing application can be developed [195]. The development of effective analytical methods for the detection of biomolecules in clinical [196], environmental [197], and industrial applications [198] has a great potential for MOF-based sensors. MOF-composite is used to sense biological macromolecules like proteins and nucleic acids (DNA and RNA) [199] and small molecules including amino acids [200], Lipids [201], fatty acids [202], glycolipids, sterols, monosaccharides [203], phenolic compounds, and alkaloids [204]. To detect chemicals in different fields chemical sensors and photochemical sensors can be applied. Different gases were detected by MOF-composite sensor including acetone, CO2, CS2, n-propanol, isopropyl alcohol, H2, pyridine, n-hexane, toluene, SO2, methanol, H2O2, ethanol, formaldehyde, etc [205, 206]. The application of MOFs in a variety of sensing applications, including the pH sensor, the detection of pesticide residues, the clinical diagnosis of diabetes, and photothermal therapy, shows significant promise [207]. For food safety, MOFs detail every use of MOFs in food safety monitoring, including sample preparation, separation, packaging, preservation, detection, and cleaning [208]. The MOF-composite and its adsorption separating materials and materials that can be detected by those sensors are listed briefly in Table 1.
CONCLUSION
The numerous research articles that have been published over the last two decades show how interesting MOFs are to both scientists and engineers. The interest in MOF-composites is due to their property including wide surface area, adjustable composition, highly flexible structures, and variable porosity. MOF-composite was made by metal ion/cluster with organic ligands with the introduction of dopants like metal, metal oxide, carbon-based materials, enzymes, etc on its surface. The most common synthesizing techniques used to produce MOF-composite were encapsulation, Solvothermal, Solution impregnation, Click chemistry (reaction), and so on. Some examples of MOF-composite synthesizing by those techniques like MOF-199/N, Co-TCPP(Fe), NH2-MIL-101(Al), DNA@ZIF-8, CA/ZIF-8@LAC/ MWCNTs/Au, etc. They are applicable for adsorption separation and as a sensor. MOF-composite is used to separate different gases like CO2/N2, CO2/CH4, C2H2/CO2, C2H2/C2H4, C2H6/C2H4, C2H2/C2H6/CO2/C2H4, C3H4/C3H6, C3H6/C3H8, noble gas separation and isotopic separation. MOF-composite material widely applicable for separation of oil/water mixture, Separation of linear/branched alkane hydrocarbons (C5‑C6‑C7), cyclic C6 isomers (benzene/cyclohexane), cyclic C8 isomers (styrene/ethyl benzene, and xylene isomers), Xylene isomers’, and Styrene/ethyl benzene separation was done by MOF-composite. They are also used as sensors for sensing different toxic chemicals, drugs, gases, trace metals, contents in foods, etc. dichlorophen, hydroquinone, catechol, Drug Idarubicin, tetracycline, Ammonia, H2S, H2O2, Al3+, glucose, etc. Generally, MOF composites are interesting materials with a wide area of application in many fields.
CONFLICTS OF INTEREST
The authors do not have any conflicts of interest.
- Zhou,H.C.,Long,J.R. and Yaghi,O.M.,2012. Introduction to metal organic frameworks. Chemical reviews, 112(2), pp.673-674.
https://doi.org/10.1021/cr300014x - Zhang, X., Wan, K., Subramanian, P., Xu, M., Luo, J. and Fransaer, J., 2020. Electroc-hemical deposition of metal organic framework films and their applications. Journal of materials chemistry A, 8(16), pp.7569-7587.
https://doi.org/10.1039/D0TA00406E - Gangu, K.K., Maddila, S., Mukkamala, S.B. and Jonnalagadda, S.B., 2016. A review on contemporary metal organic framework materials. Inorganica Chimica Acta, 446, pp.61-74.
https://doi.org/10.1016/j.ica.2016.02.062 - Li, J.R., Sculley, J. and Zhou, H.C., 2012. Metal organic frameworks for separations.Chemical reviews, 112(2), pp.869-932.
https://doi.org/10.1021/cr200190s - Salunkhe, R.R., Kaneti, Y.V. and Yamauchi, Y., 2017. Metal organic framework derived nanoporous metal oxides toward supercapacitor applications: progress and prospects. ACS nano, 11(6), pp.5293-5308.
https://doi.org/10.1021/acsnano.7b02796 - Huang, X., Huang, L., Arulmani, S.R.B., Yan, J., Li, Q., Tang, J., Wan, K., Zhang, H., Xiao, T. and Shao, M., 2022. Research progress of metal organic frameworks and their derivatives for adsorption of anions in water: A review. Environmental Research, 204, p.112381.
https://doi.org/10.1016/j.envres.2021.112381 - Valvekens, P., Vermoortele, F. and De Vos, D., 2013. Metal-organic frameworks as catalysts: the role of metal active sites. Catalysis Science & Technology, 3(6), pp.1435-1445.
https://doi.org/10.1039/c3cy20813c - Liu, D., Lu, K., Poon, C. and Lin, W., 2014. Metal-organic frameworks as sensory materials and imaging agents. Inorganic chemistry, 53(4), pp.1916-1924.
https://doi.org/10.1021/ic402194c - Allendorf, M.D., Schwartzberg, A., Stavila, V. and Talin, A.A., 2011. A roadmap to implementing metal-organic frameworks in electronic devices: challenges and critical directions. Chemistry A European Journal, 17(41), pp.11372-11388.
https://doi.org/10.1002/chem.201101595 - Wang, C., Zhang, T. and Lin, W., 2012. Rational synthesis of noncentrosymmetric metal-organic frameworks for second-order nonlinear optics. Chemical reviews, 112(2), pp.1084-1104.
https://doi.org/10.1021/cr200252n - Sun, C.Y., Qin, C., Wang, C.G., Su, Z.M., Wang, S., Wang, X.L., Yang, G.S., Shao, K.Z., Lan, Y.Q. and Wang, E.B., 2011. Chiral nanoporous metal‐organic frameworks with high porosity as materials for drug delivery. Advanced Materials, 23(47), pp.5629-5632.
https://doi.org/10.1002/adma.201102538 - Stavila, V., Talin, A.A. and Allendorf, M.D., 2014. MOF based electronic and opto electronic devices. Chemical Society Revie-ws, 43(16),pp.5994-6010.
https://doi.org/10.1039/C4CS00096J - Zhong,S.,Zhan,C.and Cao,D.,2015.Zeolitic imidazolate framework derived nitrogen doped porous carbons as high performance supercapacitor electrode materials. Carbon, 85, pp.51-59.
https://doi.org/10.1016/j.carbon.2014.12.064 - Waller,P.J.,Gándara,F. and Yaghi,O.M.,2015.Chemistry of covalent organic frameworks. Accounts of chemical research, 48(12), pp.3053-3063.
https://doi.org/10.1021/acs.accounts.5b00369 - Furukawa, H., Cordova, K.E., O’Keeffe, M. and Yaghi, O.M., 2013.The chemistry and applications of metal organic frameworks. Science, 341(6149), p.1230444.
https://doi.org/10.1126/science.1230444 - Aggarwal, V., Solanki, S. and Malhotra, B.D., 2022. Applications of metal-organic framework-based bioelectrodes. Chemical Science.
https://doi.org/10.1039/D2SC03441G - Yang, W., Li, X., Li, Y., Zhu, R. and Pang, H., 2019. Applications of metal organic‐framework‐derived carbon materials. Advanced materials, 31(6), p.1804740.
https://doi.org/10.1002/adma.201804740 - Ni, Z. and Masel, R.I., 2006. Rapid production of metal−organic frameworks via microwave-assisted solvothermal synthesis. Journal of the American Chemical Society, 128(38), pp.12394-12395.
https://doi.org/10.1021/ja0635231 - Moon, S., Kim, J., Choi, H.K., Kim, M.G., Lee,Y.S. and Lee,K.,2021.Electrochemical Synthesis of Metal organic Framework. Applied Chemistry for Engineering, 32(3), pp.229-236.
- Chen, D., Zhao, J., Zhang, P. and Dai, S., 2019. Mechanochemical synthesis of metal-organic frameworks. Polyhedron, 162, pp.59-64.
https://doi.org/10.1016/j.poly.2019.01.024 - Klinowski, J., Paz, F.A.A., Silva, P. and Rocha,J.,2011. Microwave assisted synthes-is of metal organic frameworks. Dalton Transactions, 40(2), pp.321-330.
https://doi.org/10.1039/C0DT00708K - Vaitsis, C., Sourkouni, G. and Argirusis, C., 2019. Metal organic frameworks(MOFs) and ultrasound: ai review. Ultrasonics sonochemistry, 52, pp.106-119.
https://doi.org/10.1016/j.ultsonch.2018.11.004 - Chen, L., Chen, X., Liu, H. and Li, Y., 2015. Encapsulation of Mono‐or Bimetal Nanoparticles inside Metal-Organic Frameworks via In situ Incorporation of Metal Precursors. Small, 11(22), pp.2642-2648.
https://doi.org/10.1002/smll.201403599 - Moon, H.R., Lim, D.W. and Suh, M.P., 2013. Fabrication of metal nanoparticles in metal-organic frameworks. Chemical Society Reviews, 42(4), pp.1807-1824.
https://doi.org/10.1039/C2CS35320B - Varghese, J.R., Wendt, C., Dix, F.B., Aulakh, D., Sazama, U., Yakovenko, A.A., Fröba, M., Wochnowski,J., Goia, D.V.and Wriedt, M., 2021.Design and characterization of metal nanoparticle infiltrated mesopo-rous metal-organic frameworks. Inorganic Chemistry, 60(17), pp.13000-13010.
https://doi.org/10.1021/acs.inorgchem.1c01433 - Tao, C.A. and Wang, J.F., 2020. Synthesis of metal organic frameworks by ball milling. Crystals, 11(1), p.15.
https://doi.org/10.3390/cryst11010015 - Hou, M. and Ge, J., 2017. Armoring enzymes by metal-organic frameworks by the coprecipitation method. In Methods in enzymology (Vol. 590, pp.59-75). Academic Press.
https://doi.org/10.1016/bs.mie.2016.12.002 - Wang, Z., Liu, J., Arslan, H.K., Grosjean, S., Hagendorn, T., Gliemann, H., Bräse, S. and Wöll, C., 2013. Post-synthetic modification of metal-organic framework thin films using click chemistry: the importance of strained C C triple bonds. Langmuir, 29(51), pp.15958-15964.
https://doi.org/10.1021/la403854w - Wang, Q., Liu, S., Tai, W., Li, J., Ye, M. and Yang, L., 2018. Purification, chemical structure and antioxidant activity of active ingredient (LPT-3d) separated from Lachnum sp. Process Biochemistry, 71, pp.166-174.
https://doi.org/10.1016/j.procbio.2018.04.012 - Bilal, M., Adeel, M., Rasheed, T. and Iqbal, H.M., 2019. Multifunctional metal-organic frameworks-based biocatalytic platforms: recent developments and future prospects. Journal of Materials Research and Technology, 8(2), pp.2359-2371.
https://doi.org/10.1016/j.jmrt.2018.12.001 - Salunkhe, R.R., Kaneti, Y.V. and Yamauchi, Y., 2017.Metal organic framework derived nanoporous metal oxides toward supercapacitor applications: progress and prospects. ACS nano, 11(6), pp.5293-5308.
https://doi.org/10.1021/acsnano.7b02796 - Yang, J., Zhang, Y.B., Liu, Q., Trickett, C.A., Gutiérrez-Puebla, E., Monge, M.Á., Cong, H., Aldossary, A., Deng, H. and Yaghi, O.M., 2017. Principles of designing extra-large pore openings and cages in zeolitic imidazolate frameworks. Journal of the American Chemical Society, 139(18), pp.6448-6455.
https://doi.org/10.1021/jacs.7b02272 - Yang, Q., Xu, Q. and Jiang, H.L., 2017. Metal-organic frameworks meet metal nanoparticles: synergistic effect for enhanced catalysis. Chemical Society Reviews, 46(15), pp.4774-4808.
https://doi.org/10.1039/C6CS00724D - Li, S. and Huo, F., 2015. Metal-organic framework composites: from fundamentals to applications. Nanoscale, 7(17), pp.7482-7501.
https://doi.org/10.1039/C5NR00518C - Liu, K.G., Sharifzadeh, Z., Rouhani, F., Ghorbanloo, M. and Morsali, A., 2021. Metal-organic framework composites as green/sustainable catalysts.Coordination Chemistry Reviews, 436, p.213827.
https://doi.org/10.1016/j.ccr.2021.213827 - Xue, Y., Zheng, S., Xue, H. and Pang, H., 2019. Metal organic framework composites and their electrochemical applications. Journal of Materials Chemistry A, 7(13), pp.7301-7327.
https://doi.org/10.1039/C8TA12178H - Tong, P., Liang, J., Jiang, X. and Li, J., 2020. Research progress on metal-organic framework composites in chemical sensors. Critical Reviews in Analytical Chemistry, 50(4), pp.376-392.
https://doi.org/10.1080/10408347.2019.1642732 - Olorunyomi, J.F., Geh, S.T., Caruso, R.A. and Doherty, C.M., 2021. Metal-organic frameworks for chemical sensing devices. Materials Horizons, 8(9), pp.2387-2419.
https://doi.org/10.1039/D1MH00609F - Ahmed, I. and Jhung, S.H., 2014.Composites of metal-organic frameworks: preparation and application in adsorption. Materials today, 17(3), pp.136-146.
https://doi.org/10.1016/j.mattod.2014.03.002 - Li, W., Zhang, Y., Li, Q. and Zhang, G., 2015. Metal− organic framework composite membranes: Synthesis and separation applications. Chemical Engineering Science, 135, pp.232-257.
https://doi.org/10.1016/j.ces.2015.04.011 - Yu, J., Mu, C., Yan, B., Qin, X., Shen, C., Xue,H. and Pang, H., 2017. Nanoparticle/MOF composites: preparations and applications. Materials Horizons, 4(4), pp.557-569.
https://doi.org/10.1039/C6MH00586A - Zheng, S., Li, Q., Xue, H., Pang, H. and Xu, Q., 2020. A highly alkaline stable metal oxide@ metal organic framework composite for high performance electrochemical energy storage. National science review, 7(2), pp.305-314.
https://doi.org/10.1093/nsr/nwz137 - Ren, J., Huang, Y., Zhu, H., Zhang, B., Zhu, H., Shen, S., Tan, G., Wu, F., He, H., Lan, S. and Xia, X., 2020. Recent progress on MOF‐derived carbon materials for energy storage. Carbon Energy, 2(2), pp.176-202.
https://doi.org/10.1002/cey2.44 - Liu, Y., Tang, C., Cheng, M., Chen, M., Chen, S., Lei, L., Chen, Y., Yi, H., Fu, Y. and Li, L., 2021. Polyoxometalate@metal organic framework composites as effective photocatalysts. ACS Catalysis, 11(21), pp.13374-13396.
https://doi.org/10.1021/acscatal.1c03866 - Kalaj, M., Bentz, K.C., Ayala Jr, S., Palomba, J.M., Barcus, K.S., Katayama, Y. and Cohen, S.M., 2020. MOF polymer hybrid materials: From simple composites to tailored architectures. Chemical reviews, 120(16), pp.8267-8302.
https://doi.org/10.1021/acs.chemrev.9b00575 - Lian, X., Fang, Y., Joseph, E., Wang, Q., Li, J., Banerjee, S., Lollar, C., Wang, X. and Zhou, H.C., 2017.Enzyme-MOF(metal-organic framework) composites. Chemical Society Reviews, 46(11),pp.3386-3401.
https://doi.org/10.1039/C7CS00058H - Ullah, S., Bustam, M.A., Assiri, M.A., Al-Sehemi, A.G., Kareem, F.A.A., Mukhtar, A., Ayoub, M. and Gonfa, G., 2019. Synthesis and characterization of iso-reticular metal-organic Framework-3 (IRMOF-3) for CO2/CH4 adsorption: Impact of post-synthetic aminomethyl propanol (AMP) functionalization. Journal of Natural Gas Science and Engineering, 72, p.103014.
https://doi.org/10.1016/j.jngse.2019.103014 - Tong, P., Liang, J., Jiang, X. and Li, J., 2020. Research progress on metal-organic framework composites in chemical sensors. Critical Reviews in Analytical Chemistry, 50(4), pp.376-392.
https://doi.org/10.1080/10408347.2019.1642732 - Canossa, S., Fornasari, L., Demitri, N., Mattarozzi, M., Choquesillo-Lazarte, D., Pelagatti, P. and Bacchi, A., 2019. MOF transmetalation beyond cation substitution: defective distortion of IRMOF-9 in the spotlight. CrystEngComm, 21(5), pp.827-834.
https://doi.org/10.1039/C8CE01808A - Esrafili, L., Safarifard, V., Tahmasebi, E., Esrafili, M.D. and Morsali, A., 2018. Functional group effect of Isoreticular metal-organic frameworks on heavy metal ion adsorption. New Journal of Chemistry, 42(11), pp.8864-8873.
https://doi.org/10.1039/C8NJ01150H - Cao, X.Q., Wang, X., Chen, M., Xiao, F., Huang, Y.M. and Lyu, X.J., 2021. Synthesis of nanoscale zeolitic imidazolate framework-8 (ZIF-8) using reverse micro-emulsion for Congo red adsorption. Separation and Purification Technology, 260, p.118062.
https://doi.org/10.1016/j.seppur.2020.118062 - Yi, J.T., Chen, T.T., Huo, J. and Chu, X., 2017. Nanoscale zeolitic imidazolate framework-8 for ratiometric fluorescence imaging of micro RNA in living cells. Anal-ytical chemistry, 89(22), pp.12351-12359.
https://doi.org/10.1021/acs.analchem.7b03369 - Li, Q. and Kim, H., 2012. Hydrogen production from NaBH4 hydrolysis via Co ZIF 9 catalyst. Fuel processing technology, 100, pp.43-48.
https://doi.org/10.1016/j.fuproc.2012.03.007 - Şahin, F., Topuz, B. and Kalıpçılar, H., 2018. Synthesis of ZIF 7,ZIF 8, ZIF 67and ZIF L from recycled mother liquors. Microporous and Mesoporous Materials, 261, pp.259-267.
https://doi.org/10.1016/j.micromeso.2017.11.020 - Pan, W., Cui, B., Wang, K., Shi, M., Lu, F., Li, N. and Tang, B., 2021. ATP-triggered mitochondrial cascade reactions for cancer therapy with nanoscale zeolitic imidazole framework-90. Theranostics, 11(16), p.7869.
https://doi.org/10.7150/thno.59593 - Pérez-Miana, M., Reséndiz-Ordóñez, J.U. and Coronas, J., 2021. Solventless synthesis of ZIF-L and ZIF 8 with hydraulic press and high temperature. Microporous and Mesoporous Materials, 328, p.111487.
https://doi.org/10.1016/j.micromeso.2021.111487 - Huang, A., Chen, Y., Wang, N., Hu, Z., Jiang, J. and Caro, J., 2012. A highly permeable and selective zeolitic imidazolate framework ZIF-95 membrane for H2/CO2 separation. Chemical Communications, 48(89), pp.10981-10983.
https://doi.org/10.1039/c2cc35691k - Wang, N., Liu, Y., Qiao, Z., Diestel, L., Zhou, J., Huang, A. and Caro, J., 2015. Polydopamine-based synthesis of a zeolite imidazolate framework ZIF-100 membrane with high H2/CO2 selectivity. Journal of Materials Chemistry A, 3(8), pp.4722-4728.
https://doi.org/10.1039/C4TA06763K - Park, K.S., Ni, Z., Côté, A.P., Choi, J.Y., Huang, R., Uribe-Romo, F.J., Chae, H.K., O’Keeffe, M. and Yaghi, O.M., 2006. Exceptional chemical and thermal stability of zeolitic imidazolate frameworks. Proceedings of the National Academy of Sciences, 103(27),pp.10186-10191.
https://doi.org/10.1073/pnas.0602439103 - Foo, M.L., Matsuda, R. and Kitagawa, S., 2014. Functional hybrid porous coordination polymers. Chemistry of materials, 26(1), pp.310-322.
https://doi.org/10.1021/cm402136z - Bureekaew, S., Shimomura, S. and Kitagawa, S., 2008. Chemistry and application of flexible porous coordination polymers. Scie-nce and Technology of Advanced Materials.
https://doi.org/10.1088/1468-6996/9/1/014108 - Wang, X.S., Ma, S., Rauch, K., Simmons, J.M., Yuan, D., Wang, X., Yildirim, T., Cole, W.C., López, J.J., Meijere, A.D. and Zhou, H.C., 2008. Metal− organic frameworks based on double-bond-coupled di-isophthalate linkers with high hydrogen and methane uptakes. Chemistry of Materials, 20(9), pp.3145-3152.
https://doi.org/10.1021/cm800403d - Wenzel, S.E., Fischer, M., Hoffmann, F. and Fröba, M., 2009. Highly Porous Metal-Organic Framework Containing a Novel Organosilicon Linker− a Promising Material for Hydrogen Storage. Inorganic chemistry, 48(14), pp.6559-6565.
https://doi.org/10.1021/ic900478z - Zhang, Y., Yang, X. and Zhou, H.C., 2018. Direct synthesis of functionalized PCN-333 via linker design for Fe3+ detection in aqueous media. Dalton Transactions, 47(34), pp.11806-11811.
https://doi.org/10.1039/C8DT01508B - Zhou, W., Hu, Z., Wei, J., Lu, H., Dai, H., Zhao, J., Zhang, W. and Guo, R., 2022. A ratiometric fluorescent probe based on PCN-224 for rapid and ultrasensitive detection of copper ions. Composites Communications, 33, p.101221.
https://doi.org/10.1016/j.coco.2022.101221 - Wei, C., Zhou, H. and Liu, Q., 2021. PCN-222 MOF decorated conductive PEDOT films for sensitive electrochemical determination of chloramphenicol. Materials Chemistry and Physics, 270, p.124831.
https://doi.org/10.1016/j.matchemphys.2021.124831 - Chen, C., Lee, Y.R. and Ahn, W.S., 2016. CO2 adsorption over metal-organic frameworks: A mini review. Journal of nanoscience and nanotechnology, 16(5), pp.4291-4301.
https://doi.org/10.1166/jnn.2016.10971 - Song, L., Zhang, J., Sun, L., Xu, F., Li, F., Zhang, H., Si, X., Jiao, C., Li, Z., Liu, S. and Liu, Y., 2012. Mesoporous metal organic frameworks: design and applications. Energy & Environmental Science, 5(6), pp.7508-7520.
https://doi.org/10.1039/c2ee03517k - Liu, Y., Wang, Z.U. and Zhou, H.C., 2012. Recent advances in carbon dioxide capture with metal‐organic frameworks. Greenhouse Gases: Science and Technology, 2(4), pp.239-259.
https://doi.org/10.1002/ghg.1296 - Kazemi, S. and Safarifard, V., 2018. Carbon dioxide capture on metal organic framework with amide-decorated pores. Nanochemistry Research, 3(1), pp.62-78.
- Ozturk, T.N. and Keskin, S., 2013. Predicting gas separation performances of porous coordination networks using atomistic simulations. Industrial & Enginee-ring Chemistry Research, 52(49), pp.17627-17639.
https://doi.org/10.1021/ie403159c - Yang, S., Li, X., Zeng, G., Cheng, M., Huang, D., Liu, Y., Zhou, C., Xiong, W., Yang, Y., Wang, W. and Zhang, G., 2021. Materials Institute Lavoisier (MIL) based materials for photocatalytic applications. Coordination Chemistry Reviews, 438, p.213874.
https://doi.org/10.1016/j.ccr.2021.213874 - Janiak, C. and Vieth, J.K., 2010. MOFs, MILs and more: concepts, properties and applications for porous coordination networks (PCNs). New Journal of Chemistry, 34(11), pp.2366-2388.
https://doi.org/10.1039/c0nj00275e - Araya, T., Jia, M., Yang, J., Zhao, P., Cai, K., Ma, W. and Huang,Y.,2017.Resin modified MIL 53 (Fe) MOF for improvement of photocatalytic performance. Applied Catalysis B: Environmental, 203, pp.768-777.
https://doi.org/10.1016/j.apcatb.2016.10.072 - Zhang, H., Gong, X., Song, Z., Zhang, S., Du, W., Nguyen, T.T., Guo, M. and Gao, X., 2021. Wood-based carbon quantum dots for enhanced photocatalysis of MIL-88B (Fe). Optical Materials, 113, p.110865.
https://doi.org/10.1016/j.optmat.2021.110865 - Zhang, Y., Liu, L., Yu, D., Liu, J., Zhao, L., Liu, J. and Liu, S., 2022. Preparation of Magnetic MIL-68 (Ga) Metal-Organic Framework and Heavy Metal Ion Removal Application. Molecules, 27(11), p.3443.
https://doi.org/10.3390/molecules27113443 - Xie, F., Gao, Y., Zhang, J., Bai, H., Zhang, J., Li, Z. and Zhu, W., 2022. A novel bifunctional cathode for the generation and activation of H2O2 in electro-Fenton: characteristics and mechanism. Electrochim-ica Acta, 430, p.141099.
https://doi.org/10.1016/j.electacta.2022.141099 - Yue, K., Zhang, X., Jiang, S., Chen, J., Yang, Y., Bi, F. and Wang, Y., 2021. Recent advances in strategies to modify MIL 125(Ti) and its environmental applications. Jou-rnal of Molecular Liquids, 335, p.116108.
https://doi.org/10.1016/j.molliq.2021.116108 - Iacomi, P., Gulcay-Ozcan, E., Pires Conti, P., Biswas, S., Steunou, N., Maurin, G., Rioland, G. and Devautour-Vinot, S., 2022. MIL-101 (Cr) MOF as an effective siloxane sensor. ACS Applied Materials & Interfaces, 14(15), pp.17531-17538.
https://doi.org/10.1021/acsami.2c02607 - Lei, T., Liu, Y. and Yang, J., 2022. Removal of furfural in wastewater by Al-MIL-53 prepared in various solvents. Inorganic Chemistry Communications, 140, p.109405.
https://doi.org/10.1016/j.inoche.2022.109405 - Jahromi, F.B., Elhambakhsh, A., Keshavarz, P. and Panahi, F., 2023. Insight into the application of amino acid-functionalized MIL-101 (Cr) micro fluids for high-efficiency CO2 absorption: Effect of amine number and surface area. Fuel, 334, p.126603.
https://doi.org/10.1016/j.fuel.2022.126603 - Jin, Y., Li, Y., Du, Q., Chen, B., Chen, K., Zhang, Y., Wang, M., Sun, Y., Zhao, S., Jing, Z. and Wang, J., 2023. Efficient adsorption of Congo red by MIL-53 (Fe)/chitosan composite hydrogel spheres. Microporous and Mesoporous Materials, 348, p.112404.
https://doi.org/10.1016/j.micromeso.2022.112404 - Zhou, M., Tang, T., Qin, D., Cheng, H., Wang, X., Chen, J., Wågberg, T. and Hu, G., 2023. Hematite nanoparticle decorated MIL-100 for the highly selective and sensitive electrochemical detection of trace-level paraquat in milk and honey. Sensors and Actuators B: Chemical, 376, p.132931.
https://doi.org/10.1016/j.snb.2022.132931 - Wu, L., Xue, M., Qiu, S.L., Chaplais, G., Simon-Masseron, A. and Patarin, J., 2012. Amino-modified MIL-68 (In) with enhanced hydrogen and carbon dioxide sorption enthalpy. Microporous and mesoporous materials, 157, pp.75-81.
https://doi.org/10.1016/j.micromeso.2011.12.034 - Yang, Q., Xu, Q. and Jiang, H.L., 2017. Metal-organic frameworks meet metal nanoparticles: synergistic effect for enhanced catalysis. Chemical Society Reviews, 46(15), pp.4774-4808.
https://doi.org/10.1039/C6CS00724D - Moon, H.R., Lim, D.W. and Suh, M.P., 2013. Fabrication of metal nanoparticles in metal-organic frameworks. Chemical Society Reviews, 42(4), pp.1807-1824.
https://doi.org/10.1039/C2CS35320B - Guo, K., Hussain, I., Fu, Y., Zhang, F. and Zhu, W., 2022. Strategies for improving the photocatalytic performance of metal-organic frameworks for CO2 reduction: A review. Journal of Environmental Sciences.
https://doi.org/10.1016/j.jes.2022.01.005 - Dhakshinamoorthy, A. and Garcia, H., 2012. Catalysis by metal nanoparticles embedded on metal-organic frameworks. Chemical Society Reviews, 41(15), pp.5262-5284.
https://doi.org/10.1039/c2cs35047e - Yang, Q., Yao, F., Zhong, Y., Chen, F., Shu, X., Sun, J., He, L., Wu, B., Hou, K., Wang, D. and Li, X., 2019. Metal-Organic Framework Supported Palladium Nanoparti-cles: Applications and Mechanisms. Particle & Particle Systems Characterization, 36(6), p.1800557.
https://doi.org/10.1002/ppsc.201800557 - Dai, S., Ngoc, K.P., Grimaud, L., Zhang, S., Tissot, A. and Serre, C., 2022. Impact of capping agent removal from Au NPs@ MOF core-shell nanoparticle heterogeneous catalysts. Journal of Materials Chemistry A, 10(6), pp.3201-3205.
https://doi.org/10.1039/D1TA09108E - Wang, Q., Yang, G., Fu, Y., Li, N., Hao, D. and Ma, S.,2022.Nanospace Engineering of Metal‐Organic Frameworks for Heterogeneous Catalysis. ChemNanoMat, 8(1), p.e202100396.
https://doi.org/10.1002/cnma.202100396 - Zhang, W., Shi, W., Ji, W., Wu, H., Gu, Z., Wang, P., Li, X., Qin, P., Zhang, J., Fan, Y. and Wu, T., 2020. Microenvironment of MOF channel coordination with Pt NPs for selective hydrogenation of unsaturated aldehydes. ACS Catalysis, 10(10), pp.5805-5813.
https://doi.org/10.1021/acscatal.0c00682 - Xu, W., Liu, M., Wang, S., Peng, Z., Shen, R. and Li, B., 2022. Interfacial ensemble effect of copper nanoparticles and nickel metal-organic framework on promoting hydrogen generation. International Journal of Hydrogen Energy, 47(55), pp.23213-23220.
https://doi.org/10.1016/j.ijhydene.2022.05.108 - Shen, M., Forghani, F., Kong, X., Liu, D., Ye, X., Chen, S. and Ding, T., 2020. Antibacterial applications of metal organic frameworks and their composites. Compreh-ensive Reviews in Food Science and Food Safety, 19(4), pp.1397-1419.
https://doi.org/10.1111/1541-4337.12515 - Railey, P., Song, Y., Liu, T. and Li, Y., 2017. Metal organic frameworks with immobilized nanoparticles: Synthesis and applications in photocatalytic hydrogen generation and energy storage. Materials Research Bulletin, 96, pp.385-394.
https://doi.org/10.1016/j.materresbull.2017.04.020 - Kempahanumakkagari, S., Vellingiri, K., Deep, A., Kwon, E.E., Bolan, N. and Kim, K.H., 2018. Metal-organic framework composites as electrocatalysts for electroch-emical sensing applications. Coordination Chemistry Reviews, 357, pp.105-129.
https://doi.org/10.1016/j.ccr.2017.11.028 - Wang, C., Kim, J., Tang, J., Kim, M., Lim, H., Malgras, V., You, J., Xu, Q., Li, J. and Yamauchi, Y., 2020. New strategies for novel MOF-derived carbon materials based on nanoarchitectures. Chem, 6(1), pp.19-40.
https://doi.org/10.1016/j.chempr.2019.09.005 - McHugh, L.N., Terracina, A., Wheatley, P.S., Buscarino, G., Smith, M.W. and Morris, R.E., 2019. Metal-Organic Framework‐Activated Carbon Composite Materials for the Removal of Ammonia from Contaminated Airstreams. Angewandte Chemie, 131(34), pp.11873-11877.
https://doi.org/10.1002/ange.201905779 - Zhang, Y., Bo, X., Nsabimana, A., Han, C., Li, M. and Guo, L., 2015. Electrocatalytically active cobalt-based metal-organic framework with incorporated macroporous carbon composite for electrochemical applications. Journal of materials chemistry A, 3(2), pp.732-738.
https://doi.org/10.1039/C4TA04411H - Sun, P.P., Li, Y.M., Zhang, Y.H., Shi, H. and Shi, F.N., 2022. Preparation and application of Ce-Cu based metal organic framework/biomass carbon composites in energy storage. Journal of Alloys and Compounds, 896, p.163081.
https://doi.org/10.1016/j.jallcom.2021.163081 - Shin, S. and Shin, M.W., 2021. Nickel metal-organic framework (Ni-MOF) derived NiO/C@ CNF composite for the application of high performance self-standing supercapacitor electrode. Applied Surface Science, 540, p.148295.
https://doi.org/10.1016/j.apsusc.2020.148295 - Chavali, M.S. and Nikolova, M.P., 2019. Metal oxide nanoparticles and their applications in nanotechnology. SN applied sciences, 1(6), p.607.
https://doi.org/10.1007/s42452-019-0592-3 - Jiao, L., Zhang, R., Wan, G., Yang, W., Wan, X., Zhou, H., Shui, J., Yu, S.H. and Jiang, H.L., 2020. Nanocasting SiO2 into metal-organic frameworks imparts dual protection to high-loading Fe single-atom electrocatalysts. Nature communications, 11(1),p.2831.
https://doi.org/10.1038/s41467-020-16715-6 - Liu, H., Wang, Z., Sun, C., Shao, J., Li, Z., Zhang, H., Qiu, M., Pan, G. and Yang, X., 2023. Construction of Co3O4/Fe3O4 heterojunctions from metal organic framework derivatives for high performance toluene sensor. Sensors and Actuators B: Chemical, 375, p.132863.
https://doi.org/10.1016/j.snb.2022.132863 - Amu-Darko, J.N.O., Hussain, S., Zhang, X., Alothman, A.A., Ouladsmane, M., Nazir, M.T., Qiao, G. and Liu, G., 2023. Metal-organic frameworks-derived In2O3/ZnO porous hollow nanocages for highly sensitive H2S gas sensor. Chemosphere, 314, p.137670.
https://doi.org/10.1016/j.chemosphere.2022.137670 - Liu, J., Wang, W., Li, G., Bian, X., Liu, Y., Zhang, J., Gao, J., Wang, C., Zhu, B. and Lu, H., 2023. Metal-organic framework-derived CuO tube-like nanofibers with high surface area and abundant porosities for enhanced room-temperature NO2 sensing properties. Journal of Alloys and Compounds, 934, p.167950.
https://doi.org/10.1016/j.jallcom.2022.167950 - Su, L., Wang, J., Song, T. and Tu, W., 2022. Dynamic template-driven formation of NiS/CdS nanoparticles via metal organic frameworks for efficient hydrogen evolution. International Journal of Hydrogen Energy, 47(14), pp.8776-8785.
https://doi.org/10.1016/j.ijhydene.2021.12.234 - Wang, P., Jiang, Y., Cao, Y., Wu, X. and Liu, X., 2022. MOF-derived ZnS/NC yolk-shell composites for highly reversible lithium storage. New Journal of Chemistry, 46(23), pp.11101-11107.
https://doi.org/10.1039/D2NJ01340A - Tripathy, S.P., Subudhi, S., Das, S., Ghosh, M.K., Das, M., Acharya, R., Acharya, R. and Parida, K., 2022. Hydrolytically stable citrate capped Fe3O4@ UiO-66-NH2 MOF: A hetero-structure composite with enhanced activity towards Cr (VI) adsorption and photocatalytic H2 evolution. Journal of Colloid and Interface Science, 606, pp.353-366.
https://doi.org/10.1016/j.jcis.2021.08.031 - Amu-Darko, J.N.O., Hussain, S., Zhang, X., Alothman, A.A., Ouladsmane, M., Nazir, M.T., Qiao, G. and Liu, G., 2023.Metal-organic frameworks-derived In2O3/ZnO porous hollow nanocages for highly sensitive H2S gas sensor. Chemosphere, 314, p.137670.
https://doi.org/10.1016/j.chemosphere.2022.137670 - Noor, T., Mohtashim, M., Iqbal, N., Naqvi, S.R., Zaman, N., Rasheed, L. and Yousuf, M., 2021. Graphene based FeO/NiO MOF composites for methanol oxidation reaction. Journal of Electroanalytical Chemistry, 890, p.115249.
https://doi.org/10.1016/j.jelechem.2021.115249 - Zhao, G., Qin, N., Pan, A., Wu, X., Peng, C., Ke, F., Iqbal, M., Ramachandraiah, K. and Zhu, J., 2019. Magnetic nanoparticles@ metal-organic framework composites as sustainable environment adsorbents. Journal of Nanomaterials, 2019, pp.1-11.
https://doi.org/10.1155/2019/1454358 - Nadar, S.S., Vaidya, L. and Rathod, V.K., 2020. Enzyme embedded metal organic framework (enzyme-MOF):De novo approaches for immobilization. International journal of biological macromolecules, 149, pp.861-876.
https://doi.org/10.1016/j.ijbiomac.2020.01.240 - Lian, X., Fang, Y., Joseph, E., Wang, Q., Li, J., Banerjee, S., Lollar, C., Wang, X. and Zhou, H.C., 2017.Enzyme-MOF (metal-organic framework) composites. Chemical Society Reviews, 46(11), pp.3386-3401.
https://doi.org/10.1039/C7CS00058H - Rafiei, S., Tangestaninejad, S., Horcajada, P., Moghadam, M., Mirkhani, V., Mohammadpoor-Baltork, I., Kardanpour, R. and Zadehahmadi, F., 2018. Efficient biodiesel production using a lipase@ ZIF-67 nanobioreactor. Chemical Engineering Journal, 334, pp.1233-1241.
https://doi.org/10.1016/j.cej.2017.10.094 - Ren, J., Huang, Y., Zhu, H., Zhang, B., Zhu, H., Shen, S., Tan, G., Wu, F., He, H., Lan, S. and Xia, X., 2020. Recent progress on MOF‐derived carbon materials for energy storage. Carbon Energy, 2(2), pp.176-202.
https://doi.org/10.1002/cey2.44 - Fang, R., Tian, P., Yang, X., Luque, R. and Li, Y., 2018. Encapsulation of ultrafine metal-oxide nanoparticles within mesopores for biomass derived catalytic applications. Chemical science, 9(7), pp.1854-1859.
https://doi.org/10.1039/C7SC04724J - Lian, X., 2018. Design and Synthesis of Enzyme-MOF (Metal-Organic Framework) Composites for Long-Persistent Biomedical Applications (Doctoral dissertation).
- Ahmad, M., Luo, Y., Wöll, C., Tsotsalas, M. and Schug, A., 2020. Design of metal-organic framework template materials using high throughput computational screening. Molecules, 25(21), p.4875.
https://doi.org/10.3390/molecules25214875 - Yu, J., Mu, C., Yan, B., Qin, X., Shen, C., Xue, H. and Pang, H., 2017. Nanoparticle/MOF composites: preparations and applications. Materials Horizons, 4(4), pp.557-569.
https://doi.org/10.1039/C6MH00586A - Zhang, Z., Cui, X., Yuan, W., Yang, Q., Liu, H., Xu, H. and Jiang, H.L., 2018.Encapsulating surface clean metal nanoparticles inside metal-organic frameworks for enhanced catalysis using a novel γ-ray radiation approach. Inorganic Chemistry Frontiers, 5(1), pp.29-38.
https://doi.org/10.1039/C7QI00577F - Chen, L., Zhang, X., Cheng, X., Xie, Z., Kuang, Q. and Zheng, L., 2020. The function of metal-organic frameworks in the application of MOF based composites. Nanoscale Advances, 2(7), pp.2628-2647.
https://doi.org/10.1039/D0NA00184H - Li, X., Zhang, Z., Xiao, W., Deng, S., Chen, C. and Zhang, N., 2019. Mechanochemistry-assisted encapsulation of metal nanoparticles in MOF matrices via a sacrificial strategy. Journal of Materials Chemistry A, 7(24), pp.14504-14509.
https://doi.org/10.1039/C9TA03578H - Li, Y., Wei, Z., Zhang, Y., Guo, Z., Chen, D., Jia, P., Chen, P. and Xing, H., 2019. Dual-emitting EY@ Zr-MOF composite as self-calibrating luminescent sensor for selective detection of inorganic ions and nitroaromatics. ACS Sustainable Chemistry & Engineering, 7(6), pp.6196-6203.
https://doi.org/10.1021/acssuschemeng.8b06500 - Huo, Y., Xiu, S., Meng, L.Y. and Quan, B., 2022. Solvothermal synthesis and applications of micro/nano carbons: A review. Chemical Engineering Journal, p.138572.
https://doi.org/10.1016/j.cej.2022.138572 - De Villenoisy, T., Zheng, X., Wong, V., Mofarah, S.S., Arandiyan, H., Yamauchi, Y., Koshy, P. and Sorrell, C.C., 2023. Principles of Design and Synthesis of Metal Derivatives from MOFs. Advanced Materia-ls, p.2210166.
https://doi.org/10.1002/adma.202210166 - Jin, Y., Zhao, C., Sun, Z., Lin, Y., Chen, L., Wang, D. and Shen, C., 2016. Facile synthesis of Fe-MOF/RGO and its application as a high performance anode in lithium-ion batteries. RSC advances, 6(36), pp.30763-30768.
https://doi.org/10.1039/C6RA01645F - Dang, A., Doan, T. and Huyen, P.T., 2022. CdS/MIL-101 as a Visible-light-driven Photocatalyst for Methylene Blue Degradation.
https://doi.org/10.21203/rs.3.rs-2332046/v1 - Arul, P., Gowthaman, N.S.K., John, S.A. and Lim, H.N., 2020. Ultrasonic assisted synthesis of size-controlled Cu-metal-organic framework decorated graphene oxide composite: sustainable electrocatalyst for the trace-level determination of nitrite in environmental water samples. ACS omega, 5(24), pp.14242-14253.
https://doi.org/10.1021/acsomega.9b03829 - Ling, J., Zhou, A., Wang, W., Jia, X., Ma, M. and Li, Y., 2022. One-pot method synthesis of bimetallic MgCu-MOF-74 and its CO2 adsorption under visible light. Acs Omega, 7(23), pp.19920-19929.
https://doi.org/10.1021/acsomega.2c01717 - Ke, F., Peng, C., Zhang, T., Zhang, M., Zhou, C., Cai, H., Zhu, J. and Wan, X., 2018. Fumarate-based metal-organic frameworks as a new platform for highly selective removal of fluoride from brick tea. Scientific reports, 8(1), p.939.
https://doi.org/10.1038/s41598-018-19277-2 - Tanhaei, M., Mahjoub, A.R. and Safarifard, V., 2019. Energy-efficient sonochemical approach for the preparation of nanohybrid composites from graphene oxide and metal-organic framework. Inorganic Chemistry Communications, 102, pp.185
https://doi.org/10.1016/j.inoche.2019.02.024 - Qian, Y., Zhang, F. and Pang, H., 2021. A review of MOFs and their composites‐based photocatalysts: synthesis and applications. Advanced Functional Materials, 31(37), p.2104231.
https://doi.org/10.1002/adfm.202104231 - Wakeman, M.D. and Rudd, C.D., 2000. Compression molding of thermoplastic composites.
https://doi.org/10.1016/B0-08-042993-9/00173-X - Lehto, V.P. and Riikonen, J., 2014. Drug loading and characterization of porous silicon materials. In Porous silicon for biomedical applications (pp. 337-355). Woodhead Publishing.
https://doi.org/10.1533/9780857097156.3.337 - Tsao, K.C. and Yang, H., 2018. Oxygen reduction catalysts on nanoparticle electrodes. In Encyclopedia of Interfacial Chemistry: Surface Science and Electrochemistry (pp. 796-811). Elsevier.
https://doi.org/10.1016/B978-0-12-409547-2.13334-7 - Liu, L., Ping, E., Sun, J., Zhang, L., Zhou, Y., Zhong, Y. and Zhou, Y., 2019. Multifunctional Ag@ MOF-5@ chitosan non-woven cloth composites for sulfur mustard decontamination and hemostasis. Dalton Transactions, 48(20), pp.6951-6959.
https://doi.org/10.1039/C9DT00503J - Belyaeva, E.V., Isaeva, V.I., Said-Galiev, E.E., Tkachenko, O.P., Savilov, S.V., Egorov, A.V., Kozlova, L.M., Sharf, V.Z. and Kustova, L.M., 2014. New method for catalyst preparation based on metal-organic framework MOF 5 for the partial hydrogenation of phenylacetylene. Russian Chemical Bulletin, 63, pp.396-403.
https://doi.org/10.1007/s11172-014-0443-8 - Surya, S.G., Bhanoth, S., Majhi, S.M., More, Y.D., Teja, V.M. and Chappanda, K.N., 2019. A silver nanoparticle-anchored UiO-66 (Zr) metal-organic framework (MOF)-based capacitive H2S gas sensor. CrystEngComm, 21(47), pp.7303-7312.
https://doi.org/10.1039/C9CE01323G - Devaraj, N.K. and Finn, M.G., 2021. Introduction: click chemistry. Chemical Reviews, 121(12), pp.6697-6698.
https://doi.org/10.1021/acs.chemrev.1c00469 - Nandivada, H., Jiang, X. and Lahann, J., 2007. Click chemistry: versatility and control in the hands of materials scientists. Advanced Materials, 19(17),pp.2197-2208.
https://doi.org/10.1002/adma.200602739 - Hein, C.D., Liu, X.M. and Wang, D., 2008. Click chemistry, a powerful tool for pharmaceutical sciences. Pharmaceutical research, 25, pp.2216-2230.
https://doi.org/10.1007/s11095-008-9616-1 - Battigelli, A., Almeida, B. and Shukla, A., 2022. Recent advances in bioorthogonal click chemistry for biomedical applications. Bioconjugate Chemistry, 33(2), pp.263-271.
https://doi.org/10.1021/acs.bioconjchem.1c00564 - Mueller, E., Poulin, I., Bodnaryk, W.J. and Hoare, T., 2022. Click chemistry hydrogels for extrusion bioprinting: progress, challenges, and opportunities. Biomacromolecules, 23(3), pp.619-640.
https://doi.org/10.1021/acs.biomac.1c01105 - Singh, R., Singh, G., George, N., Singh, G., Gupta, S., Singh, H., Kaur, G. and Singh, J., 2023. Copper-Based Metal-Organic Frameworks (MOFs) as an Emerging Catalytic Framework for Click Chemistry. Catalysts, 13(1), p.130.
https://doi.org/10.3390/catal13010130 - Mandal, S., Natarajan, S., Mani, P. and Pankajakshan, A., 2021. Post‐synthetic modification of metal-organic frameworks toward applications. Advanced Functional Materials, 31(4), p.2006291.
https://doi.org/10.1002/adfm.202006291 - Kirchon, A., Feng, L., Drake, H.F., Joseph, E.A. and Zhou, H.C., 2018. From fundamentals to applications: a toolbox for robust and multifunctional MOF materials. Chemical Society Reviews, 47(23), pp.8611-8638.
https://doi.org/10.1039/C8CS00688A - Yan, A.X., Yao, S., Li, Y.G., Zhang, Z.M., Lu, Y., Chen, W.L. and Wang, E.B., 2014. Incorporating polyoxometalates into a porous MOF greatly improves its selective adsorption of cationic dyes. Chemistry-A European Journal, 20(23), pp.6927-6933.
https://doi.org/10.1002/chem.201400175 - Brahmi, C., Benltifa, M., Vaulot, C., Michelin, L., Dumur, F., Millange, F., Frigoli, M., Airoudj, A., Morlet-Savary, F., Bousselmi, L. and Lalevée, J., 2021. New hybrid MOF/polymer composites for the photodegradation of organic dyes. European Polymer Journal, 154, p.110560.
https://doi.org/10.1016/j.eurpolymj.2021.110560 - Tong, H., Ji, Y., He, T., He, R., Chen, M., Zeng, J. and Wu, D., 2022. Preparation and photocatalytic performance of UIO-66/La-MOF composite. Water Science and Technology, 86(1), pp.95-109.
https://doi.org/10.2166/wst.2022.197 - Ramohlola, K.E., Masikini, M., Mdluli, S.B., Monama, G.R., Hato, M.J., Molapo, K.M., Iwuoha, E.I. and Modibane, K.D., 2017. Electrocatalytic hydrogen production properties of poly (3-aminobenzoic acid) doped with metal organic frameworks. Int. J. Electrochem. Sci, 12, pp.4392-4405.
https://doi.org/10.20964/2017.05.58 - Bagheri, A., Taghizadeh, M., Behbahani, M., Asgharinezhad, A.A., Salarian, M., Dehghani, A., Ebrahimzadeh, H. and Amini, M.M., 2012. Synthesis and characterization of magnetic metal-organic framework (MOF) as a novel sorbent, and its optimization by experimental design methodology for determination of palladium in environmental samples. Talanta, 99, pp.132-139.
https://doi.org/10.1016/j.talanta.2012.05.030 - Xiao, H., Zhang, W., Yao, Q., Huang, L., Chen, L., Boury, B. and Chen, Z., 2019. Zn-free MOFs like MIL-53 (Al) and MIL-125 (Ti) for the preparation of defect-rich,ultrafine ZnO nanosheets with high photoca-talytic performance. Applied Catalysis B: Environmental, 244, pp.719-731.
https://doi.org/10.1016/j.apcatb.2018.11.026 - Wang, Y., Xie, J., Wu, Y., Ge, H. and Hu, X., 2013. Preparation of a functionalized magnetic metal-organic framework sorbent for the extraction of lead prior to electrothe-rmal atomic absorption spectrometer analys-is. Journal of Materials Chemistry A, 1(31), pp.8782-8789.
https://doi.org/10.1039/c3ta11178d - Cao, K., Yu, Z. and Yin, D., 2019. Preparation of Ce-MOF@ TEOS to enhance the anti-corrosion properties of epoxy coatings. Progress in Organic Coatings, 135, pp.613-621.
https://doi.org/10.1016/j.porgcoat.2019.06.015 - Ren, J., Li, T., Zhou, X., Dong, X., Shorokhov, A.V., Semenov, M.B., Krevchik, V.D. and Wang, Y., 2019. Encapsulating all-inorganic perovskite quantum dots into mesoporous metal organic frameworks with significantly enhanced stability for optoelectronic applic-ations. Chemical Engineering Journal, 358,pp.30-39.
https://doi.org/10.1016/j.cej.2018.09.149 - Yang, Z., Zhang, D. and Chen, H., 2019. MOF-derived indium oxide hollow microtubes/MoS2 nanoparticles for NO2 gas sensing. Sensors and Actuators B: Chemical, 300, p.127037.
https://doi.org/10.1016/j.snb.2019.127037 - Da Silva, C.T.P., Veregue, F.R., Moisés, M.P., Guilherme, M.R. and Rinaldi, A.W., 2019. Synthesis of Al2O3-nanowhisker-based HKUST1 MOF composites. Materials Chemistry and Physics, 232, pp.446-451.
https://doi.org/10.1016/j.matchemphys.2019.05.009 - Meng, W., Xu, S., Dai, L., Li, Y., Zhu, J. and Wang, L., 2017. An enhanced sensitivity towards H2O2 reduction based on a novel Cu metal-organic framework and acetylene black modified electrode. Electro-chimica Acta, 230, pp.324-332.
https://doi.org/10.1016/j.electacta.2017.02.017 - Chen, L., Chen, H. and Li, Y., 2014. One-pot synthesis of Pd@ MOF composites without the addition of stabilizing agents. Chemical Communications, 50(94), pp.14752-14755.
https://doi.org/10.1039/C4CC06568A - Wang, Y. and Zhang, L., 2022. High sensitive label-free electrochemical sensor using polydopamine-coated Zr-MOF composites for rapid detection of amoxicillin. Int. J. Electrochem. Sci, 17(221293), p.2.
https://doi.org/10.20964/2022.12.85 - Lan, Y.C., Kamal, S., Lin, C.C., Liu, Y.H. and Lu, K.L., 2022. Ultra-Thin Zr-MOF/PVA/Melamine Composites with Remarkable Sound Attenuation Effects.
https://doi.org/10.21203/rs.3.rs-2257532/v1 - Duan, J., Pan, Y., Liu, G. and Jin, W., 2018. Metal-organic framework adsorbents and membranes for separation applications. Current Opinion in Chemical Engineering, 20, pp.122-131.
https://doi.org/10.1016/j.coche.2018.04.005 - Qian, Q., Asinger, P.A., Lee, M.J., Han, G., Mizrahi Rodriguez, K., Lin, S., Benedetti, F.M., Wu, A.X., Chi, W.S. and Smith, Z.P., 2020. MOF-based membranes for gas separations. Chemical reviews, 120(16), pp.8161-8266.
https://doi.org/10.1021/acs.chemrev.0c00119 - Li, Y., Lin, Z., Wang, X., Duan, Z., Lu, P., Li, S., Ji, D., Wang, Z., Li, G., Yu, D. and Liu, W., 2021. High-hydrophobic ZIF-8@ PLA composite aerogel and application for oil-water separation. Separation and Purification Technology, 270, p.118794.
https://doi.org/10.1016/j.seppur.2021.118794 - Yang, X., Li, C., Qi, M. and Qu, L., 2016. Graphene-ZIF8 composite material as stationary phase for high-resolution gas chromatographic separations of aliphatic and aromatic isomers. Journal of Chromatogra-phy A, 1460, pp.173-180.
https://doi.org/10.1016/j.chroma.2016.07.029 - Mahmoodi, N.M., Taghizadeh, M., Taghizadeh, A., Abdi, J., Hayati, B. and Shekarchi, A.A., 2019. Bio-based magnetic metal-organic framework nanocomposite: Ultrasound-assisted synthesis and pollutant (heavy metal and dye) removal from aqueous media. Applied Surface Science, 480, pp.288-299.
https://doi.org/10.1016/j.apsusc.2019.02.211 - Habib, N., Durak, O., Zeeshan, M., Uzun, A. and Keskin, S., 2022. A novel IL/MOF/polymer mixed matrix membrane having superior CO2/N2 selectivity. Journal of Membrane Science, 658, p.120712.
https://doi.org/10.1016/j.memsci.2022.120712 - Zhang, X.F., Feng, Y., Wang, Z., Jia, M. and Yao, J., 2018. Fabrication of cellulose nanofibrils/UiO-66-NH2 composite membrane for CO2/N2 separation. Journal of Membrane Science, 568, pp.10-16.
https://doi.org/10.1016/j.memsci.2018.09.055 - Zeeshan, M., Keskin, S. and Uzun, A., 2018. Enhancing CO2/CH4 and CO2/N2 separation performances of ZIF 8 by post synthesis modification with [BMIM][SCN]. Polyhedron, 155, pp.485-492.
https://doi.org/10.1016/j.poly.2018.08.073 - Guo, X., Huang, H., Ban, Y., Yang, Q., Xiao, Y., Li, Y., Yang, W. and Zhong, C., 2015.Mixed matrix membranes incorporated with amine-functionalized titanium-based metal-organic framework for CO2/CH4 separation. Journal of Membrane Science, 478, pp.130-139.
https://doi.org/10.1016/j.memsci.2015.01.007 - Wang, T., Lin, E., Peng, Y.L., Chen, Y., Cheng, P. and Zhang, Z., 2020. Rational design and synthesis of ultramicroporous metal-organic frameworks for gas separation. Coordination Chemistry Reviews, 423, p.213485.
https://doi.org/10.1016/j.ccr.2020.213485 - Noonikara‐Poyil, A., Cui, H., Yakovenko, A.A., Stephens, P.W., Lin, R.B., Wang, B., Chen, B. and Dias, H.R., 2021. A Molecular Compound for Highly Selective Purification of Ethylene. Angewandte Chemie, 133(52), pp.27390-27394.
https://doi.org/10.1002/ange.202109338 - Chen, Y., Wu, H., Lv, D., Shi, R., Chen, Y., Xia, Q. and Li, Z., 2018. Highly adsorptive separation of ethane/ethylene by an ethane-selective MOF MIL-142A. Industrial & Engineering Chemistry Research, 57(11), pp.4063-4069.
https://doi.org/10.1021/acs.iecr.7b05260 - Xu, M.M., Kong, X.J., He, T., Wu, X.Q., Xie, L.H. and Li, J.R., 2019. Reaction duration-dependent formation of two Cu (ii)-MOFs with selective adsorption properties of C3 H4 over C 3 H 6. Dalton Transactions, 48(25), pp.9225-9233.
https://doi.org/10.1039/C9DT01677E - Zhang, Q., Li, H., Chen, S., Duan, J. and Jin, W., 2020. Mixed-matrix membranes with soluble porous organic molecular cage for highly efficient C3H6/C3H8 separation. Jour-nal of Membrane Science, 611, p.118288.
https://doi.org/10.1016/j.memsci.2020.118288 - Feng, X., Zong, Z., Elsaidi, S.K., Jasinski, J.B., Krishna, R., Thallapally, P.K. and Carreon, M.A., 2016. Kr/Xe separation over a chabazite zeolite membrane. Journal of the American Chemical Society, 138(31), pp.9791-9794.
https://doi.org/10.1021/jacs.6b06515 - Xiao, D. and Xiao, D., 2016. Insulating characteristics of SF6 gas mixtures. Gas Discharge and Gas Insulation, pp.231-270.
https://doi.org/10.1007/978-3-662-48041-0_8 - Nakles, M.R., Hargus Jr, W.A., Delgado, J.J. and Corey, R.L., 2011. A Performance Comparison of Xenon and Krypton Propellant on an SPT-100 Hall Thruster (Preprint). Air force research lab Edwards afb ca.
https://doi.org/10.21236/ADA549666 - Gong, L., Ye, Y., Liu, Y., Li, Y., Bao, Z., Xiang, S., Zhang, Z. and Chen, B., 2022. A microporous hydrogen bonded organic framework for efficient Xe/Kr separation. ACS Applied Materials &Interfaces, 14(17), pp.19623-19628.
https://doi.org/10.1021/acsami.2c04746 - Wang, F., Bi, Z., Ding, L. and Yang, Q., 2023. Large-scale computational screening of metal-organic frameworks for D2/H2 separation. Chinese Journal of Chemical Engineering, 54, pp.323-330.
https://doi.org/10.1016/j.cjche.2022.04.003 - Xue, J., Xu, M., Gao, J., Zong, Y., Wang, M. and Ma, S., 2021. Multifunctional porphyrinic Zr-MOF composite membrane for high-performance oil-in-water separation and organic dye adsorption/photocatalysis. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 628, p.127288.
https://doi.org/10.1016/j.colsurfa.2021.127288 - Rana, A., Ghosh, S. and Biswas, S., 2023. An eco-friendly approach using a nonfluorous self-cleaning metal-organic framework composite and membrane for oil-water separation. Inorganic Chemistry Frontiers.
https://doi.org/10.1039/D2QI02062A - Liu, X., Shan, Y., Zhang, S., Kong, Q. and Pang, H., 2022. Application of metal organic framework in wastewater treatment. Green Energy & Environment.
https://doi.org/10.1016/j.gee.2022.03.005 - Shokri, A. and Nasernejad, B., 2023. Treatment of spent caustic wastewater by electro-Fenton process: Kinetics and cost analysis. Process Safety and Environmental Protection, 172, pp.836-845.
https://doi.org/10.1016/j.psep.2023.02.077 - Yusuf, M., Khan, M.A., Otero, M., Abdullah, E.C., Hosomi, M., Terada, A. and Riya, S., 2017. Synthesis of CTAB intercalated graphene and its application for the adsorption of AR265 and AO7 dyes from water. Journal of colloid and interface science, 493, pp.51-61.
https://doi.org/10.1016/j.jcis.2017.01.015 - Shokri, A., 2022. Employing electro-peroxone process for degradation of Acid Red 88 in aqueous environment by Central Composite Design: A new kinetic study and energy consumption. Chemosphere, 296, p.133817.
https://doi.org/10.1016/j.chemosphere.2022.133817 - Shokri, A., 2021. Using NiFe2O4 as a nano photocatalyst for degradation of polyvinyl alcohol in synthetic wastewater. Environmental Challenges, 5, p.100332.
https://doi.org/10.1016/j.envc.2021.100332 - Tan, Y., Chen, M. and Hao, Y., 2012. High efficient removal of Pb (II) by amino-functionalized Fe3O4 magnetic nano-particles. Chemical engineering journal, 191, pp.104-111.
https://doi.org/10.1016/j.cej.2012.02.075 - Wibowo, A., Marsudi, M.A., Pramono, E., Belva, J., Parmita, A.W., Patah, A., Eddy, D.R., Aimon, A.H. and Ramelan, A., 2021. Recent improvement strategies on metal-organic frameworks as adsorbent, catalyst, and membrane for wastewater treatment. Molecules, 26(17), p.5261.
https://doi.org/10.3390/molecules26175261 - Shi, Z., Xu, C., Guan, H., Li, L., Fan, L., Wang, Y., Liu, L., Meng, Q. and Zhang, R., 2018. Magnetic metal organic frameworks (MOFs) composite for removal of lead and malachite green in wastewater. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 539, pp.382-390.
https://doi.org/10.1016/j.colsurfa.2017.12.043 - Ramezanalizadeh, H., Zakeri, F. and Manteghi, F., 2018. Immobilization of BaWO4 nanostructures on a MOF-199-NH2: An efficient separable photocatalyst for the degradation of organic dyes. Optik, 174, pp.776-786.
https://doi.org/10.1016/j.ijleo.2018.08.052 - Lei, J., Qian, R., Ling, P., Cui, L. and Ju, H., 2014. Design and sensing applications of metal-organic framework composites. TrAC Trends in Analytical Chemistry, 58, pp.71-78.
https://doi.org/10.1016/j.trac.2014.02.012 - Lopa, N.S., Rahman, M.M., Ahmed, F., Sutradhar, S.C., Ryu, T. and Kim, W., 2018. A base-stable metal-organic framework for sensitive and non-enzymatic electrochemical detection of hydrogen peroxide. Electrochimica Acta, 274, pp.49-56.
https://doi.org/10.1016/j.electacta.2018.03.148 - Lustig, W.P., Mukherjee, S., Rudd, N.D., Desai, A.V., Li, J. and Ghosh, S.K., 2017. Metal-organic frameworks: functional luminescent and photonic materials for sensing applications. Chemical Society Reviews, 46(11), pp.3242-3285.
https://doi.org/10.1039/C6CS00930A - Er, E., Recent Developments in MOF-based Sensors for Pharmaceutical Compounds. Metal-Organic Frameworks-Based Hybrid Materials for Environmental Sensing and Monitoring, pp.263-276.
- Mehta, J., Dhaka, S., Bhardwaj, N., Paul, A.K., Dayananda, S., Lee, S.E., Kim, K.H. and Deep, A., 2019. Application of an enzyme encapsulated metal organic frame work composite for convenient sensing and degradation of methyl parathion. Sensors and Actuators B: Chemical, 290, pp.267-274.
https://doi.org/10.1016/j.snb.2019.03.116 - Freund, P., Senkovska, I. and Kaskel, S., 2017. Switchable conductive MOF-nanocarbon composite coatings as threshold sensing architectures. ACS applied materials & interfaces, 9(50), pp.43782-43789.
https://doi.org/10.1021/acsami.7b13924 - Majeed, S., Farooq, M.U., Naqvi, S.T.R., Fatima, B., Najam-ul-Haq, M., Majeed, S., Ali, F. and Khan, N.A., MOF-based Electrochemical Sensors for DNA/RNA/ ATP. In Metal-Organic Frameworks-Based Hybrid Materials for Environmental Sensing and Monitoring (pp. 237-247). CRC Press.
https://doi.org/10.1201/9781003188148-25 - Liu, Q., Tang, S., Wang, H. and Han, P., 2022. Stereospecific recognition and rapid determination of d-amino acids in human serum based on luminescent metal organic frameworks. New Journal of Chemistry, 46(17), pp.8029-8036.
https://doi.org/10.1039/D1NJ05570D - Wang, X., Zhang, L., Ye, N. and Xiang, Y., 2022. Synthesis of a dual metal-organic framework heterostructure as a fluorescence sensing platform for rapid and sensitive detection of tetracycline in milk and beef samples. Food Analytical Methods, 15(10), pp.2801-2809.
https://doi.org/10.1007/s12161-022-02332-0 - Zhang, Q., Luo, Q., Wu, Y., Yu, R., Cheng, J. and Zhang, Y., 2021. Construction of a Keggin heteropolyacid/Ni-MOF catalyst for esterification of fatty acids. RSC advances, 11(53), pp.33416-33424.
https://doi.org/10.1039/D1RA06023F - Ma, Z.Z., Wang, Y.S., Liu, B., Jiao, H. and Xu, L., 2022. A Non-Enzymatic Electrochemical Sensor of Cu@ Co-MOF Composite for Glucose Detection with High Sensitivity and Selectivity. Chemosensors, 10(10), p.416.
https://doi.org/10.3390/chemosensors10100416 - Liu, Y., Lei, P., Liao, X. and Wang, C., 2022. Nanoscale metal-organic frameworks as smart nanocarriers for cancer therapy. Journal of Nanostructure in Chemistry, pp.1-19.
https://doi.org/10.1007/s40097-022-00493-2 - Anik, Ü., Timur, S. and Dursun, Z., 2019. Metal organic frameworks in electrochemical and optical sensing platforms: a review. Microchimica Acta, 186, pp.1-15.
https://doi.org/10.1007/s00604-019-3321-0 - He, J., Xu, J., Yin, J., Li, N. and Bu, X.H., 2019. Recent advances in luminescent metal-organic frameworks for chemical sensors. Science China Materials, 62(11), pp.1655-1678.
https://doi.org/10.1007/s40843-019-1169-9 - Cai, G., Yan, P., Zhang, L., Zhou, H.C. and Jiang, H.L., 2021. Metal organic framework-based hierarchically porous materials: synthesis and applications.Chemical Reviews, 121(20),pp.12278 12326.
https://doi.org/10.1021/acs.chemrev.1c00243 - Cheng, W., Tang, X., Zhang, Y., Wu, D. and Yang, W., 2021. Applications of metal-organic framework (MOF)-based sensors for food safety: Enhancing mechanisms and recent advances. Trends in Food Science & Technology, 112, pp.268-282.
https://doi.org/10.1016/j.tifs.2021.04.004 - Habib, N., Durak, Ö., Uzun, A. and Keskin, S., 2023. Incorporation of a pyrrolidinium-based ionic liquid/MIL-101 (Cr) composite into Pebax sets a new benchmark for CO2/N2 selectivity. Separation and Purification Technology, p.123346.
https://doi.org/10.1016/j.seppur.2023.123346 - Guo, X., Huang, H., Ban, Y., Yang, Q., Xiao, Y., Li, Y., Yang, W. and Zhong, C., 2015. Mixed matrix membranes incorporated with amine-functionalized titanium-based metal-organic framework for CO2/CH4 separation. Journal of Membrane Science, 478, pp.130-139.
https://doi.org/10.1016/j.memsci.2015.01.007 - Wu, H.Q., Yan, C.S., Luo, F. and Krishna, R., 2018. Beyond crystal engineering: Significant enhancement of C2H2/CO2 separation by constructing composite material. Inorganic Chemistry, 57(7), pp.3679-3682.
https://doi.org/10.1021/acs.inorgchem.8b00341 - Liu, H., Ding, W., Lei, S., Tian, X. and Zhou, F., 2019. Selective adsorption of CH4/N2 on Ni-based MOF/SBA 15 composite materials. Nanomaterials, 9(2), p.149.
https://doi.org/10.3390/nano9020149 - Raptopoulou, C.P., 2021. Metal-Organic Frameworks: Synthetic Methods and Potential Applications. Materials 2021, 14, 310.
https://doi.org/10.3390/ma14020310 - Bendt, S., Hovestadt, M., Böhme, U., Paula, C., Döpken, M., Hartmann, M. and Keil, F.J., 2016. Olefin/Paraffin Separation Potential of ZIF‐9 and ZIF‐71: A Combined Experimental and Theoretical Study. European Journal of Inorganic Chemistry, 2016(27), pp.4440-4449.
https://doi.org/10.1002/ejic.201600695 - Li, L., Wen, H.M., He, C., Lin, R.B., Krishna, R., Wu, H., Zhou, W., Li, J., Li, B. and Chen, B., 2018. A metal-organic framework with suitable pore size and specific functional sites for the removal of trace propyne from propylene. Angewandte Chemie, 130(46), pp.15403-15408.
https://doi.org/10.1002/ange.201809869 - Cadiau, A., Adil, K., Bhatt, P.M., Belmabkhout, Y. and Eddaoudi, M., 2016. A metal-organic framework-based splitter for separating propylene from propane. Science, 353(6295), pp.137-140.
https://doi.org/10.1126/science.aaf6323 - Xiang, F., Marti, A.M. and Hopkinson, D.P., 2018. Layer by layer assembled polymer/MOF membrane for H2/CO2 separation. Jour-nal of Membrane Science, 556, pp.146-153.
https://doi.org/10.1016/j.memsci.2018.03.081 - Jia, Z., Yan, Z., Zhang, J., Zou, Y., Qi, Y., Li, X., Li, Y., Guo, X., Yang, C. and Ma, L., 2020. Pore size control via multiple-site alkylation to homogenize sub-nanoporous covalent organic frameworks for efficient sieving of xenon/krypton. ACS Applied Materials & Interfaces, 13(1), pp.1127-1134.
https://doi.org/10.1021/acsami.0c14610 - Chen, X., Plonka, A.M., Banerjee, D., Krishna, R., Schaef, H.T., Ghose, S., Thallapally, P.K. and Parise, J.B., 2015. Direct observation of Xe and Kr adsorption in a Xe-selective microporous metal-organic framework. Journal of the American Chemical Society, 137(22), pp.7007-7010.
https://doi.org/10.1021/jacs.5b02556 - Si, Y., He, X., Jiang, J., Duan, Z., Wang, W. and Yuan, D., 2021. Highly effective H2/D2 separation in a stable Cu based metal organic framework. Nano Research, 14, pp.518-525.
https://doi.org/10.1007/s12274-019-2571-9 - Chen, X., Liu, M., Zhang, L., Zhou, Y., Ping, E. and Ding, C., 2021. New gas chromatographic packing ZIF-67@ NH2-SiO2 for separation of hydrogen isotope H2/D2. International Journal of Hydrogen Energy, 46(24), pp.13029-13037.
https://doi.org/10.1016/j.ijhydene.2021.01.153 - Ping, E., Chen, X., Zhou, Y., Zhang, L., Kong, L. and Chen, N., 2023. H2/D2 Separation Using UTSA-16@ CAU-10-H@ γ-AlOOH Composites as the Stationary Phase in Gas Chromatography via the Additive Effects of Kinetic Sieving and Chemical Affinity Quantum Sieving. Inorganic Chemistry.
https://doi.org/10.1021/acs.inorgchem.2c03795 - Jiang, M., Cui, X., Yang, L., Yang, Q., Zhang, Z., Yang, Y. and Xing, H., 2018. A thermostable anion-pillared metal-organic framework for C2H2/C2H4 and C2H2/CO2 separations. Chemical Engineering Journal, 352, pp.803-810.
https://doi.org/10.1016/j.cej.2018.07.104 - Lei, C., Gao, J., Ren, W., Xie, Y., Abdalkarim, S.Y.H., Wang, S., Ni, Q. and Yao, J., 2019. Fabrication of metal-organic frameworks@ cellulose aerogels composite materials for removal of heavy metal ions in water. Carbohydrate polymers, 205, pp.35-41.
https://doi.org/10.1016/j.carbpol.2018.10.029 - Abdollahi, N., Razavi, S.A.A., Morsali, A. and Hu, M.L., 2020. High capacity Hg (II) and Pb (II) removal using MOF-based nanocomposite: Cooperative effects of pore functionalization and surface charge modulation. Journal of hazardous materials, 387, p.121667.
https://doi.org/10.1016/j.jhazmat.2019.121667 - Gul Zaman, H., Baloo, L., Kutty, S.R., Aziz, K., Altaf, M., Ashraf, A. and Aziz, F., 2023. Insight into microwave-assisted synthesis of the chitosan-MOF composite: Pb (II) adsorption. Environmental Science and Pollution Research, 30(3), pp.6216-6233.
https://doi.org/10.1007/s11356-022-22438-6 - Xue, J., Xu, M., Gao, J., Zong, Y., Wang, M. and Ma, S., 2021. Multifunctional porphyrinic Zr-MOF composite membrane for high-performance oil-in-water separation and organic dye adsorption/photocatalysis. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 628, p.127288.
https://doi.org/10.1016/j.colsurfa.2021.127288 - Qu, W., Wang, Z., Wang, X., Wang, Z., Yu, D. and Ji, D., 2023. High-hydrophobic ZIF-67@ PLA honeycomb aerogel for efficient oil-water separation. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 658, p.130768.
https://doi.org/10.1016/j.colsurfa.2022.130768 - Wang, H., Meng, J., Li, F. and Li, T., 2023. Graphitic carbon nitride/metal-organic framework composite functionalized cotton for efficient oil-water separation and dye degradation. Journal of Cleaner Production, 385, p.135758.
https://doi.org/10.1016/j.jclepro.2022.135758 - Tu, X., Xie, Y., Ma, X., Gao, F., Gong, L., Wang, D., Lu, L., Liu, G., Yu, Y. and Huang, X., 2019. Highly stable reduced graphene oxide-encapsulated Ce-MOF composite as sensing material for electrochemically detecting dichlorophen. Journal of Electroanalytical Chemistry, 848, p.113268.
https://doi.org/10.1016/j.jelechem.2019.113268 - Zhou, J., Li, X., Yang, L., Yan, S., Wang, M., Cheng, D., Chen, Q., Dong, Y., Liu, P., Cai, W. and Zhang, C., 2015.TheCu MOF 199/single walled carbon nanotubes modified electrode for simultaneous determination of hydroquinone and catechol with extended linear ranges and lower detection limits. Analytica chimica acta, 899, pp.57-65.
https://doi.org/10.1016/j.aca.2015.09.054 - Wang, H., Hu, Q., Meng, Y., Jin, Z., Fang, Z., Fu, Q., Gao, W., Xu, L., Song, Y. and Lu, F., 2018. Efficient detection of hazardous catechol and hydroquinone with MOF rGO modified carbon paste electrode. Journal of hazardous materials, 353, pp.151-157.
https://doi.org/10.1016/j.jhazmat.2018.02.029 - Dehdashtian,S., Hashemi, B. and Aeenmehr, A., 2019. The application of perlite/cobalt oxide/reduced graphene oxide (PC-rGO)/metal organic framework (MOF) composite as electrode modifier for direct sensing of anticancer drug idarubicin. IEEE Sensors Journal, 19(24), pp.11739-11745.
https://doi.org/10.1109/JSEN.2019.2937400 - Li, C., Xu, X., Xing, J., Wang, F., Shi, Y., Zhao, X., Liu, J., Yang, Y. and Zhao, Z., 2023. A fluorescent dual-emissions UiO-66-NH2@ TpTt-COF core-shell composite for sensitive and optosmart sensing of tetracycline. Applied Surface Science, p.156455.
https://doi.org/10.1016/j.apsusc.2023.156455 - Bhardwaj, S.K., Mohanta, G.C., Sharma, A.L., Kim, K.H. and Deep, A., 2018. A three phase copper MOF graphene polyaniline composite for effective sensing of ammonia. Analytica chimica acta, 1043, pp.89-97.
https://doi.org/10.1016/j.aca.2018.09.003 - Zheng, X., Zhao, Y., Jia, P., Wang, Q., Liu, Y., Bu, T., Zhang, M., Bai, F. and Wang, L., 2020. Dual-emission Zr-MOF-based composite material as a fluorescence turn-on sensor for the ultrasensitive detection of Al3+. Inorganic Chemistry, 59(24), pp.18205-18213.
https://doi.org/10.1021/acs.inorgchem.0c02674 - Karuppasamy, K., Sharma, B., Vikraman, D., Lee, J.H., Islam, M., Santhoshkumar, P. and Kim, H.S., 2022. Metal organic framework derived Ni4Mo/MoO2@C composite nanospheres as the sensing materials for hydrogen sulfide detection. Journal of Alloys and Compounds, 900, p.163421.
https://doi.org/10.1016/j.jallcom.2021.163421 - Roztocki, K., Bon, V., Senkovska, I., Matoga, D. and Kaskel, S., 2022. A Logic Gate Based on a Flexible Metal-Organic Framework (JUK‐8) for the Concomitant Detection of Hydrogen and Oxygen. Chemi-stry. A European Journal, 28(59), p.e202202255.
https://doi.org/10.1002/chem.202202255 - Lu, Q., Su, T., Shang, Z., Jin, D., Shu, Y., Xu, Q. and Hu, X., 2021. Flexible paper-based Ni-MOF composite/AuNPs/CNTs film electrode for HIV DNA detection. Biosensors and Bioelectronics, 184, p.113229.
https://doi.org/10.1016/j.bios.2021.113229 - Cheng, D., Xiao, X., Li, X., Wang, C., Liang, Y., Yu, Z., Jin, C., Zhou, N., Chen, M., Dong, Y. and Lin, Y., 2018. A non-enzymatic electrochemical sensing platform based on hemin@ MOF composites for detecting hydrogen peroxide and DNA. Journal of the Electrochemical Society, 165(16), p.B885.
https://doi.org/10.1149/2.0841816jes - Ma, Z.Z., Wang, Y.S., Liu, B., Jiao, H. and Xu, L., 2022. A Non Enzymatic Electrochemical Sensor of Cu@ Co MOF Composite for Glucose Detection with High Sensitivity and Selectivity. Chemosensors, 10(10), p.416.
https://doi.org/10.3390/chemosensors10100416