Seyedeh Elahe Badiei Khairabadi; Hasan Rahmani; Mohammad Bagher Miranzadeh; Rouhullah Dehghani; Fatemeh Atoof
Abstract
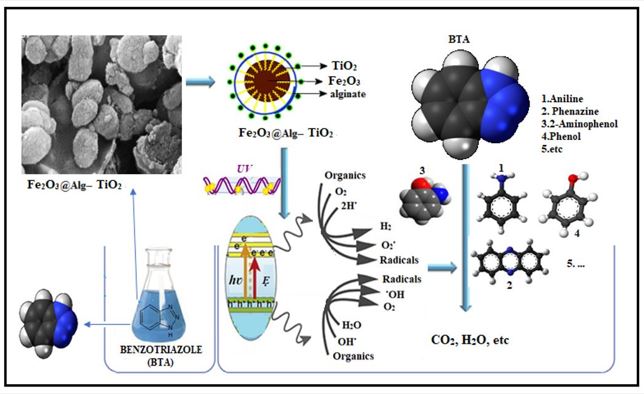
In this study, the decomposition of BTA was examined using a photocatalytic process (UV/Fe2O3@Alg-TiO2). The effects of various operational parameters such as the initial pH(3-7-10), catalyst dosage (0.05, 0.1, 0.15 g/L), BTA initial concentration (15–45 mg/L), UV intensity ( 2, 4, and 6 watts), ...
Read More
In this study, the decomposition of BTA was examined using a photocatalytic process (UV/Fe2O3@Alg-TiO2). The effects of various operational parameters such as the initial pH(3-7-10), catalyst dosage (0.05, 0.1, 0.15 g/L), BTA initial concentration (15–45 mg/L), UV intensity ( 2, 4, and 6 watts), and detention time (15,30, and 45 minutes) on the decomposition of BTA in the reactor with Batch conditions and in deionized water, real wastewater (three different matrices saline water, and real raw wastewater after activated sludge and after stabilization ponds) were investigated. SEM, XRD, and FTIR determined the synthesized catalyst's specifications. The results showed that 73.5% of BTA and 30% TOC was removed during the UV/Fe2O3@Alg-TiO2 photocatalytic process at the optimal condition (BTA=30 mg/L, pH = 3, UV = 4 Watt and catalyst dosage = 0.15 g/L) in deionized water. Also, the efficiency of the process in removing contaminants from the Saline, real raw sewage after activated, and after activated sludge and stabilization ponds was 23%, 47%, and 51%, respectively. The results showed that the toxicity decreased in 200 minutes that if sufficient time is provided to complete the reaction toxicity can be reduced. The results related to the amount of electrical energy consumed showed that considering both the advantages of BTA removal efficiency and energy consumption, the best performance was obtained in optimum conditions.