Rejani Padmavathiamma; Rani Kunjiraman Pillai
Abstract
Heavy metals are known to be toxic for living organisms even if they are present at low levels. Water pollution by heavy metals from industries is a dangerous environmental problem. Due to the ease, flexibility, and cost-effectiveness of the remediation process, adsorption has been widely implemented ...
Read More
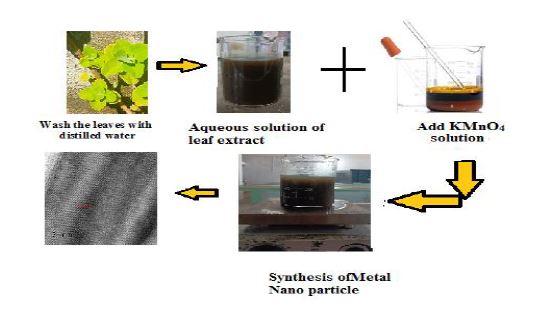
Heavy metals are known to be toxic for living organisms even if they are present at low levels. Water pollution by heavy metals from industries is a dangerous environmental problem. Due to the ease, flexibility, and cost-effectiveness of the remediation process, adsorption has been widely implemented in heavy metal wastewater treatment. In the present study, nanostructured manganese oxide was used for the removal of the heavy metal ions from aqueous solutions by a batch adsorption method and have been modelled using classical Langmuir and Freundlich adsorption isotherms. we have successfully synthesized an efficient adsorbent through a cost-effective and eco-friendly method. Bio synthesis is widely applied for the synthesis of nano materials . Various techniques such as XRD, FTIR, SEM EDX ,TEM and UV–VIS spectroscopy have been used to characterize the nanometal oxide. The obtained nano manganese oxide rods have very good adsorption efficiency due to the presence of some functionalities associated with the oxide material.
Nandini Venkat Iyer; Jayant A Kher; Shekhar D Bhame
Abstract
The present study features the synthesis of Dodecylbenzene sulphonic acid-doped polypyrrole tungsten oxide (PPy-WO3) nanocomposites and its photocatalyic studies on Methylene Blue (MB). The nanocomposites of PPy with a very low concentration of WO3 were prepared using 0.05 to 0.3 wt.%, of WO3 nanoparticles. ...
Read More
The present study features the synthesis of Dodecylbenzene sulphonic acid-doped polypyrrole tungsten oxide (PPy-WO3) nanocomposites and its photocatalyic studies on Methylene Blue (MB). The nanocomposites of PPy with a very low concentration of WO3 were prepared using 0.05 to 0.3 wt.%, of WO3 nanoparticles. The composites were characterized by using-ray Diffraction, FTIR and FESEM for phase identification, morphological studies. The electrical conduction of the nanostructured materials at room temperature exceeded that of PPy, with the electrical conductivity increasing linearly with higher WO3 concentrations. The band gap for the nanocomposite was found to be 2.12eV. The PL spectra substantiated the adequate segregation of charge carriers photoexcited in the samples. The nanocomposites were tested as photocatalysts for the degradation of methylene blue dye (MB). A two-step mechanism has been propsed for dye removal: adsorption (in the absence of UV light) and photodegradation on the photocatalyst surface (in the presence of UV light). The maximum removal efficiency for methylene blue dye was 98.31% in UV light for 0.16 g/L of the 0.3 wt.% PPy- WO3 nanocomposite, with a dye concentration of 5 mg/L. The addition of p-benzoquinone (*O2- scavenger) contributed to a significant decrease in the photodegradation efficiency of the catalyst, i.e.,63.21%, and thus can be believed as the main active species for the degradation of the methylene blue dye.

Pingal Sarmah; Khemnath Patir; Sonit Kumar Gogoi
Abstract
Here we report a simple and eco-friendly solvothermal synthesis of graphitic carbon nitride nanospheres (g-CNNS) at 180 oC. The synthesized g-CNNS is characterized by various analytical techniques such as FESEM, PXRD, BET isotherm, Zeta potential, EDX and FT-IR spectroscopy. The adsorption property of ...
Read More
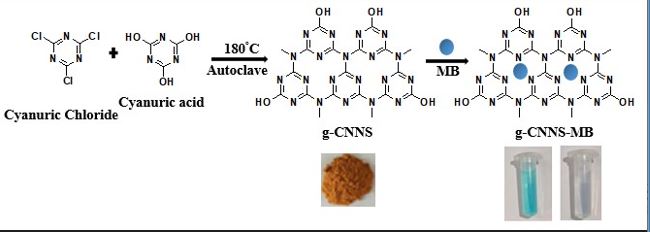
Here we report a simple and eco-friendly solvothermal synthesis of graphitic carbon nitride nanospheres (g-CNNS) at 180 oC. The synthesized g-CNNS is characterized by various analytical techniques such as FESEM, PXRD, BET isotherm, Zeta potential, EDX and FT-IR spectroscopy. The adsorption property of g-CNNS is studied using four different dyes in aqueous medium and found that g-CNNS is an efficient material for cationic dye adsorption. A Comprehensive investigation of the kinetics, isotherms and thermodynamics of methylene blue (MB) adsorption, is carried out. The adsorption of MB on g-CNNS is well described by Langmuir isotherm model, and the experimental data fits well with pseudo-second order kinetics. The high rate of adsorption (94.92% MB removal in 120 minute at neutral pH) is attributed to electrostatic interaction between negative charged g-CNNS and cationic organic dye molecule. Additionally, g-CNNS demonstrated good reusability, retaining its efficiency for at least three cycles. Over all our findings suggests that g-CNNS has potential as an efficient adsorbent for wastewater treatment.
Ahdieh Bavi; Mohammad Ghorbanpour; Mohammed Jaafar Ali Alatabe
Abstract
This study investigated the removal of methylene blue dye from aqueous solutions using Mg solid state exchanged bentonite. Parent bentonite and magnesium exchanged bentonite were characterized by scanning electron microscopy (SEM), X-ray diffraction (XRD), Brunauer Emmett Teller (BET), Energy dispersive ...
Read More
This study investigated the removal of methylene blue dye from aqueous solutions using Mg solid state exchanged bentonite. Parent bentonite and magnesium exchanged bentonite were characterized by scanning electron microscopy (SEM), X-ray diffraction (XRD), Brunauer Emmett Teller (BET), Energy dispersive X-ray spectroscopy (EDX), and Fourier transform infrared spectroscopy (FTIR). The parent bentonite showed the lowest removal efficiency (40.3%) and prepared magnesium exchanged bentonite for 10 min showed the highest removal efficiency (54.8%). Thus, dye absorption by modified bentonite increased compared to parent bentonite. After its modification by the ion exchange method, the specific surface was changed from 44.5 to 56.05 M2/g. EDX results indicate that the cation exchange process has been completed successfully. Ion exchange causes some minor changes in bentonite morphology and crystallinity. The best conditions for removing methylene blue using magnesium exchanged bentonite was pH 10, adsorbent dosage 0.05 mg/liter, initial concentration of solution 100 mg per liter, and contact time of 120 minutes with a Removal efficiency of 96.67. The Langmuir isotherm had a better correlation coefficient than the Freundlich adsorption model, which indicated the homogeneous surface of the adsorbent.

Noorallah Delijeh; Taher Yousefi; Hasan Aghayan; Ramin Yavari; Mehdi Parvini; Hossein Ghasemi mobtaker
Abstract
In the current work synthesis and modification of graphene oxide with Nickel Hexa Ferrocyanide (NiHCF) nanoparticles has been reported. The Graphene oxide- Nickel Hexa Ferrocyanide (GO-NiHCF) was used as an adsorbent to remove Cesium (Cs) ions from a simulated solution. The obtained product was characterized ...
Read More
In the current work synthesis and modification of graphene oxide with Nickel Hexa Ferrocyanide (NiHCF) nanoparticles has been reported. The Graphene oxide- Nickel Hexa Ferrocyanide (GO-NiHCF) was used as an adsorbent to remove Cesium (Cs) ions from a simulated solution. The obtained product was characterized with XRD, SEM, TGA, FTIR, and BET techniques. The SEM images and XRD pattern confirms the successful immobilization of Nickel Hexa Ferrocyanide on graphene oxide sheet. The cesium removal ability of GO-NiHCF was evaluated in batch mode. Effect of various parameters such as pH, initial concentration, contact time, and interferences ions were studied. The results cleared that the maximum adsorption for Cs removal was 240 mg g-1. Equilibrium modeling studies suggest that the data are reasonably and relatively fitted well to the Langmuir adsorption isotherm. Kinetic studies show that sorption process is fairly rapid and the kinetic data are fitted well to the pseudo-second order rate model. This composite offers strong potential in the field of elimination of Cs that requires rapid and complete decontamination.
Taher Yousefi; Ramin Yavari; D Rezaei Uchbelagh; Abbas Ariamanesh
Abstract
Al-Ce-Mn oxide samples were synthesized by the cathodic electrochemical method at current densities of 5, 15, and 35 mAcm-2. The XRD, SEM, and EDX techniques were used for the characterization of samples. The SEM images show that at high current density the one-dimensional(nanowire) structure and at ...
Read More
Al-Ce-Mn oxide samples were synthesized by the cathodic electrochemical method at current densities of 5, 15, and 35 mAcm-2. The XRD, SEM, and EDX techniques were used for the characterization of samples. The SEM images show that at high current density the one-dimensional(nanowire) structure and at low current density two-dimensional (nanosheet) structure were obtained. Moreover, the particle sizes are decreased with increasing the current density. The samples were applied for the uptake of fluorine (F-) ions from solutions. The influence of the contact time, initial fluoride concentration, and solution pH on the adsorption was investigated. The results showed more than 80 % of F- ions were uptake from solution during the three hours initial contact times and the uptake capacity has little change at pH below 6 and it has a sharp decline with increasing solution pH. The kinetic data were well fitted to the pseudo-second-order model and the equilibrium adsorption data was well described by the Langmuir isotherm model. The adsorption capacity was 48 mg/g at pH 6 and room temperature.
Dhanraj S Shirsath
Abstract
Magnetic nano adsorbent is cost-effective and easily synthesized in the laboratory by chemical Co-precipitation method that provides not only high adsorption capacity but also rapid adsorption rate. The magnetic nano adsorbents were synthesized by Ferric and Ferrous ions precursor solution in the presence ...
Read More
Magnetic nano adsorbent is cost-effective and easily synthesized in the laboratory by chemical Co-precipitation method that provides not only high adsorption capacity but also rapid adsorption rate. The magnetic nano adsorbents were synthesized by Ferric and Ferrous ions precursor solution in the presence of ammonium hydroxide. In the present investigation, a magnetic nano adsorbent has been employed for the removal of Pb (II) from an aqueous solution by batch adsorption technique along with photocatalysis. The different parametric study also carried out such as initial concentration of Pb (II), adsorbent dose, contact time, and Solution pH. The Pb (II) was fast adsorption and the equilibrium was achieved within 45 minutes. The amount Pb (II) adsorbed increases as the temperature increase. The optimal pH for Pb (II) was around 5.4 and for the removal of Pb (II) ions was up to 96.00%. The employed adsorbents were characterized by SEM, X-ray diffraction (XRD), Vibrating spinning magnetometer (VSM), and FTIR. The Kinetic of adsorption study was examined for the pseudo-first-order model and pseudo-second-order models. This Photocatalytic adsorption study obeys Pseudo second-order kinetic. The reusability and regeneration of magnetic nano adsorbents were studied and were recycled up to 87.00 %.
Maryam Ghasemi; Javad Azimi-Amin
Abstract
Here, graphene oxide was synthesized and reduced by lemon extract (source of vitamin C) in an aqueous solution under different pH (3 and 10). The lemon extract was prepared using a solvent-free method. The proposed mechanisms for the reduction of GO may be due to the nucleophilic attack of oxygen anion ...
Read More
Here, graphene oxide was synthesized and reduced by lemon extract (source of vitamin C) in an aqueous solution under different pH (3 and 10). The lemon extract was prepared using a solvent-free method. The proposed mechanisms for the reduction of GO may be due to the nucleophilic attack of oxygen anion of ascorbic acid to the epoxy or hydroxyl groups of GO sheets. Based on Raman spectra, with increasing the solution pH, the repair of the graphitic sp2 domain of the RGOs decreased. Reduced graphene oxide was successfully used to synthesize Fe3O4/RGO nanocomposite and remove Pb ions from aqueous media. The obtained Fe3O4/RGO nanocomposite was characterized by XRD, FTIR, SEM, and BET analysis. Based on these characterization techniques, reduced graphene oxide is distinguishably coated by Fe3O4 nanoparticles. The effect of different parameters: contact time (1-60 min), initial lead concentration (25-200 mg/L), adsorbent dosage (0.01-0.07 g), and the solution’s initial pH (1-8) on the removal of lead ions was studied using batch-scale tests. The maximum lead ion removal was achieved up to 90 % for Pb ions, respectively at optimum operating conditions viz. pH 5, Pb initial concentration 100 mg/L, Fe3O4/RGO dose 0.05 g, and contact time 30 min. Obtained results showed that the maximum adsorption capacity of Fe3O4/RGO for lead ion was 107.52 mg/g within 60 min of contact time. The adsorption behavior can be well described with the Langmuir isotherm and the pseudo-second-order models, indicating that the adsorption process was a monolayer and chemisorption adsorption.
Latha Vatchalan; Pandiselvam S.
Abstract
Paper industries will be using a different kind of dyes for producing various kinds of paper. Rhodamine–B (Rh-B) dye is one of the major sources of color effluents from textile and paper dyeing industries, and they cause long-term effects for on an aquatic environment. So this work mainly focused ...
Read More
Paper industries will be using a different kind of dyes for producing various kinds of paper. Rhodamine–B (Rh-B) dye is one of the major sources of color effluents from textile and paper dyeing industries, and they cause long-term effects for on an aquatic environment. So this work mainly focused on the synthesis of carbon nanoparticles from neem leaves, characterization and its adsorption and photocatalytic action against Rh-B dye. In this study, Carbon nanoparticles (C NPs) from neem leaves were synthesized for adsorption and photo catalytic degradation of Rh-B dye used in paper industries. The synthesized carbon NPs were characterized by the powder X-Ray Diffraction and Scanning Electron Microscopy. The adsorption and photocatalytic properties of Ccarbon NPs were examined for 1 hour by studying the degradation of the Rh-B dye at every 5 minutes time interval through UV-Visible spectrophotometer. The results obtained shown that carbonC NPs acts as better adsorbents than as photo catalysts.
Sajjad Lotfi; Bagher Aslibeiki; Mahmoud Zarei
Abstract
Tri-ethylene glycol (TEG) coated Fe3O4 nanoparticles ferrofluid were used for Pb (II) removal from simulated wastewater. The samples were synthesized using a modified co-precipitation method. The prepared samples were characterized by different techniques including X-ray diffraction, Rietveld method, ...
Read More
Tri-ethylene glycol (TEG) coated Fe3O4 nanoparticles ferrofluid were used for Pb (II) removal from simulated wastewater. The samples were synthesized using a modified co-precipitation method. The prepared samples were characterized by different techniques including X-ray diffraction, Rietveld method, FTIR, FESEM, TEM, VSM, TGA, BET and atomic adsorption experiments. The crystallinity of nanoparticles with a cubic spinel ferrite structure and absence of impurity phases were verified using X-ray diffraction and Rietveld method. The presence of TEG was approved by FTIR and thermogravimetric analysis. The VSM results showed that the bonding between the TEG molecules and ferrite nanoparticles, reduces the surface spin disorder, influences the morphology and magnetization, and consequently increases the Pb (II) removal efficiency to a high value of 97%. The obtained high value of adsorption capacity of q=363.4 mg.g-1 with R= 91 % and q=129.4 mg.g-1 with R=97 %shows effective role of TEG coating on Pb (II) adsorption. The interesting results of this study imply that the TEG coated ferrofluid sample is suitable candidate for practical applications.
Nik Rashida Nik Abdul Ghani; Siti Syakirah Sulaiman; Amina Tahreen; Mohammed Saedi Jami
Abstract
Arsenic contamination poses a major public health concern and harms the environment with its toxicity. Long term exposure to a high concentration of arsenic is harmful to human health as well as the environmental biodiversity. This study is aimed to fabricate and investigate the possibility of polyethersulfone-graphene ...
Read More
Arsenic contamination poses a major public health concern and harms the environment with its toxicity. Long term exposure to a high concentration of arsenic is harmful to human health as well as the environmental biodiversity. This study is aimed to fabricate and investigate the possibility of polyethersulfone-graphene oxide-polyvinyl pyrrolidone (PES-GO-PVP) nanocomposite adsorptive membrane and use it to enhance the removal of arsenic from wastewater. The nanocomposite membrane in this study was fabricated via the non-solvent induced phase separation (NIPS) method with the addition of polyvinylpyrrolidone (PVP) as a pore-forming agent. Based on the characterization results of GO through Fourier-Transform Infrared Spectroscopy (FTIR), X-ray diffraction (XRD) and Raman spectroscopy, the existence of high quantity of oxygen based functional groups with high degree of oxidation was observed, which indicated that the GO were well-synthesized. The characterization of the membrane indicated that the addition of GO and PVP could impact the membrane hydrophilicity and mechanical stability. Three adsorption parameters (initial concentration of arsenic, pH and contact time) were then optimized using a face-centred central composite design (FCCCD). The arsenic removal efficiency of 88.6 % was obtained with 55 mg/L of initial arsenic concentration, at pH 8 and 75 minutes of contact time between PES-GO-PVP membrane and the arsenic ion. The Langmuir isotherm model fitted the equilibrium data, describing the monolayer adsorption mechanism occurred on the surface of the membrane. Therefore, the results obtained in this study prove the suitability and promising potential of the nanocomposite membrane for effective removal of arsenic through adsorption.
Sumon Chakrabarty; Md. Anas Mahmud; Mosummath Hosna Ara; Shovon Bhattacharjee
Abstract
The present study aimed to synthesize copper oxide (CuO) nanoparticles (NPs), which were used as an adsorbent for the sequestration of Iron (Fe) (III) ion from aqueous solution. The synthesized NPs were characterized with the help of X-ray diffraction (XRD) spectroscopy, Field Emission scanning electron ...
Read More
The present study aimed to synthesize copper oxide (CuO) nanoparticles (NPs), which were used as an adsorbent for the sequestration of Iron (Fe) (III) ion from aqueous solution. The synthesized NPs were characterized with the help of X-ray diffraction (XRD) spectroscopy, Field Emission scanning electron microscopy (FESEM), and Energy-dispersive X-ray spectroscopy (EDS). The SEM and XRD analyses indicated the average size of CuO NPs were ~25 nm with a rod-like shape. Based on the batch experiments the maximum adsorption observed at pH 9 with removal efficiency 98.38%, initial metal ion concentration of 10 ppm, and contact time 60 min. This study also revealed that adsorption capacity increases when the concentration of adsorbents decreases. To specify the adsorption characteristics of CuO NPs, the adsorption equilibrium data were treated with Langmuir and Freundlich models, which demonstrated that the removal of Fe (III) ions was mostly favored by the physical process followed by the multilayer adsorption on the heterogeneous surface of the adsorbents. Finally, this study concludes that CuO NPs could be used as a promising material for the removal of Fe(III) ion from aqueous Solution.
Mahdieh Chegeni; Mozhgan Mehri; Mehdi Hosseini
Abstract
The MoS2/S-doped graphitic carbon nitride (MoS2/S-g-C3N4) was synthesized by a simple method and applied for methylene blue (MB) removal as an organic pollutant. The structure of MoS2/S-doped graphitic carbon nitride was characterized using FTIR, XRD, SEM, TGA and BET techniques. The accomplishment of ...
Read More
The MoS2/S-doped graphitic carbon nitride (MoS2/S-g-C3N4) was synthesized by a simple method and applied for methylene blue (MB) removal as an organic pollutant. The structure of MoS2/S-doped graphitic carbon nitride was characterized using FTIR, XRD, SEM, TGA and BET techniques. The accomplishment of MoS2/S-doped graphitic carbon nitride as an adsorbent was investigated to removal of MB from aqueous solution. The various parameters were studied such as: pH, initial MB concentration, adsorbent dose, temperature and time. The best findings were obtained at pH=8, 8 ppm MB concentration, 0.05 g MoS2/S-g-C3N4, 30 min and 22 ˚C. The Langmuir isotherm model was adopted with the obtained data. The kinetic studies were showed that the adsorption of methylene blue can be well described by the second-order equation. Maximum adsorption was calculated as 166 mg/g. The degradation of MB was studied by MoS2/S-doped graphitic carbon nitride under Light Emmition Diode (LED). Results showed that the MoS2/S-doped graphitic carbon nitride can enhance photocatalytic activity compared to pure g-C3N4 and MoS2/g-C3N4. The findings confirmed that the MoS2/S-doped graphitic carbon nitride can be applied as an efficient, low-cost adsorbent, and photocatalyst to remove of cationic dyes such as methylene blue.
Hessam Jafari; Gholam Reza Mahdavinia; Bagher Kazemi; Shahrzad Javanshir; Samira Alinavaz
Abstract
Abstract: In this project, new magnetic Fucus vesiculosus (m-FV) nanoparticles with a high adsorption capacity of cationic dyes were prepared. To reach a nanocomposite with effective performance, Fucus vesiculosus (FV) was modified using ultrasound. Then, the Fe2+/Fe3+ ions were co-precipitated in situ ...
Read More
Abstract: In this project, new magnetic Fucus vesiculosus (m-FV) nanoparticles with a high adsorption capacity of cationic dyes were prepared. To reach a nanocomposite with effective performance, Fucus vesiculosus (FV) was modified using ultrasound. Then, the Fe2+/Fe3+ ions were co-precipitated in situ to induce magnetic feature to FV particles. Solutions contaminated with the model cationic dyes, methylene blue (MB) and crystal violet (CV), were treated by employing m-FV particles. Study on time of dyes removal showed a fast removing rate of MB and CV, reaching equilibrium at 10 and 5 minutes, respectively. Analysis of experimental kinetic data by the pseudo-first-order and pseudo-second-order models indicated a well-describing of data by the pseudo-second-order model. The isotherm data of adsorption of both cationic dyes on m-FV were modeled and revealed a well-describing with the Langmuir model. According to the Langmuir model, maximum adsorption capacities of 577 mg/g for MB and 1062 mg/g for CV on m-FV observed. Easy recovery, good recyclability, pH-independent property, as well as the high capability in the removal of cationic dyes, the m-FV can be considered an effective and eco-friendly bioadsorbent in the treatment of dye contaminated solutions.
Peghah Ghomashi; Amir Charkhi; Mohammad Kazemeini; Taher Yousefi
Abstract
In this study adsorption of fluoride ion from high-fluorine solutions (
Read More
In this study adsorption of fluoride ion from high-fluorine solutions (
Zeinab Ibrahim; Yehya Mohsen; Joumana Toufaily; Wassim Rammal; Tayssir Hamieh; T. Jean Daou; Maria-Laura Foddis; bachar Koubaissy
Abstract
Activated Montmorillonite (AM) reveals as a low-cost and efficient adsorbent for the adsorption of nicotine and pyridine from aqueous solutions. In this study, the influence of several operation conditions (initial compounds concentration, volumetric flow rate, and height of bed) on the shape of breakthrough ...
Read More
Activated Montmorillonite (AM) reveals as a low-cost and efficient adsorbent for the adsorption of nicotine and pyridine from aqueous solutions. In this study, the influence of several operation conditions (initial compounds concentration, volumetric flow rate, and height of bed) on the shape of breakthrough curves and the mass transfer resistance was evaluated. Adsorption experiments were developed in order to determine the adsorption isotherm of the system, then the adsorption of pyridine and nicotine onto activated Montmorillonite in single and binary system has been studied using fixed bed reactor. In continuous adsorption, Results show that the maximum nicotine uptake 110 mg/g of AM was achieved through electrostatic attraction and hydrogen bond at a pH = 6.3, a flow rate of 1 ml/min and a height of bed equal to 12 mm. In binary mixtures, zeolites adsorption is governed primarily by the size of pollutants present in water. Thus, the bigger compound (in this case, Nicotine), was adsorbed more easily than the pyridine present in the mixture. Experimental data were fitted according to Fowler Guggenheim for the isotherms and Wolborska model for the breakthroughs. AM was regenerated by ethanol and the results show that about 94% of the adsorption capacity is maintained after three times of cyclic adsorption-desorption process.
Reza Jazini Zadeh; Mohammad Sayadi; Mohammad Reza Rezaei
Abstract
The present study was performed on the adsorption of 2,4-Dichlorophenoxyacetic acid from aqueous solutions by amine-modified magnetic nanoparticles. The adsorbent was synthesized by the co-precipitation method. The adsorbent properties of Fe3O4@SiO2-NH2 were investigated using XRD, FTIR, TGA, VSM and ...
Read More
The present study was performed on the adsorption of 2,4-Dichlorophenoxyacetic acid from aqueous solutions by amine-modified magnetic nanoparticles. The adsorbent was synthesized by the co-precipitation method. The adsorbent properties of Fe3O4@SiO2-NH2 were investigated using XRD, FTIR, TGA, VSM and TEM. Adsorbent efficacy was studied by investigating the effect of pH, initial concentration of pollutants, and adsorbent dose, and times. Finally, kinetics equations and isotherms models were used to describe the data. The results showed that the highest removal percentage was observed at pH 6 and the initial concentration 20 mg/l of 2,4-Dichlorophenoxyacetic acid. Adsorption capacity was increased by 65.3% bypassing the time from the beginning of the process to 60 min. The results from the study of isotherms and adsorption kinetics presented that the sorption procedure follows the pseudo-second-order kinetics and the Langmuir isotherm with R2> 99. The maximum adsorption capacity of Fe3O4@SiO2-NH2 is 116.3 mg/g. Besides, thermodynamic studies have shown that the adsorption process in the present study is endothermic and spontaneous. The experiments showed that Fe3O4@SiO2-NH2 synthesized nanoparticles could be an excellent method to remove 2,4- Dichlorophenoxyacetic acid contaminants from the aqueous solutions due to the high efficiency, simplicity, and lack of secondary contamination in the solution.
Arezoo Ghadi; Sharieh Hosseini
Abstract
The aim of this research work is to investigate sorption characteristic of synthesized polypyrrole / polyaniline (PPy / PANI) nano fiber for Cu (II) removal from paper mill wastewater. The sorption is carried out by batch method. The structural characterization of this sorbent was performed by SEM and ...
Read More
The aim of this research work is to investigate sorption characteristic of synthesized polypyrrole / polyaniline (PPy / PANI) nano fiber for Cu (II) removal from paper mill wastewater. The sorption is carried out by batch method. The structural characterization of this sorbent was performed by SEM and FTIR analysis. The optimum conditions of copper sorption from paper mill wastewater onto PPy / PANI were found to be: pH=6, contact time 12 min and 0.4 g of sorbent in 100 ml in wastewater. The kinetic studies showed that the sorption process was well described by the pseudo second order kinetic model. Also, Langmuir isotherm provided the best fit to the equilibrium data. Also the effect of temperature on the process was found that the temperature has positive effect on the process. Thermodynamic parameters showed that the sorption of copper onto nano fiber were feasible, spontaneous and endothermic under studied conditions.
Mohammad Reza Jalali Sarvestani; Zohreh Doroudi
Abstract
Reactive black 5 is a toxic dye that has adverse effects on the environmental ecosystems and the health of human beings. Therefore, its removal is very important. Among the reported methods adsorption gathered a huge attention in the recent years because of its simplicity and low-cost. In this review ...
Read More
Reactive black 5 is a toxic dye that has adverse effects on the environmental ecosystems and the health of human beings. Therefore, its removal is very important. Among the reported methods adsorption gathered a huge attention in the recent years because of its simplicity and low-cost. In this review paper, removal of reactive black 5 by adsorption method from waste waters was evaluated and all of the achievements from the past to the present were discussed in detail. The influence of important operational parameters on the adsorption efficiency of reactive black 5 such as pH, temperature, adsorbent dosage and initial dye concentration was investigated. In addition, the reported adsorbents for reactive black 5 were divided into different groups on the basis of their nature (like nanostructures, natural materials, by products and chitosan based adsorbents) and their important characteristics, including adsorption capacity, removal percentage, initial dye concentration, repeatability, the synthesis cost and optimized experimental parameters are compared with each other in detail. Moreover, important conclusions have been made from the surveyed literature and some suggestions are proposed for the future works.
Behgam Rahmanivahid; Freshteh Naderi; Hamed Nayebzadeh
Abstract
In this paper, Fe3O4 and MgFe2O4 as magnetic samples were successfully synthesized by coprecipitation and combustion methods, respectively, to be used for adsorption of toxic methyl orange molecules from the aqueous solution. Characteristics of the synthesized samples were evaluated using various analyses. ...
Read More
In this paper, Fe3O4 and MgFe2O4 as magnetic samples were successfully synthesized by coprecipitation and combustion methods, respectively, to be used for adsorption of toxic methyl orange molecules from the aqueous solution. Characteristics of the synthesized samples were evaluated using various analyses. The results of crystalline and surface bonding assessment confirmed the successful synthesis of both samples with an appropriate structure. Moreover, Fe3O4 presented higher magnetic properties and surface area as well as lower pore diameter than MgFe2O4 sample. However, the maximum adsorption of methyl orange was obtained for MgFe2O4 (56.54 mg/g) which was around three times of Fe3O4 in the same conditions. This may be related to larger pore diameter of MgFe2O4 and the ease of access to the internal surface of the adsorbent by the adsorbate molecules. Among the evaluated isotherms, the predicted Freundlich model showed well correlation with actual results of the adsorption process and the process could kinetically explained by the pseudo-second-order equation. Thermodynamic investigation of the process showed the adsorption of methyl orange was exothermic and spontaneous. The results revealed that MgFe2O4 sample (qmax = 181.34 mg/g) can be suggested as a good adsorbent for the removal of toxic dyes and water pollutants.
Mohammad Reza Jalali Sarvestani; Roya Ahmadi
Abstract
This paper investigated boron nitride nanocage performance as an adsorbent and sensing material for removal and detection of trinitroanisole by density functional theory. The calculated adsorption energies, Gibbs free energy changes (ΔGad), adsorption enthalpy changes (ΔHad) and thermodynamic ...
Read More
This paper investigated boron nitride nanocage performance as an adsorbent and sensing material for removal and detection of trinitroanisole by density functional theory. The calculated adsorption energies, Gibbs free energy changes (ΔGad), adsorption enthalpy changes (ΔHad) and thermodynamic equilibrium constants (Kth) revealed the adsorption process is experimentally feasible, spontaneous, exothermic and Irreversible. The highly negative adsorption energy values and bond lengths between B12N12 and trinitroanisole indicated the interaction between the adsorbate and the adsorbent is a chemisorption process. The N-O and C-N bond lengths and the density values showed that trinitroanisole complexes with boron nitride cage have higher explosive velocity and detonation pressure than the pure trinitroanisole without B12N12. The frontier molecular orbital parameters such as band gap, chemical hardness, electrophilicity, chemical potential and charge capacity were also studied and the findings proved B12N12 is an excellent sensing material for fabricating novel electrochemical and thermal sensors for detection of trinitroanisole.
Susan Samadi; Mostafa Mirzaie Shalmani; Amirabbas Zakaria
Abstract
The zeolite N.P./PEG/GO composite was synthesized by hydrothermal assisted sol-gel method to be used as Pb2+ and Cd2+ ions nano-adsorbent from the agricultural water. The FT-IR spectrum confirmed the expected microstructure of synthesized adsorbent. The SEM revealed the formation of zeolite nanoparticles ...
Read More
The zeolite N.P./PEG/GO composite was synthesized by hydrothermal assisted sol-gel method to be used as Pb2+ and Cd2+ ions nano-adsorbent from the agricultural water. The FT-IR spectrum confirmed the expected microstructure of synthesized adsorbent. The SEM revealed the formation of zeolite nanoparticles and the layer structure of graphene. The existence of expected elements and crystalline structures were confirmed by EDS and XRD analysis, respectively. The pH, contact time, temperature and adsorption amount was optimized in the removal process of lead (II) and cadmium ions. The results were 6, 20 min, 25 oC and 0.005g for Pb2+ and 4, 20 min, 25 oC and 0.005g for Cd2+ respectively. Adsorption capacity of Lead (II) and cadmium ions were 49.6 and 50.2 mg.g-1 of adsorbent, respectively. Interference ions don’t show any considerable effects on the efficiency of adsorption for both ions. The equilibrium data can well be fixed using both Langmuier and Freundlich equations.
Mohammad Reza Jalali Sarvestani; Roya Ahmadi
Abstract
In this study, the impact of doping graphene with silicon and germanium on the adsorption of Ag+ was evaluated by density functional theory. At the outset, the structures of silver, adsorbents and their derived products at ten different configurations were optimized geometrically. Then, IR and frontier ...
Read More
In this study, the impact of doping graphene with silicon and germanium on the adsorption of Ag+ was evaluated by density functional theory. At the outset, the structures of silver, adsorbents and their derived products at ten different configurations were optimized geometrically. Then, IR and frontier molecular orbital calculations were implemented on them and some important parameters such as adsorption energy, Gibbs free energy changes, enthalpy variations, the thermodynamic equilibrium constant, specific heat capacity, chemical hardness, energy gap and electrophilicity were obtained and inspected. The achieved results indicate that by doping graphene with silicon and germanium the adsorption process has become more spontaneous, exothermic and experimentally feasible. The influence of temperature on the adsorption procedure was also checked out and the results indicate that 298.15 K is the optimum temperature for the desired process at all of the evaluated configurations. The HOMO-LUMO related parameters reveal that the pure and also doped nano-adsorbents are not appropriate sensing material in the construction of conductometric sensors but they can act as an eminent neutral ion carrier in the development of a potentiometric ion selective electrode for determination of silver (I) cations.
Atiyeh Yazdani; Mohammad Sayadi; Ava Heidari
Abstract
The green synthesis of palladium oxide nanoparticles using Dictyota indica seaweed extract was investigated. Dictyotales is a large order in the brown algae (class Phaeophyceae). The color of the reaction mixture changed which indicated the formation of palladium oxide nanoparticles. UV-Visible, SEM, ...
Read More
The green synthesis of palladium oxide nanoparticles using Dictyota indica seaweed extract was investigated. Dictyotales is a large order in the brown algae (class Phaeophyceae). The color of the reaction mixture changed which indicated the formation of palladium oxide nanoparticles. UV-Visible, SEM, TEM, XRD, and FTIR analysis determined the characteristics of synthesized nanoparticles. The UV-Visible analysis showed the formation of palladium oxide nanoparticles. SEM and TEM analyses presented the palladium oxide nanoparticles have a spherical shape and based on DLS analysis with the average particle diameter of 19nm. The comparisons of the nanoparticle size with different biological synthesis were studied which revealed this methodology offer smallest size. The crystallographic parameters of the synthesized Pd nanoparticles were as follows: crystalline structure (cubic), space group (Fm-3m), and lattice parameter (a=5.6500, b=5.6500, c=5.6500, α=90, β=90, and γ=90). FTIR analysis indicated the presence of a palladium oxide group in the product. The removal efficacy of cadmium by the palladium oxide nanoparticles was investigated to optimize the pH, contact time, dose of adsorbent and concentration of cadmium. The results showed that optimum conditions for cadmium removal from water were obtained at pH 8, 500 mg/l adsorbent dose and 20 min contact time, wherein in these circumstances the removal of cadmium was 82.82%. The adsorption isotherms primacy was as follows, Langmuir R2=0.9904> Freundlich R2=0.9857> Temkin R2=0.8791.
Hossein Khoshsang; Ali Ghaffarinejad; Hojjat Kazemi; Sedighe Jabarian
Abstract
The magnetic mesoporous Fe3O4 and Fe3O4/Carbon Nanocomposite (Fe3O4/C) are synthesized by a facile hydrothermal method in one- step and are used for methylene blue dye removal. Nanomaterials are characterized by field-emission scanning electron microscope (FE-SEM), transition electron microscopy ...
Read More
The magnetic mesoporous Fe3O4 and Fe3O4/Carbon Nanocomposite (Fe3O4/C) are synthesized by a facile hydrothermal method in one- step and are used for methylene blue dye removal. Nanomaterials are characterized by field-emission scanning electron microscope (FE-SEM), transition electron microscopy (TEM), energy dispersive X-ray spectrometry (EDX), X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR) and Vibrating sample magnetometry (VSM). The specific surface area of the samples and mean pore diameter were measured via Brunauer–Emmett–Teller (BET) surface area measurement technique. To improve the adsorption performance some important parameters affecting dye removal are optimized. Maximum capacity for methylene blue (MB) adsorption on to Fe3O4 and Fe3O4/C Nanocompositeis 169.5 and 208.33 mg. g-1, respectively, which compared to some same recent reports, has a better adsorption capacity. Thermodynamic parameters (ΔGο, ΔΗο and ΔSο) were calculated and the result showed a spontaneous, endothermic and increase in randomness for dye adsorption. The obtained data has the best fitting with Langmuir isotherm and the kinetic analysis has the best fit by pseudo-second order model.



