Ali Bahari; Mohammad Ali Mousavi; Hosein Milani Moghadarm
Abstract
The use of nanotechnology ( and nanocomposites and nanohybrids) in replacing cement with nanomaterials due to environmental aspects and reduction of pollutants is presently an important issue and of course a significant problem in the whole world. In this regard, the effect of various weight percentages ...
Read More
The use of nanotechnology ( and nanocomposites and nanohybrids) in replacing cement with nanomaterials due to environmental aspects and reduction of pollutants is presently an important issue and of course a significant problem in the whole world. In this regard, the effect of various weight percentages of functionalized carbon nanotubes together with montmorillonite nanoparticles and with titanium oxide nanoparticles were studied as replacements for the cement to improve the mechanical and microstructural properties of hardened cement paste composites. In the present study, carbon nanotubes, TiO2, and montmorillonite nanoparticles were synthesized as additions into cement matrix, and their nano-structural characterizations were studied using the TEM, XRD, X-Powder, DSC-TGA, FTIR, and GPS132A techniques. The obtained results indicate that addition of nanomaterials to cement-based composites improves mechanical, electrical and nano-structural properties and the quality as well as emission factors of samples with optimization portion of nano-particles found by Response surface methodology (RSM) method.
Mansoor Kazemimoghadam; Zahra Amiri Rigi
Abstract
Nano pore ZSM-5type membranes were prepared on the outer surface of a porous-mullite tube by in situ liquid phase hydrothermal synthesis. The hydrothermal crystallization was carried out under an autogenous pressure, at a static condition and at a temperature of 180°C with tetra propyl ammonium bromide ...
Read More
Nano pore ZSM-5type membranes were prepared on the outer surface of a porous-mullite tube by in situ liquid phase hydrothermal synthesis. The hydrothermal crystallization was carried out under an autogenous pressure, at a static condition and at a temperature of 180°C with tetra propyl ammonium bromide (TPABr) as a template agent. The molar composition of the starting gel of ZSM-5 zeolite membrane was: SiO2/Al2O3=100, Na2O/Al2O3=0.292, H2O/Al2O3=40–65, TPABr/ SiO2=0.02-0.05. The zeolites calcinations were carried out in the air at 530°C, to burn off the template (TPABr) within the zeolites. X-ray diffraction (XRD) patterns of the membranes consisted of peaks corresponding to the support and zeolite. The crystal species were characterized by XRD, and morphology of the supports subjected to crystallization was characterized by scanning electron microscopy (SEM). Performance of Nano-porous ZSM-5 membranes was studied for separation of water–unsymmetrical dimethylhydrazine (UDMH) mixtures using pervaporation (PV). Finally, a comprehensive unsteady-state model was developed for the pervaporation of water-UDMH mixture by COMSOL Multiphysics software version 5.2. The developed model was strongly capable of predicting the effect of various dimensional factors on concentration and velocity distributions within the membrane module. The best ZSM-5 zeolite membranes had a water flux of 2.22 kg/m2.h at 27°C. The best PV selectivity for ZSM-5 membranes was obtained to be 55.
Siroos Shojaei; Somaye Khammarnia; Saeed Shojaei; Mojtaba Sasani
Abstract
Although dyes are widely used in textile industries, they are carcinogenic, teratogenic and mutagenic. Industries discharge their wastewater containing a variety of colors into water resources and make harmful effect on the environment. The present study aims to Evaluate removal of reactive red 198 by ...
Read More
Although dyes are widely used in textile industries, they are carcinogenic, teratogenic and mutagenic. Industries discharge their wastewater containing a variety of colors into water resources and make harmful effect on the environment. The present study aims to Evaluate removal of reactive red 198 by nanoparticle zero valent iron (NZVI) in the presence of hydrogen peroxide from aqueous solution. The effective parameters on the removal of dye such as the hydrogen peroxide concentration of NZVI, contact time, pH and dye concentration were investigated and optimized. According to the results, the combination of NZVI with hydrogen peroxide is more effective than single hydrogen peroxide. At pH = 4, contact time= 40 min, 200 M of hydrogen peroxide, dye concentration= 75 mg/L and concentration of NZVI 2g/L, color removal was achieved 91% approximately. Based on the results of experiments, using hydrogen peroxide- NZVI has high efficiency in removal of azo dye type.
Nika Gholamzadeh; Majid Peyravi; Mohsen Jahanshahi
Abstract
The olive mill wastewater (OMW) is generated from olive oil extraction in olive mills. It contains a very high organic load and considerable quantities of phytotoxicity compounds. Comprehensive articles with different methods have been published about the treatment of OMW. This paper reviews the recent ...
Read More
The olive mill wastewater (OMW) is generated from olive oil extraction in olive mills. It contains a very high organic load and considerable quantities of phytotoxicity compounds. Comprehensive articles with different methods have been published about the treatment of OMW. This paper reviews the recent reports on the variety methods of OMW treatment. Biological process, containing aerobic pre-treatment by using different cultures and anaerobic co-digestion with other sewage and also added external nutrient with optimum ratio attracted much attention in the treatment of OMW. However, advanced oxidation process (AOP) due to the high oxidation potential which causes destruction of organic pollutants, toxic and chlorinated compounds have been considered. Furthermore, membrane technologies consist of microfiltration, ultrafiltration and especially nanofiltrationin wastewater treatment are growing in recent years. They offer high efficiency and mediocre investments owing to novel membrane materials, membrane design technics, module figures and improvement of the skills. In addition, fouling reduces the membrane performances in time, which is a main problem of cost efficiency.
Leila Mahdavian
Abstract
The aim of this study is to investigate the performance of zinc oxide nano-cages (Zn12O12-NC) to detect and reduce nitrate (NO3-) ions from aqueous media and convert them to oxygen and nitrogen gases by the density functional theory (DFT) method on a B3LYP level with basis set of 6-31+G*. Due to the ...
Read More
The aim of this study is to investigate the performance of zinc oxide nano-cages (Zn12O12-NC) to detect and reduce nitrate (NO3-) ions from aqueous media and convert them to oxygen and nitrogen gases by the density functional theory (DFT) method on a B3LYP level with basis set of 6-31+G*. Due to the structure of the nano-cage (Zn12O12-NC), there are some location positions with different potential on it. The results showed that the first N atom of nitrate ion strongly prefers to be adsorbed on O atoms and O of nitrate ion is adsorbed on Zn atoms of the 4-membered ring (4-MR). The electronic, structure and thermodynamic properties of these conversions are calculated and investigated. The energy gap (Eg) of the Zn12O12-NC is dramatically reduced from 3.88 to 1.22 eV upon the adsorption of NO3- ion, suggesting that it is transformed to n-type semiconductor ascribed to the large charge transfer from the ion to the nano-cage and ions convert into oxygen and nitrogen on it. The data show that Zn12O12 nano-cage can be used to identify and reduce of nitrate ions from the environment and may be helpful in several fields of study such as sensors, catalysts, and field emission investigations.
Mansoor Kazemimoghadam; Zahra Amiri Rigi
Abstract
Effect of crystallization time and temperature on the membrane structure and performance has been investigated for Nano-pore Hydroxysodalite (HS) zeolite membranes. Molar composition of the starting gel of the HS zeolite membranes were: SiO2/Al2O3=1.0-5.0, Na2O/Al2O3=15-65, and H2O/Al2O3=500-1500. X-ray ...
Read More
Effect of crystallization time and temperature on the membrane structure and performance has been investigated for Nano-pore Hydroxysodalite (HS) zeolite membranes. Molar composition of the starting gel of the HS zeolite membranes were: SiO2/Al2O3=1.0-5.0, Na2O/Al2O3=15-65, and H2O/Al2O3=500-1500. X-ray diffraction (XRD) patterns of the membranes exhibited peaks corresponding to the support and the zeolite. The crystal species was characterized by XRD and the morphology of the supports subjected to crystallization was characterized by Scanning Electron Microscopy (SEM). Separation performance of HS zeolite membranes was studied for water-Ethanol mixtures using pervaporation (PV). The membranes showed good selectivity towards water in the water-Ethanol mixtures. Water permeates faster because of its preferential adsorption into the Nano-pores of the hydrophilic zeolite membrane. In PV of water-Ethanol mixtures, the membrane exhibits a hydrophilic behavior, with a high selectivity towards water and a good flux. The best flux and separation factor of the membranes were 2.05 kg/m2.h and 10000, respectively. Effect of operating condition (temperature, flow rate and pressure) on the membrane performance was investigated for HS zeolite membranes grown onto seeded mullite supports. Finally, a comprehensive 2D model was developed for the PV of water-Ethanol mixture through HS zeolite membrane using Finite Element Method (FEM). Effect of varying dimensional factors, temperature and feed flow rate on the PV performance was studied. The proposed model was masterfully capable of predicting concentration distribution within two sub-domains of feed and membrane.
Salman Ghasemi-Kahrizsangi; Ebrahim Karamian; Hassan Gheisari Dehsheikh; Ahmad Ghasemi-Kahrizsangi
Abstract
Due to exhibiting an excellent chemical resistance against basic environments at high temperature, good thermal shock resistance, thermodynamic stability in the presence of carbon, and a suitable abrasion resistance, MgO-CaO refractories are widely used in argon-oxygen decarburization furnaces in the ...
Read More
Due to exhibiting an excellent chemical resistance against basic environments at high temperature, good thermal shock resistance, thermodynamic stability in the presence of carbon, and a suitable abrasion resistance, MgO-CaO refractories are widely used in argon-oxygen decarburization furnaces in the metallurgy industry and cement rotary kilns. Furthermore, MgO-CaO refractories are beneficial to removing inclusions from molten steels; thus, they have been considered to be one of the effective refractory types for processing clean steel products. Also, MgO-CaO materials have become one of the attractive steelmaking refractories because of their low cost and high ore reserves. However, in spite of these primary advantages, the application of MgO-CaO refractories has not been popular due to its tendency to hydration when exposed to the atmosphere. In world most of MgO-CaO bricks producers used of organic components such as tar, pitch, and peck for produce MgO-CaO refractories. But during the application of these bricks in steel and cement and industrials, they released CO and CO2 gases to air and pollute the atmosphere. For this reason, recently some researcher investigate the effect of additive nanoparticles on MgO-CaO refractories performance. They reported the used of Nano-additive have acceptable results and additive nanoparticles can replace by aforementioned environment contaminating organic compounds. In this study, we reviewed all effort that done for improving the hydration resistance of MgO-CaO refractories by application of Nano-additives with an emphasis on the beneficial the use of additives nanoparticle for reduction of environmental pollution by various industries which used of MgO-CaO refractory bricks.
Kalpesh Isai; Vinod Shrivastava
Abstract
ZnO and 2%Fe-ZnO nanomaterials were prepared by using a low-cost sol-gel method. The synthesized nanomaterials were characterized by X-ray diffraction (XRD), Scanning Electron Microscopy (SEM) and Energy Dispersive X-ray Spectroscopy (EDX). The XRD and SEM studies reveal that the synthesized nanomaterials ...
Read More
ZnO and 2%Fe-ZnO nanomaterials were prepared by using a low-cost sol-gel method. The synthesized nanomaterials were characterized by X-ray diffraction (XRD), Scanning Electron Microscopy (SEM) and Energy Dispersive X-ray Spectroscopy (EDX). The XRD and SEM studies reveal that the synthesized nanomaterials have a hexagonal wurtzite structure with average crystalline size ~ 22-23 nm. EDX analysis confirmed the composition and purity of synthesized nanomaterial. The photocatalytic activity of synthesized nanomaterials was monitored using the spectrophotometric method. Also, the photocatalytic removal of methylene blue (MB) dye from its aqueous solution by using ZnO and 2%Fe-ZnO nanopowder under UV light irradiation was studied. The effect of various parameters such as pH of dye solution, dye concentration, contact time and catalyst dose were investigated. Results of the current study demonstrated that, the maximum degradation using ZnO was 86 % and that for 2%Fe-ZnO was 92 % (under the optimum condition initial dye concentration=10 mg/L and pH =2). This study showed that 2%Fe-ZnO is a promising and better photocatalysts than ZnO.
Bahareh Hakimi; Mohammad Ghorbanpour; Atabak Feizi
Abstract
ZnO/bentonite photocatalyst was synthesized using different methods, including: precipitation, liquid-state ion exchange (LSIE) and solid-state Ion exchange (SSIE) methods. In most studies done so far, LSIE and precipitation method have been used to prepare composites. The SSIE is a new method. The purpose ...
Read More
ZnO/bentonite photocatalyst was synthesized using different methods, including: precipitation, liquid-state ion exchange (LSIE) and solid-state Ion exchange (SSIE) methods. In most studies done so far, LSIE and precipitation method have been used to prepare composites. The SSIE is a new method. The purpose of this research is to produce nanocomposites with each of these methods and compare their photocatalytic activity. The physicochemical properties of the prepared photocatalysts were determined by scanning electron microscope (SEM), energy dispersive X-ray (EDX) and diffusive reflective spectroscopy (DRS) analysis. The Photocatalytic activity was evaluated by degradation of methyl orange (MO) with the prepared photocatalysts. SEM images showed that the ZnO particles were successfully distributed on the bentonite in the samples prepared by precipitation method. Nevertheless, after ion exchange-based methods, no particles were formed on the bentonite surface. This is because of very small size of ZnO particles which in the interlayer space of bentonite. The EDX and DRS analysis confirmed the formation of composites. The photodegradation of MO were 84, 85 and 87% for the composites prepared by LSIE, SSIE and precipitation methods, respectively. Ultimately, the photocatalytic composites prepared by the solid-state method are very bright due to their simple production, low price and short time due through direct heating of the reaction.
Mansoor Kazemimoghadam; Zahra Amiri-Rigi
Abstract
Membrane ultrafiltration (UF) is widely used in dairy industries like milk concentration and dehydration processes. The limiting factor of UF systems is fouling which is defined as the precipitation of solutes in the form of a cake layer on the surface of the membrane. In this study, the combined cake ...
Read More
Membrane ultrafiltration (UF) is widely used in dairy industries like milk concentration and dehydration processes. The limiting factor of UF systems is fouling which is defined as the precipitation of solutes in the form of a cake layer on the surface of the membrane. In this study, the combined cake filtration-complete blocking model was compared to cake filtration mechanism for flux data through ultrafiltration of skim milk at constant flow rate. The resistance data also was modeled using cake filtration model and standard blocking model. The effect of different trans-membrane pressures and temperatures on flux decline was then investigated. Based on the results obtained here, the combined complete blocking-cake formation model was in excellent agreement with experimental data. The cake filtration model also provided good data fits and can be applied to solutions whose solutes tend to accumulate on the surface of the membrane in the form of a cake layer. With increasing pressure, the differences between the model and experimental data increased.
Hamidreza Kazemeini; Asghar Azizian; Mohammad Hassan Shahavi
Abstract
This research aimed to assess the effect of biodegradable coating chitosan nano-gel/emulsion loaded by Bunium persicum essential oil and nisin on E. coli O157:H7 in rainbow trout fillet during 12 days at refrigeration (4˚C). Trout fillet Sample was divided into 6 groups after inoculation of bacteria ...
Read More
This research aimed to assess the effect of biodegradable coating chitosan nano-gel/emulsion loaded by Bunium persicum essential oil and nisin on E. coli O157:H7 in rainbow trout fillet during 12 days at refrigeration (4˚C). Trout fillet Sample was divided into 6 groups after inoculation of bacteria (E. coli O157:H7), including control (without any coating), coated with chitosan 2% and other groups including nano-emulsion chitosan 2%, Nano-emulsion of chitosan containing Bunium persicum essential oil (0.5%), Nano-gel of chitosan containing nisin (200 IU/g) and Nano-gel/emulsion of chitosan containing Bunium persicum essential oil (0.5%) and nisin (200 IU/g). The samples were stored at the cool condition, and the bacterial count was performed on days: 0, 1, 2, 4, 8, and 12. The mean number of the bacterial count was significantly different among treatment (p<0.001). The most significant inhibitory effect on the growth of E.coli O157:H7 was observed in chitosan Nano-emulsion coating containing Bunium persicum essential oil (0.5%) and nisin (200 IU/g). According to this study, it was concluded that the use of Nano-gel/emulsion of chitosan coating Bunium persicum essential oil and nisin could be effective on the decrease of E.coli O157:H7 growth in food.
Trifa Sheikhaghaiy; Bahram Golestani Eimani
Abstract
Given the gradual development of drug resistance in different bacterial species, it is necessary to search for new drugs with effective broad spectrum antimicrobial activity. Therefore, recent studies on various nanometal oxides such as copper oxide and on antibacterial peptides including nisin as antibacterial ...
Read More
Given the gradual development of drug resistance in different bacterial species, it is necessary to search for new drugs with effective broad spectrum antimicrobial activity. Therefore, recent studies on various nanometal oxides such as copper oxide and on antibacterial peptides including nisin as antibacterial agents are especially important. The present study aimed to investigate the synergistic effect of nisin-conjugated copper oxide nanoparticles (CuO NPs) on the genome of E. coli selected as a Gram-negative model. After being cultured in a Nutrient Broth medium, the bacteria were treated with CuO NPs at 15, 30, 40, and 60μg/mL, with nisin at 30, 60, 90, and 120μg/mL, and with nisin-conjugated CuO NPs at 10, 20, and 30μg/mL and were then incubated. The optical densities of the samples were read at 600nm and their DNA was extracted. RAPD-PCR was used to study genomic effects, and statistical analysis was performed employing NTSYS-PC based on the DICE coefficient, the similarity matrix, and the drawn dendrogram. Results showed that the combination of CuO NPs and nisin had synergistic effects and was able to inhibit growth more than either of them used alone. However, this combination had no synergistic effects on the genome and caused minimal changes in the DNA sequence.
Koilparambil Divya; Liya C. Kurian; Smitha Vijayan; Jisha Manakulam Shaikmoideen
Abstract
The emerging infectious diseases and the development of drug resistance in the pathogenic microorganism is a matter of serious concern. Despite the increased knowledge of microbial pathogenesis and application of modern therapeutics, the morbidity and mortality associated with the microbial infections ...
Read More
The emerging infectious diseases and the development of drug resistance in the pathogenic microorganism is a matter of serious concern. Despite the increased knowledge of microbial pathogenesis and application of modern therapeutics, the morbidity and mortality associated with the microbial infections still remains high. Therefore, there is a pressing demand to discover novel strategies and identify new antimicrobial agents to develop the next generation of drugs or agents to control microbial infections. The use of nanoparticles is gaining impetus in the present century as they possess defined chemical, optical and mechanical properties. In the present study green synthesis of silver nanoparticles by Escherichia coli has been done. Various parameters such as mixing ratio of culture supernatant and silver nitrate, media, temperature and pH for production of silver nanoparticles were optimised. The nanoparticles synthesised was characterized using SEM, FTIR and XRD. The antibacterial activity of silver nanoparticles synthesised using both pellet and supernatant against human pathogens Salmonella typhi, Vibrio cholerae, Bacillus subtilis and Klebsiella pneumoniae was analysed and MIC was calculated as 20µg and 50µg respectively.
Behgam Rahmanivahid; Freshteh Naderi; Hamed Nayebzadeh
Abstract
In this paper, Fe3O4 and MgFe2O4 as magnetic samples were successfully synthesized by coprecipitation and combustion methods, respectively, to be used for adsorption of toxic methyl orange molecules from the aqueous solution. Characteristics of the synthesized samples were evaluated using various analyses. ...
Read More
In this paper, Fe3O4 and MgFe2O4 as magnetic samples were successfully synthesized by coprecipitation and combustion methods, respectively, to be used for adsorption of toxic methyl orange molecules from the aqueous solution. Characteristics of the synthesized samples were evaluated using various analyses. The results of crystalline and surface bonding assessment confirmed the successful synthesis of both samples with an appropriate structure. Moreover, Fe3O4 presented higher magnetic properties and surface area as well as lower pore diameter than MgFe2O4 sample. However, the maximum adsorption of methyl orange was obtained for MgFe2O4 (56.54 mg/g) which was around three times of Fe3O4 in the same conditions. This may be related to larger pore diameter of MgFe2O4 and the ease of access to the internal surface of the adsorbent by the adsorbate molecules. Among the evaluated isotherms, the predicted Freundlich model showed well correlation with actual results of the adsorption process and the process could kinetically explained by the pseudo-second-order equation. Thermodynamic investigation of the process showed the adsorption of methyl orange was exothermic and spontaneous. The results revealed that MgFe2O4 sample (qmax = 181.34 mg/g) can be suggested as a good adsorbent for the removal of toxic dyes and water pollutants.
Aiman M.A Noman; Mohammed A. Alghobar; Sidduraiah Suresha
Abstract
In this study, p-n junction photocatalyst CuO/CeO2ZrO2 with different concentrations of CuO was prepared by auto solution combustion method using glycine as fuel. This method is simple, fast and cost effective compared with other preparation methods. The photocatalyst was characterized by X-ray diffraction ...
Read More
In this study, p-n junction photocatalyst CuO/CeO2ZrO2 with different concentrations of CuO was prepared by auto solution combustion method using glycine as fuel. This method is simple, fast and cost effective compared with other preparation methods. The photocatalyst was characterized by X-ray diffraction (XRD), energy-dispersive spectrometer (EDS), UV-vis DRS. The assembly of p-type CuO nanoparticles produces a large number of nano p–n junction heterostructures on the surface of the CeO2ZrO2 nanocrystals, where CuO and CeO2ZrO2 form p- and n-type semiconductors. The experimental results reveal that p–n junction CuO/CeO2-ZrO2 heterojunction nanostructures exhibit much higher visible-light photocatalytic activities than the n-CeO2-ZrO2 for the removal of dye from industerial waste water. The photocatalytic activity of the p-n CuO/CeO2-ZrO2 heterojunction photocatalyst was evaluated using the degradation of aqueous methylene blue solution (MB) under visible light irradiation(λ>420 nm). The photo-degradation rate of this catalyzed is much faster than those occurring on n-type CeO2ZrO2. The sample with a p-n CuO/ CeO2-ZrO2 molar ratio of 0.021 presented the best photocatalytic activity, which was 30% higher than that of n-type CeO2ZrO2. The heat treatment condition also influences the photocatalytic activity strongly, and the best preparation condition is about 400ºC for 4h.
Sheetal .; Savita Sihag; Monika Yadav; Jitender Pal
Abstract
This study is focused on the synthesis and characterization of cotton straw residue. Nanocellulose was synthesized by chemical method and followed by ultrasonication and cryocrushing. The results of the present study show that the cotton straw residue consists of lignin (27%), hemicellulose (15%), cellulose ...
Read More
This study is focused on the synthesis and characterization of cotton straw residue. Nanocellulose was synthesized by chemical method and followed by ultrasonication and cryocrushing. The results of the present study show that the cotton straw residue consists of lignin (27%), hemicellulose (15%), cellulose (32%), and ash content (2.3%). Nanocellulose was characterized by FTIR, XRD, FESEM, TEM, DSC, TGA, and AFM. Two aromatic rings were observed at wavelength 1650.47cm-1 and 1436.53 cm-1 which indicates that there is a presence of cellulose in the prepared sample which was characterized by FTIR. The structural analysis shows that the material was amorphous and the nanocellulose crystallinity is 23 %. The morphological analysis using FESEM indicates even elongated fiber with a smooth surface and it contains pore in the nanocellulose of cotton residue. TEM analysis indicates that nanocellulose has an irregular shape with a circular rod-like structure of different sizes. The enthalpy of nanocellulose changes at 168.48℃ due to endothermic transition. TGA results show that the nanocellulose is degraded in the temperature range 300-355℃ and low thermal stability was observed during the experiment. AFM result shows the needle shape particle ( root square mean roughness = 0.1738nm) and the size of nanocellulose was observed 7.1 nm.
Mehrdad Hajian; Mohammad Rostamizadeh; Federico Galli
Abstract
Catalysis for environmental remediation is becoming of paramount importance as industrial and urban activities multiply, and by-products contaminate soils and wastewater. Effluents from industrial and urban activities have led to many environmental problems involving water contamination. Here, we propose ...
Read More
Catalysis for environmental remediation is becoming of paramount importance as industrial and urban activities multiply, and by-products contaminate soils and wastewater. Effluents from industrial and urban activities have led to many environmental problems involving water contamination. Here, we propose a new iron-incorporated metal-organic framework (MOF) photocatalyst to decontaminate water. The nanocatalyst was synthesized by the solvothermal method, and Fe was added to the structure as a promoter and active phase. In this study, we examined the degradation of methylene blue (MB) as a cationic azo dye. The nanocatalysts were characterized by XRD, FE-SEM, BET, NH3-TPD, and FTIR techniques. The results showed high crystallinity, a large specific surface area, and a uniform promoter distribution. At a pH = 9, a catalyst amount of 110 mg and an initial MB concentration in the effluent of 2 ppm resulted in the highest removal percentage (98 %). The kinetic analysis provided a quasi-first-order model that reasonably matched the experimental data (R2 = 95 %). The results verified the catalyst’s great capability for efficient and fast MB removal in 60 minutes of photocatalytic processing.
Seyedeh Elahe Badiei Khairabadi; Hasan Rahmani; Mohammad Bagher Miranzadeh; Rouhullah Dehghani; Fatemeh Atoof
Abstract
In this study, the decomposition of BTA was examined using a photocatalytic process (UV/Fe2O3@Alg-TiO2). The effects of various operational parameters such as the initial pH(3-7-10), catalyst dosage (0.05, 0.1, 0.15 g/L), BTA initial concentration (15–45 mg/L), UV intensity ( 2, 4, and 6 watts), ...
Read More
In this study, the decomposition of BTA was examined using a photocatalytic process (UV/Fe2O3@Alg-TiO2). The effects of various operational parameters such as the initial pH(3-7-10), catalyst dosage (0.05, 0.1, 0.15 g/L), BTA initial concentration (15–45 mg/L), UV intensity ( 2, 4, and 6 watts), and detention time (15,30, and 45 minutes) on the decomposition of BTA in the reactor with Batch conditions and in deionized water, real wastewater (three different matrices saline water, and real raw wastewater after activated sludge and after stabilization ponds) were investigated. SEM, XRD, and FTIR determined the synthesized catalyst's specifications. The results showed that 73.5% of BTA and 30% TOC was removed during the UV/Fe2O3@Alg-TiO2 photocatalytic process at the optimal condition (BTA=30 mg/L, pH = 3, UV = 4 Watt and catalyst dosage = 0.15 g/L) in deionized water. Also, the efficiency of the process in removing contaminants from the Saline, real raw sewage after activated, and after activated sludge and stabilization ponds was 23%, 47%, and 51%, respectively. The results showed that the toxicity decreased in 200 minutes that if sufficient time is provided to complete the reaction toxicity can be reduced. The results related to the amount of electrical energy consumed showed that considering both the advantages of BTA removal efficiency and energy consumption, the best performance was obtained in optimum conditions.
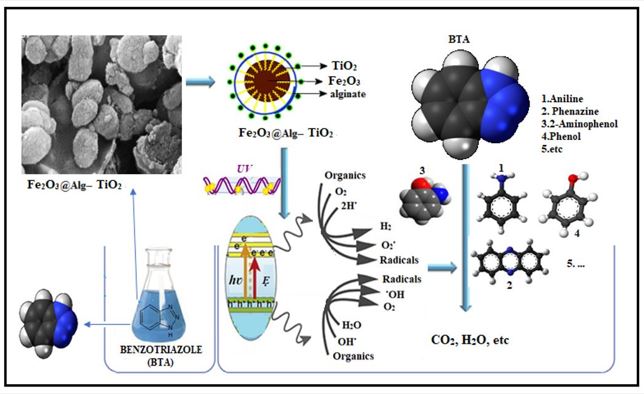
Seyed Hamed Mousavi; Fatemeh Ajoudani; Taher Yousefi; Amir Charkhi; Nima Rezaee Mojdehi; Ramin Yavari
Abstract
{[Dy(BTC)(H2O)]•DMF}n metal organic framework nanoparticles was synthezed through solvthermal method. The product was characterized by XRD, TG, BET, and SEM techniques. SEM images showed that the synthesized sample has semi-cubic particles with average size of 70 nm in length.For improve the gas ...
Read More
{[Dy(BTC)(H2O)]•DMF}n metal organic framework nanoparticles was synthezed through solvthermal method. The product was characterized by XRD, TG, BET, and SEM techniques. SEM images showed that the synthesized sample has semi-cubic particles with average size of 70 nm in length.For improve the gas separation performance,the MOF nano particles were dispersed in polydimethylsiloxane (PDMS) for preparation of mixed matrix membrane (MMM) on support of polyethersulphone (PES). The performance of obtained MMM in separation of NO, N2 and O2 gas were investigated, and the effect of MOF nanoparticles (5, 10, and 15% wt)and feed pressure (100-250 kPa) on permeability and selectivity were studied. It was found that the membrane performance is evaluated by addition of MOF nano particles in membrane (polymeric matrix), and the feed pressure have not important effect on separation. The performance (NO/N2 and NO/O2 selectivity) increased as the loading of MOF particles (up to 15% wt) being dispersed within the polymer matrices.
Ghader Hosseinzadeh
Abstract
In the current work, a novel ZnO-Cu2O heterojunction was synthesized from ZnO nanorods and Cu2O nanoparticles via hydrothermal route and was applied for the first time as a visible light active photocatalyst for decomposition of Paraoxon insecticide. Crystallinity, shape and size of particles, and optical ...
Read More
In the current work, a novel ZnO-Cu2O heterojunction was synthesized from ZnO nanorods and Cu2O nanoparticles via hydrothermal route and was applied for the first time as a visible light active photocatalyst for decomposition of Paraoxon insecticide. Crystallinity, shape and size of particles, and optical properties of the synthesized heterojunction nanocomposites were evaluated by XRD, FESEM, EDS, Mott-Schottky, photocurrent analysis and UV-Visible spectroscopy analyses. Based on the obtained results the ZnO-Cu2O heterojunction nanocomposite was successfully synthesized and compared to pure ZnO semiconductor has the enhanced photocatalytic efficiency. The nanocomposite with 40% weight percentage of Cu2O has the best photocatalytical activity of 0.0201 min-1, which could be related to the improvement of optical properties (increasing of the visible light harvesting ability) and the reduction of the recombination of the photoinduced electron-hole pairs. In addition, according to the radical trapping tests and Mott-Schottky experiments, superoxide radical was determined as the main oxidizing species for photocatalytic degradation of Paraoxon, and a type II charge transfer process was proposed for the improved photocatalytic activity.
Mona Nabizad; Ahmad Dadvand Koohi; Zahra Erfanipour
Abstract
In this study, alginate, magnetite, and hydroxyapatite were used to fabricate alginate-hydroxyapatite (Alg-Hap), alginate-Fe3O4 (Alg-Fe3O4), and alginate-magnetic hydroxyapatite (Alg-mHap) using ferric chloride (III) crosslinker to remove cefixime from an aqueous solution. FTIR, SEM, VSM, BET, and XRD ...
Read More
In this study, alginate, magnetite, and hydroxyapatite were used to fabricate alginate-hydroxyapatite (Alg-Hap), alginate-Fe3O4 (Alg-Fe3O4), and alginate-magnetic hydroxyapatite (Alg-mHap) using ferric chloride (III) crosslinker to remove cefixime from an aqueous solution. FTIR, SEM, VSM, BET, and XRD tests were used to determine the functional groups, morphology, magnetization behavior, surface area, and crystallinity of catalysts, respectively. The optimal pH for the Fenton reaction was determined to be 3.3 for Alg-Hap and Alg-Fe3O4 catalysts and 4 for Alg-mHap catalysts. Increases in the concentration of hydrogen peroxide (1 to 3 mM) and the amount of catalyst (50 to 90 gr/L) increased the percentage of degradation to approximately 8% and 6%, respectively. The degradation efficiency of cefixime by using Alg-mHap as the best catalyst in the Fenton process was achieved 91%, at optimum condition (pH of 4, catalyst amount of 90 gr/L, initial cefixime concentration of 5 mg/L, H2O2 concentration of 3 mM within 90 min). Moreover, the second-order kinetic equation fits the experimental data for cefixime degradation for all three catalysts. Furthermore, not only did the catalysts display a negligible iron leaching (0.92 mg/L for Alg-mHap) but also after three consecutive cycles, the catalysts indicated long-term stability. Comparison between synthesized catalysts and other methods proved its effectiveness.
Jacinth Mispa Kanagaraj; Regini Chelladurai; Subramaniam Perumal; Murugesan Rajamani
Abstract
A three component nanocomposite Poly(O-Toluidine) Zr(IV) based ion exchangers are synthesized by sol-gel method and characterized by Fourier transform-infrared spectra, , X-ray diffraction, scanning electron microscopy, thermo gravimetric analysis, ion exchange studies, conductivity and antimicrobial ...
Read More
A three component nanocomposite Poly(O-Toluidine) Zr(IV) based ion exchangers are synthesized by sol-gel method and characterized by Fourier transform-infrared spectra, , X-ray diffraction, scanning electron microscopy, thermo gravimetric analysis, ion exchange studies, conductivity and antimicrobial studies. The organic polymeric part of the composites provides mechanical and chemical stability whereas the inorganic part supports the ion-exchange behavior, thermal stability and also increases the electrical conductivity due to the good ion-exchange behavior of Zirconium (IV) molybdophosphate and Zirconium(IV) iodovanadate . The nano composite of POT/ Zr(IV) Zirconium(IV) iodovanadate exhibited an excellent ion exchange capacity value for Na+ is 4.84 meq g −1 and POT/Zr(IV) molybdo phosphate ion-exchanger has the value 4.60 meq/g . While compared to other nanocomposite ion-exchangers both the ion–exchangers have significant and effective ion-exchange behaviour . From the sorption studies and the distribution coefficient values , both the composite ion exchangers show maximum selectivity towards Pb2+.They can conjugate the mechanical properties of the organic polymers with intrinsic properties of the inorganic ion exchangers creating a new class of hybrid organic – inorganic materials with improvement in thermal stability and good electrical conductivity, ion – exchange capacity and also showed higher antimicrobial activity against Gram positive and Gram negative bacteria like Escherchia coli, Pseudomonas and Staphylococcus saprophitocus, which leads to their usage for environmental remediation like water purification.POT/Zr(IV) molybdophosphate and POT/Zr(IV) iodovanadate nanocomposite ion exchangers were used as promising ion exchangers and applied as electrochemically switchable ion exchanger for water treatment, especially water softening
Aklilu Melese; Kenaegzer Mulate; Abdu Hussen; Afework Hailekiros; Walelign Wubet
Abstract
A group of supramolecular solid materials known as "metal organic frameworks"(MOFs) are a type of hybrid networks made up of a variety of inorganic and organic linkers that are all tightly bonded to metal ions. These classes of compounds have a larger surface area with the benefit of variable pore sizes, ...
Read More
A group of supramolecular solid materials known as "metal organic frameworks"(MOFs) are a type of hybrid networks made up of a variety of inorganic and organic linkers that are all tightly bonded to metal ions. These classes of compounds have a larger surface area with the benefit of variable pore sizes, a diverse structure, and a lovely appearance. They are promising materials for a range of applications because they are easy to develop and have consistent, fine-tunable pore structures. Controlled mixing of MOFs with functional materials is resulting in the development of new multifunctional composites and hybrids that display unique properties that outperform those of their component parts as a whole. The structural characteristics, classification, The most widely used and successful strategies for MOFs composite synthesis are presented like (Encapsulation method, Solvothermal method, Solution impregnation, and Click chemistry (reaction) method), numerous characterization techniques and their applications MOFs composite have all been covered in this review. Crystals with extremely high porosity and good thermal and chemical stability can be produced by carefully choosing the MOF ingredients. Because of these properties, MOFs composites can be used for a wide range of applications, including, sensing toxic chemicals, drugs, gases, and trace metals, components of foods and many more and also for detection of different materials. This is a rapidly developing interdisciplinary research area therefore to present the current situation of the field; this article has covered recent achievements as well as new avenues to investigate the future scope and uses of MOFs composites/hybride.
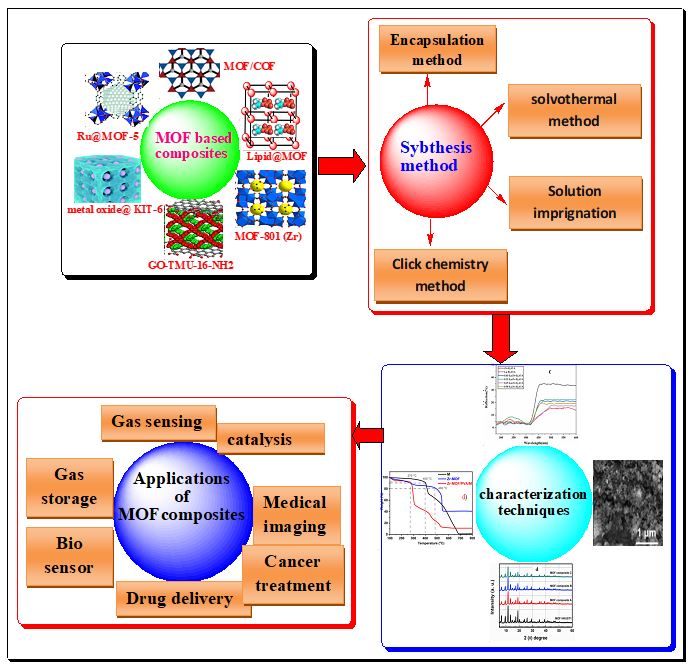
Bipul Sarkar; Palash Mondal
Abstract
Monitoring the levels of toxic Hg2+ metal ion in aquatic environment is important issue because this ion can have adverse effect in human health and environment. Therefore, detection of Hg2+ ion in water is very important issue for improving human health and water quality. Metallic nanoparticles such ...
Read More
Monitoring the levels of toxic Hg2+ metal ion in aquatic environment is important issue because this ion can have adverse effect in human health and environment. Therefore, detection of Hg2+ ion in water is very important issue for improving human health and water quality. Metallic nanoparticles such as gold and silver nanoparticles (AuNPs & AgNPs) have received much attention due to their colorimetric properties as well as localized surface plasmon resonance (LSPR) properties. AuNPs and AgNPs can easily change their colour (AuNPs: Red to Pink/Blue; AgNPs: Yellow to orange/red) which is easily discriminate by visual inspection. Functionalization of AuNPs and AgNPs offers an excellent application in many scientific worlds as the choice of ligands/functionalizing groups is outmost importance for their colloidal stability and function of the nanoparticles. In this review, we have discussed the colorimetric sensors of gold and silver nanoparticles based on functionalization of organic ligands, polymers, amino acid, and proteins for the detection of Hg2+ ion in aqueous medium.
Nandini V Iyer; Ganesh Agawane; Abhishjek Bhapkar; Jayant Kher; Shekhar Bhame
Abstract
In this paper, we report, the synthesis of conducting polymer nanocomposites of nickel oxide polypyrrole (NiO-PPy) doped with dodecyl benzene sulphonic acid for its application as a photocatalyst. In-situ polymerization of the pyrrole technique was employed along with oxidant ammonium persulphate and ...
Read More
In this paper, we report, the synthesis of conducting polymer nanocomposites of nickel oxide polypyrrole (NiO-PPy) doped with dodecyl benzene sulphonic acid for its application as a photocatalyst. In-situ polymerization of the pyrrole technique was employed along with oxidant ammonium persulphate and dodecyl benzene sulphonic acid as a dopant. The nanostructures were synthesized at different concentrations of NiO nanoparticles viz. 0.05 wt.%, 0.1 wt.%, 0.2 wt.% and 0.3 wt.%. The development of nanostructures was explored by Fourier Transform Infrared Spectrophotometer, Field Emission Scanning Electron Microscope, X-ray diffraction spectrometer, and electrical conductivity measurements. FTIR studies revealed a shift in the absorption band when pure PPy and NiO-PPy nanocomposites were studied, exhibiting the substantial interaction between the PPy network and the NiO. FE-SEM analysis demonstrated the consistent distribution of NiO with globular-shaped metal oxide materials in the PPy host template. The XRD studies for pure PPy revealed its amorphous nature while nanocomposites indicated the prominent NiO peaks arising from (111), (200) and (220) planes. The nanocomposites' direct electrical conductivity at room temperature was much higher than pure PPy. It was observed that the electrical conductivity for pure PPy was 0.409×10-5 S/cm while it substantially increased to 4.2×10-5 (S/cm) for 0.3% nanocomposite. The electrical studies revealed that the electrical conductivity goes on increasing with increased NiO concentration and then after a saturation point more PPy encapsulates the NiO and in turn reduces the electrical conductivity. With 50 mg of 0.3% nanocomposite, the photocatalytic degradation of the Methylene-Blue dye was 84.98%.

