Sumathi Harohally Paramesh; Veerendra Shetty Ananthpur; Nagaraju Rajendraprasad
Abstract
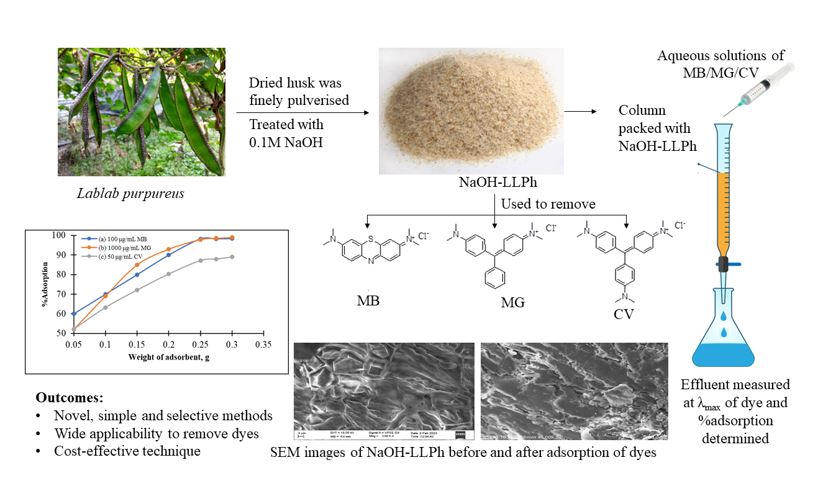
Novel results in this study showcase the utilization of sunlight-dried, ground Lablab purpureus husk (LLPh), treated with water and alkali, as a highly efficient bio-adsorbent for the removal of cationic dyes from aqueous solutions. Methylene blue (MB), malachite green (MG), and crystal violet (CV) were ...
Read More
Novel results in this study showcase the utilization of sunlight-dried, ground Lablab purpureus husk (LLPh), treated with water and alkali, as a highly efficient bio-adsorbent for the removal of cationic dyes from aqueous solutions. Methylene blue (MB), malachite green (MG), and crystal violet (CV) were effectively adsorbed onto NaOH activated LLPh (NaOH-LLPh) as bio-adsorbent. Employing the Chromatrap method within a column, successfully removed these dyes, while the surface morphology of the bio-adsorbent was elucidated through scanning electron microscopy (SEM) analysis. FTIR spectrometric data revealed valuable insights into the extent of adsorption. The impact of factors including adsorbate concentration, adsorbent dose, pH, contact time, and flow rate on the adsorption process was systematically studied and optimized. Up to 1000 µg/mL of MB and MG, 50 µg/mL of CV were found to be effectively removed by adsorption at pH 4-5, 3 and 2, respectively, at the flow rate of 1 mL/min. The results of kinetic studies and adsorption isotherms of above-mentioned dyes indicates that, all the three dyes follow the pseudo-second order kinetics. The adsorption of MB and MG are well fitted with the Langmuir isotherm model. The other dye CV suits with the Freundlich isotherm model. Based on the results, NaOH-LLPh, as an inexpensive and eco-friendly adsorbent, is suitable for the removal of cationic organic dyes from aqueous samples.
Hessam Jafari; Gholam Reza Mahdavinia; Bagher Kazemi; Shahrzad Javanshir; Samira Alinavaz
Abstract
Abstract: In this project, new magnetic Fucus vesiculosus (m-FV) nanoparticles with a high adsorption capacity of cationic dyes were prepared. To reach a nanocomposite with effective performance, Fucus vesiculosus (FV) was modified using ultrasound. Then, the Fe2+/Fe3+ ions were co-precipitated in situ ...
Read More
Abstract: In this project, new magnetic Fucus vesiculosus (m-FV) nanoparticles with a high adsorption capacity of cationic dyes were prepared. To reach a nanocomposite with effective performance, Fucus vesiculosus (FV) was modified using ultrasound. Then, the Fe2+/Fe3+ ions were co-precipitated in situ to induce magnetic feature to FV particles. Solutions contaminated with the model cationic dyes, methylene blue (MB) and crystal violet (CV), were treated by employing m-FV particles. Study on time of dyes removal showed a fast removing rate of MB and CV, reaching equilibrium at 10 and 5 minutes, respectively. Analysis of experimental kinetic data by the pseudo-first-order and pseudo-second-order models indicated a well-describing of data by the pseudo-second-order model. The isotherm data of adsorption of both cationic dyes on m-FV were modeled and revealed a well-describing with the Langmuir model. According to the Langmuir model, maximum adsorption capacities of 577 mg/g for MB and 1062 mg/g for CV on m-FV observed. Easy recovery, good recyclability, pH-independent property, as well as the high capability in the removal of cationic dyes, the m-FV can be considered an effective and eco-friendly bioadsorbent in the treatment of dye contaminated solutions.
siroos shojaei; Jamal Ahmadi; Meysam Davoodabadi Farahani; Bentolhoda Mehdizadehd; Mohammadreza Pirkamali
Abstract
The remaining dye in the wastewater is not desirable as it damages the ecosystem and nature, and also is very toxic. The Crystal Violet (CV) dye is toxic and potentially carcinogenic. In addition, it reduces light in water and prevents the process of photosynthesis of aquatic plants. Therefore, nanozeolite-X ...
Read More
The remaining dye in the wastewater is not desirable as it damages the ecosystem and nature, and also is very toxic. The Crystal Violet (CV) dye is toxic and potentially carcinogenic. In addition, it reduces light in water and prevents the process of photosynthesis of aquatic plants. Therefore, nanozeolite-X (NX) was utilized as an adsorbent to remove the CV; effects of pH, catalyst mass, sonication time, and concentration of dye were also investigated. Effects of variables on the removal efficiency were studied via the Central Composite Design (CCD) to determine the dye removal percentage. The quadratic model was selected to predict the removal efficiency using the software. Optimal conditions for CV removal from aqueous solution were: pH= 8, sonication time= 6 min, concentration of dye= 13 mg L-1, and catalyst mass= 0.26 g. In these circumstances, the recovery efficiency was 97.60%. The research results indicated that NX could be applied potentially for CV removal.
