Malak Barakat; Rassil Khoder; Fatima Kassir; Zeinab Harajli; Mouhiaddine Mohamed El Jamal; Azadeh Ebrahimian Pirbazari
Abstract
Sol–gel method was applied for synthesis of TiO2 nanoparticles in the existence of different volumes of ethanol (10–50 mL) with the purpose to find optimized synthesis conditions. Also, nickel doped TiO2 nanoparticles (Ni/TiO2 molar ratio: 0.1-1.0%) were prepared by the similar technique ...
Read More
Sol–gel method was applied for synthesis of TiO2 nanoparticles in the existence of different volumes of ethanol (10–50 mL) with the purpose to find optimized synthesis conditions. Also, nickel doped TiO2 nanoparticles (Ni/TiO2 molar ratio: 0.1-1.0%) were prepared by the similar technique but in the existence of 10 mL ethanol and heated at different temperatures (300 °C –600 °C). XRD, SEM/EDX, UV-Vis DRS, FTIR and Raman spectroscopy were applied to identify the structural and morphological characteristics of the as-synthesized samples. XR diffraction results verified that TiO2 samples prepared with various volumes of ethanol (10–50 mL) consist of anatase and brookite phases up to 500 °C and rutile phase at 600 °C. The intensity of brookite diffraction decreased with the increase of calcination temperatures. Also, the low ethanol volume favored for formation of rutile phase at 600 °C. The addition of Ni(II) during the preparation of TiO2 nanoparticles prevented the formation of rutile phase. The undoped samples were synthesized with 10 and 20 mL ethanol and treated at 500 °C displayed the best catalytic performance for photocatalytic treatment of E 131 VF dye solution (rate constant: 0.051 and 0.061 (a.u) respectively). Ni doped TiO2 samples displayed lower photoactivity and rate constant.
S. Sibmah Stalin; E.K. Kirupa Vasam Jino
Abstract
This present study involves the sonochemical and microwave synthesis of an efficient light harvesting nanocomposite photocatalyst, ZnO/GO embedded with copper nanoparticles. The synthesised nanocomposite was characterised by various spectroscopic methods like XRD, SEM, EDX, FT-IR, UV-DRS and fluorescence ...
Read More
This present study involves the sonochemical and microwave synthesis of an efficient light harvesting nanocomposite photocatalyst, ZnO/GO embedded with copper nanoparticles. The synthesised nanocomposite was characterised by various spectroscopic methods like XRD, SEM, EDX, FT-IR, UV-DRS and fluorescence analysis. The catalytic activity of the synthesised nanocomposite was tested using an organophosphorous pesticide, as the test solution and its percentage efficiency of degradation were studied. About 99 % of 40 ppm of Quinalphos pesticide could be degraded using ZnO/Cu/GO nanocomposite (3 mg/L) under visible light radiation within20 min at neutral pH. The presence of an intrinsic defects and the fluorescence property of the prepared nanocomposite were also detected. The degradation efficiency was estimated by COD and TOC measurements. The reaction rate followed pseudo - first order kinetics with a rate constant of 0.042 min-1. Furthermore, the composite has demonstrated a reusable feature and was utilised for eight cycles without any change in its activity. These findings has illustrated an ecofriendly, more stable and well organised photocatalytic nanocomposite, which could be preferred much for the treatment of industrial and agricultural waste water containing organic contaminants within a short span of time under irradiation using sunlight.
Anisha Russelraj; Sibmah Stalin; Kirupa Vasam Jino
Abstract
In view of growing environmental issues there is a requirement for the immediate eradication of pesticides and its residues from local and commercial streamlets. Quinalphos is one of the most common organophosphorous pesticide especially used in agricultural fields bring about a great impact in environment ...
Read More
In view of growing environmental issues there is a requirement for the immediate eradication of pesticides and its residues from local and commercial streamlets. Quinalphos is one of the most common organophosphorous pesticide especially used in agricultural fields bring about a great impact in environment thereby affecting the health of aquatic organisms and humans. Among the various type of method that has been implemented, photocatalytic reaction is considered as the most relevant technique for the removal of toxic organic contaminants. In this study, modified TiO2 has been synthesized by doping with Fe, S using a simple one-step sol-gel method. The incorporation of Fe3+ and the distribution of S nonmetal in TiO2 crystal lattice were affirmed by the XRD, SEM and FT-IR analysis. The band gap energy of Fe/S/TiO2 nanocomposite has been narrowed to 2.5eV and its photocatalytic activity was extended to the visible region. Thus Fe/S/TiO2 photocatalyst was employed for the eradication of Quinalphos and the complete mineralization was achieved in 12 min of visible light irradiation and it is analyzed by TOC analysis. Furthermore, the reusable feature of the catalyst was demonstrated to be utilised for twelve cycles. The penetration of pesticide residues on protein pockets of fishes was predicted by molecular docking. Furthermore, the reduction in toxicity level of the effluent was examined by using aquatic organisms. Thus Fe/S/TiO2 nanocomposite is an efficient photocatalyst to oxidize emerging organic contaminants due to its high synergetic effect of visible light absorbing tendency and low recombination effect of charge carriers.

Maryam Ghasemi; Javad Azimi-Amin
Abstract
Here, graphene oxide was synthesized and reduced by lemon extract (source of vitamin C) in an aqueous solution under different pH (3 and 10). The lemon extract was prepared using a solvent-free method. The proposed mechanisms for the reduction of GO may be due to the nucleophilic attack of oxygen anion ...
Read More
Here, graphene oxide was synthesized and reduced by lemon extract (source of vitamin C) in an aqueous solution under different pH (3 and 10). The lemon extract was prepared using a solvent-free method. The proposed mechanisms for the reduction of GO may be due to the nucleophilic attack of oxygen anion of ascorbic acid to the epoxy or hydroxyl groups of GO sheets. Based on Raman spectra, with increasing the solution pH, the repair of the graphitic sp2 domain of the RGOs decreased. Reduced graphene oxide was successfully used to synthesize Fe3O4/RGO nanocomposite and remove Pb ions from aqueous media. The obtained Fe3O4/RGO nanocomposite was characterized by XRD, FTIR, SEM, and BET analysis. Based on these characterization techniques, reduced graphene oxide is distinguishably coated by Fe3O4 nanoparticles. The effect of different parameters: contact time (1-60 min), initial lead concentration (25-200 mg/L), adsorbent dosage (0.01-0.07 g), and the solution’s initial pH (1-8) on the removal of lead ions was studied using batch-scale tests. The maximum lead ion removal was achieved up to 90 % for Pb ions, respectively at optimum operating conditions viz. pH 5, Pb initial concentration 100 mg/L, Fe3O4/RGO dose 0.05 g, and contact time 30 min. Obtained results showed that the maximum adsorption capacity of Fe3O4/RGO for lead ion was 107.52 mg/g within 60 min of contact time. The adsorption behavior can be well described with the Langmuir isotherm and the pseudo-second-order models, indicating that the adsorption process was a monolayer and chemisorption adsorption.
Sepideh Arbabi Bidgoli
Abstract
The etiological factors for increased risk of endocrine and reproductive disorders remain largely unclear but huge number of data from in vitro, in vivo and epidemiological studies, support the association of their incidence with long term exposure to endocrine disrupting chemicals /agrochemicals in ...
Read More
The etiological factors for increased risk of endocrine and reproductive disorders remain largely unclear but huge number of data from in vitro, in vivo and epidemiological studies, support the association of their incidence with long term exposure to endocrine disrupting chemicals /agrochemicals in the modern world. Engineered Nanomaterials (ENMs) could be considered as new alternatives to overcome the environmental challenges of endocrine disrupting pesticides and fertilizers and to reduce human health risks of cancer and endocrine toxicity based on their unique physicochemical properties. Carbon nanotubes (CNTs) are the initiative members of the big family of ENMs used for developing “Nanotechnology Based Agrochemical” but despite remarkable detoxifying effects mediated by CNTs, a number of controversies and key questions address the toxicity and endocrine disrupting properties of these authoritative agents which may introduce to the global markets new generation of as nanofertilizers ,nanoadsorbents and nanopesticides soon .The actual issue stems from limited number of studies in valid toxicology models on CNTs related endocrine disruption and absence of systematic reviews on CNTs exposure-mediated endocrine health hazards especially with respect to epidemiological and human data. In this direction this systematic review focused on the following sub-topics: (1) an overview on CNTs applications as novel agrochemicals (2) environmental risks and benefits of CNTs 3) toxicokinetic and toxicodynamic of CNTs (4) contribution of CNTs in pathogenesis of obesity, diabetes and cardiovascular effects 4) evidence on the involvement of CNTs in developmental and reproductive toxicities from in vitro and in vivo studies (5) conclusions and perspectives.
Rajni Lasyal; Shakunj Rajput
Abstract
In this study, the degradation of azo-dye acid orange 10 has been investigated using Polyvinylpyrrolidone and Brij-35 stabilized iridium oxide nanoclusters as catalysts. Simple chemical reduction method was used to synthesize the above-mentioned nanoclusters. The characteristics of the nanocatalysts ...
Read More
In this study, the degradation of azo-dye acid orange 10 has been investigated using Polyvinylpyrrolidone and Brij-35 stabilized iridium oxide nanoclusters as catalysts. Simple chemical reduction method was used to synthesize the above-mentioned nanoclusters. The characteristics of the nanocatalysts were determined by UV-visible spectrophotometer, TEM and XRD. The kinetic study has been carried out at λmax of reaction mixture i.e. 479 nm spectrophotometrically. The degradation follows first order kinetics with respect to oxidant and catalyst concentration while order is one at lower substrate concentration tending towards zero at higher concentration. The degradation kinetics has been supported by the derived rate law. The results showed that Polyvinylpyrrolidone stabilized iridium oxide nanoclusters outperformed Brij-35 stabilized iridium oxide nanoclusters, exhibiting the fastest degradation rate. The progress of the degradation process was monitored by UV-vis spectroscopy. Using Polyvinylpyrrolidone stabilized iridium oxide nanoclusters as a catalyst is a very promising approach for the remediation of acid orange 10 due to the fast degradation rate and high degradation efficiency. In addition, Polyvinylpyrrolidone stabilized iridium oxide nanoclusters can be easily recovered and recycled for three consecutive cycles. It can be inferred from this study that catalytic oxidation methods are active and environment-friendly for the remediation of dyes.
Sajjad Lotfi; Bagher Aslibeiki; Mahmoud Zarei
Abstract
Tri-ethylene glycol (TEG) coated Fe3O4 nanoparticles ferrofluid were used for Pb (II) removal from simulated wastewater. The samples were synthesized using a modified co-precipitation method. The prepared samples were characterized by different techniques including X-ray diffraction, Rietveld method, ...
Read More
Tri-ethylene glycol (TEG) coated Fe3O4 nanoparticles ferrofluid were used for Pb (II) removal from simulated wastewater. The samples were synthesized using a modified co-precipitation method. The prepared samples were characterized by different techniques including X-ray diffraction, Rietveld method, FTIR, FESEM, TEM, VSM, TGA, BET and atomic adsorption experiments. The crystallinity of nanoparticles with a cubic spinel ferrite structure and absence of impurity phases were verified using X-ray diffraction and Rietveld method. The presence of TEG was approved by FTIR and thermogravimetric analysis. The VSM results showed that the bonding between the TEG molecules and ferrite nanoparticles, reduces the surface spin disorder, influences the morphology and magnetization, and consequently increases the Pb (II) removal efficiency to a high value of 97%. The obtained high value of adsorption capacity of q=363.4 mg.g-1 with R= 91 % and q=129.4 mg.g-1 with R=97 %shows effective role of TEG coating on Pb (II) adsorption. The interesting results of this study imply that the TEG coated ferrofluid sample is suitable candidate for practical applications.
Lydia Rohmawati; Lytha Rizqika Lailia; Nugrahani Primary Putri; Munasir Nasir; Darminto Darminto
Abstract
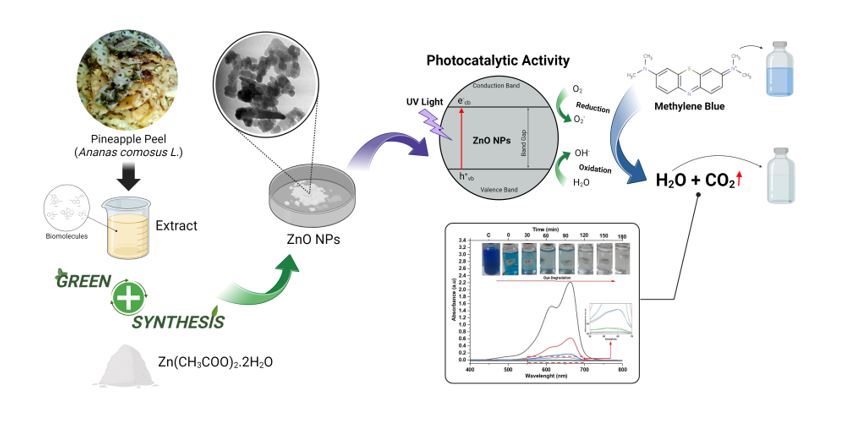
ZnO nanoparticles can be used as a photocatalyst in waste treatment because they have good photodegradation. Synthesis of ZnO nanoparticles using green synthesis method from pineapple peel extract, whose results were characterized by XRD (Xray Diffraction), FTIR (Fourier Transform Infra-red), FESEM (Field ...
Read More
ZnO nanoparticles can be used as a photocatalyst in waste treatment because they have good photodegradation. Synthesis of ZnO nanoparticles using green synthesis method from pineapple peel extract, whose results were characterized by XRD (Xray Diffraction), FTIR (Fourier Transform Infra-red), FESEM (Field Emission-Scanning Electron Microscopy), TEM (Transmission Electron Microscopy), Raman spectroscopy, photoluminescence, and a photocatalytic activity assay. This research showed that ZnO nanoparticles had wurtzite phase, alcohol functional groups, and phenol O-H, C=C alkenes, C-O, C-N, and Zn-O. ZnO nanoparticles had a particle size of 20.04 nm, a spherical shape, and a band gap energy of 3.28 eV. The Raman active mode E2(High) at 439.05 cm-1 confirmed the formation of pure phase wurtzite. Photoluminescence results indicated that two emission peaks at 392.07 nm and 595.07 nm were associated with defects such as oxygen and zinc vacancies. The results of the photocatalytic effectiveness test showed the highest percent degradation value of 99.86% at 180 minutes using UV light.
Zeinab Ibrahim; Yehya Mohsen; Joumana Toufaily; Wassim Rammal; Tayssir Hamieh; T. Jean Daou; Maria-Laura Foddis; bachar Koubaissy
Abstract
Activated Montmorillonite (AM) reveals as a low-cost and efficient adsorbent for the adsorption of nicotine and pyridine from aqueous solutions. In this study, the influence of several operation conditions (initial compounds concentration, volumetric flow rate, and height of bed) on the shape of breakthrough ...
Read More
Activated Montmorillonite (AM) reveals as a low-cost and efficient adsorbent for the adsorption of nicotine and pyridine from aqueous solutions. In this study, the influence of several operation conditions (initial compounds concentration, volumetric flow rate, and height of bed) on the shape of breakthrough curves and the mass transfer resistance was evaluated. Adsorption experiments were developed in order to determine the adsorption isotherm of the system, then the adsorption of pyridine and nicotine onto activated Montmorillonite in single and binary system has been studied using fixed bed reactor. In continuous adsorption, Results show that the maximum nicotine uptake 110 mg/g of AM was achieved through electrostatic attraction and hydrogen bond at a pH = 6.3, a flow rate of 1 ml/min and a height of bed equal to 12 mm. In binary mixtures, zeolites adsorption is governed primarily by the size of pollutants present in water. Thus, the bigger compound (in this case, Nicotine), was adsorbed more easily than the pyridine present in the mixture. Experimental data were fitted according to Fowler Guggenheim for the isotherms and Wolborska model for the breakthroughs. AM was regenerated by ethanol and the results show that about 94% of the adsorption capacity is maintained after three times of cyclic adsorption-desorption process.
Nik Rashida Nik Abdul Ghani; Siti Syakirah Sulaiman; Amina Tahreen; Mohammed Saedi Jami
Abstract
Arsenic contamination poses a major public health concern and harms the environment with its toxicity. Long term exposure to a high concentration of arsenic is harmful to human health as well as the environmental biodiversity. This study is aimed to fabricate and investigate the possibility of polyethersulfone-graphene ...
Read More
Arsenic contamination poses a major public health concern and harms the environment with its toxicity. Long term exposure to a high concentration of arsenic is harmful to human health as well as the environmental biodiversity. This study is aimed to fabricate and investigate the possibility of polyethersulfone-graphene oxide-polyvinyl pyrrolidone (PES-GO-PVP) nanocomposite adsorptive membrane and use it to enhance the removal of arsenic from wastewater. The nanocomposite membrane in this study was fabricated via the non-solvent induced phase separation (NIPS) method with the addition of polyvinylpyrrolidone (PVP) as a pore-forming agent. Based on the characterization results of GO through Fourier-Transform Infrared Spectroscopy (FTIR), X-ray diffraction (XRD) and Raman spectroscopy, the existence of high quantity of oxygen based functional groups with high degree of oxidation was observed, which indicated that the GO were well-synthesized. The characterization of the membrane indicated that the addition of GO and PVP could impact the membrane hydrophilicity and mechanical stability. Three adsorption parameters (initial concentration of arsenic, pH and contact time) were then optimized using a face-centred central composite design (FCCCD). The arsenic removal efficiency of 88.6 % was obtained with 55 mg/L of initial arsenic concentration, at pH 8 and 75 minutes of contact time between PES-GO-PVP membrane and the arsenic ion. The Langmuir isotherm model fitted the equilibrium data, describing the monolayer adsorption mechanism occurred on the surface of the membrane. Therefore, the results obtained in this study prove the suitability and promising potential of the nanocomposite membrane for effective removal of arsenic through adsorption.
Reddy Prasad Puthalapattu; Bebi Vakati; Sudheer Kaveti; Santosh Singh; Sreedhar Neelam Yugandhar
Abstract
Butachlor is a herbicide that belongs to the acetanilide family. It is widely used as a granule-based post-emergence herbicide on rice in India. As a result of the ongoing usage of these synthetic substances, soil fertility and soil organisms are declining. Differential pulse voltammetry was used to ...
Read More
Butachlor is a herbicide that belongs to the acetanilide family. It is widely used as a granule-based post-emergence herbicide on rice in India. As a result of the ongoing usage of these synthetic substances, soil fertility and soil organisms are declining. Differential pulse voltammetry was used to determine butachlor herbicide in soil samples with a modified glassy carbon electrode voltammetric sensor with palladium-supported multiwalled carbon nanotubes (Pd@MWCNTs). Scanning electron microscopy, energy dispersive x-ray spectroscopy, and X-ray diffraction spectroscopy were used to investigate the morphology of Pd@MWCNTs, while cyclic and differential pulse techniques were used to investigate the voltammetric properties. The butachlor herbicide under voltammetric investigation involves irreversible, two-electron reduction based on the protonation of the carbonyl group (>C=O). The voltammetric method was developed for the determination of butachlor in phosphate buffer solution at pH 6.0 as a supporting electrolyte. A good linear response to butachlor in the concentration ranging from 0.10 μg⸳mL−1 to 32.0 μg⸳mL−1 was observed, and a limit of detection of 0.044 μg⸳mL−1 was obtained with the calculation based on signal/noise=3. The suggested method was efficaciously applied for the detection of butachlor in soil samples.
Feizollah Dinarvand; Nematollah Jaafarzadeh; Mehdi Ahmadi Moghadam; Mohammad Bagher Miranzadeh; Nezam Mirzaei
Abstract
Introduction and Aims: Compounds containing dye are toxic, carcinogenic and mutagenic for aquatic organisms and lead to mutagenicity, carcinogenicity, and dysfunction of human beings’ kidney, liver, brain, reproductive system and central nervous system. Advanced oxidation processes can remove pollutants ...
Read More
Introduction and Aims: Compounds containing dye are toxic, carcinogenic and mutagenic for aquatic organisms and lead to mutagenicity, carcinogenicity, and dysfunction of human beings’ kidney, liver, brain, reproductive system and central nervous system. Advanced oxidation processes can remove pollutants faster than other processes due to active hydroxyl radical production. This study was aimed at investigating feasibility of dye removal using UVA/TiO2 process.Materials and methods: This study was done in a batch reactor and the effects of initial dye concentrations, TiO2 nanoparticles dosage, time, pH and interference compounds on efficiency of dye degradation was investigated. The Daphnia Magna as bioassay test and biodegradability index (BOD5/COD rate) were used for detoxification assessment.Results: The D.Y 50 dye effluent degradation at pH 2, 20 mg/l initial dye concentration and 1 g/l TiO2 catalyst was (lnC0/C= 1.4), (lnC0/C= 3) and (lnC0/C= 2.9) respectively. Dye removal rate by 50 mg/l COD concentration was (88%), Daphnia Magna mortality rate after maximum contact time (96 h) decreased from 96.7% to 43.3% and biodegradability index increased from 0.25 to 0.68.
Narasimha Raghavendra; Leena V Hublikar; Soumya R Chitnis; Rachel A Joseph; Deeksha S Sheelimath; Paravati S Pattan
Abstract
At present, the synthetic compounds are wide choice as indicators in various acid-base titrations. Due to strict environmental regulations, search for eco-friendly compounds as an effective indicator for various acid-base titrations was started. The current vocation highlights the exploit of Areca catechu ...
Read More
At present, the synthetic compounds are wide choice as indicators in various acid-base titrations. Due to strict environmental regulations, search for eco-friendly compounds as an effective indicator for various acid-base titrations was started. The current vocation highlights the exploit of Areca catechu seed extract as an efficacious indicator for various acid-base titrations and to determine their Ka values. The Areca catechu seed is easily available and easy to extract. The extraction performed by using the Soxhlet extraction apparatus. UV-Vis spectroscopy, FT-IR spectroscopy, XRD and physical properties (density, viscosity, surface tension and refractive index) and qualitative phytochemical screening was performed for the proper identification of the Areca catechu seed extract. The Areca catechu seed extract exhibits sharp colour change at the end point during the various acid-base titrations. The specific contrast between their colors in both the acid and alkali media made species present in the Areca catechu seed extract suitable for the eco-friendly indicator for four acid-base titrations. The end point obtained by the extract of Areca catechu seed coincide with end point obtained by standard synthetic indicator. Areca catechu seed is found to be economical, useful, accurate, simple and eco-friendly in nature.
Mahmood Hajiani; Efat Ezami; Mohammad Reza Rezaei
Abstract
Fe3O4/ ZnO/Ag magnetic nanocomposite was synthesized for the first time and its ability was evaluated for photocatalytic degradation of albumin in aqueous solutions under UV-A light. The resulting nanoparticles were then characterized using X-ray diffraction (XRD), scanning electron microscopy (FESEM), ...
Read More
Fe3O4/ ZnO/Ag magnetic nanocomposite was synthesized for the first time and its ability was evaluated for photocatalytic degradation of albumin in aqueous solutions under UV-A light. The resulting nanoparticles were then characterized using X-ray diffraction (XRD), scanning electron microscopy (FESEM), vibration magnetometer (VSM), and Fourier infrared (FTIR). The effects of some parameters such as pH, initial albumin concentration, catalyst concentration, and temperature were also investigated in the photodegradation of albumin. The results showed that the maximum removal of albumin was obtained at pH 9, catalyst concentration of 0. 5 g/l, initial albumin concentration of 150 mg/l, and room temperature in 90 min. Under the optimum conditions, the total amount of organic carbon (TOC) was 56%. Kinetic degradation experiments followed the pseudo-first-order kinetic model with a constant rate (k) of 0.0255 min-1. Therefore, due to the high performance of Fe3O4/ ZnO/Ag magnetic nanocomposite in the degradation of albumin as well as its easy synthesis and separation with an external magnetic field, it can be used as a suitable and environmentally friendly catalyst for the degradation of organic and resistant pollutants in the wastewater.
Mugashini Vasudevan; Aiswarya Kanesan; Sathaniswarman Remesh; Veeradasan Perumal; Pandian Bothi Raja; Mohamad Nasir Mohamad Ibrahim; Saravanan Karuppanan; Subash C.B Gopinath; Mark Ovinis
Abstract
The purpose of this research is to develop a sustainable and organic energy storage system from oil palm lignin waste-derived Laser Scribed Graphene embedded with molybdenum disulfide (LSG/MoS2). In this study, LSG/MoS2 hybrids were fabricated to overcome the graphene zero band gap, restacking issues ...
Read More
The purpose of this research is to develop a sustainable and organic energy storage system from oil palm lignin waste-derived Laser Scribed Graphene embedded with molybdenum disulfide (LSG/MoS2). In this study, LSG/MoS2 hybrids were fabricated to overcome the graphene zero band gap, restacking issues of molybdenum disulfide and to induce the electrical conductivity. Therefore, various amounts of LSG (0.1,0.5,1.0 g) were added in MoS2 precursor to produce nanoscale LSG/MoS2 hybrid nanostructure via the hydrothermal method. To justify the findings of LSG/MoS2 hybrid nanostructures, Raman spectroscopy, FESEM, TEM, and XRD were conducted. The D, G and 2D bands found in LSG confirm the formation of graphene from lignin. The morphology of LSG/MoS2 hybrids is porous and has a large surface area anchored with 3D MoS2 nanoflower on LSG. TEM has proved that LSG was wrapped with MoS2, where the presence of lattice spacing of 0.62 and 0.27nm, which corresponded to the (002) and (100) planes of MoS2 was observed. The electrochemical performance of the hybrids was conducted through Electrochemical Impedance Spectroscopy (EIS) demonstrated that increment in LSG in MoS2 precursor effects the impedance and resistances performances.
shima Amani; Mohammad Rostamizadeh; Ali Ghadimi
Abstract
In this study, zeolitic imidazolate framework (ZIF-8) nanocatalyst was synthesized by the thermal solvent method and doped by Fe species through wet impregnation technique. The nanocatalysts were applied for the degradation of Phenazopyridine Hydrochloride (PHP) through the heterogeneous Electro-Fenton ...
Read More
In this study, zeolitic imidazolate framework (ZIF-8) nanocatalyst was synthesized by the thermal solvent method and doped by Fe species through wet impregnation technique. The nanocatalysts were applied for the degradation of Phenazopyridine Hydrochloride (PHP) through the heterogeneous Electro-Fenton (HEF) process. The nanocatalysts were characterized by XRD, BET-BJH, FT-IR, FE-SEM, TEM, and acidimetric-alkalimetric titration techniques. The results showed the high surface area (1335 m2g-1) and homogenous dispersion of Fe species. The influence of different operating conditions was investigated, including pH level, nanocatalyst concentration, applied current, and PHP concentration. The optimum conditions for the HEF system over the Fe-ZIF-8 nanocatalyst were pH=7, 0.2 g L-1 of the Fe-ZIF-8 nanocatalyst, 100 mA, and 10 ppm of PHP concentration, which resulted in 99% PHP removal. The developed nanocatalyst had high reusability for the PHP removal in the HEF process. The results confirm the high potential of ZIF-8 nanocatalyst for pharmaceutical wastewater treatment through the HEF process.
Bahram Bagheri; Seyed Ali Hosseini; Habib Mehrizadeh
Abstract
The catalytic wet peroxide oxidation of phenol from aqueous wastewater and COD reduction over Cu-Mn2 and Cu-Co2 nano mixed oxides are reported. The effects of process variables of pH, reaction time and hydrogen peroxide dosage were investigated in the process over both catalysts. The catalysts were characterized ...
Read More
The catalytic wet peroxide oxidation of phenol from aqueous wastewater and COD reduction over Cu-Mn2 and Cu-Co2 nano mixed oxides are reported. The effects of process variables of pH, reaction time and hydrogen peroxide dosage were investigated in the process over both catalysts. The catalysts were characterized by X-ray diffraction (XRD) and it was concluded that the mixed oxides are in the form of the spinel structure. However, a little bit CuO was found in the mixed oxides. The morphology and particles size of the catalysts were investigated by scanning electron microscope (SEM). The morphologies and particle size of the catalyst were approximately the same with an average range of 40-60 nm. The catalytic results indicated the higher activity of CuCo2O4 spinel. The phenol oxidation on Cu-Co2 oxide was 82% after 40 min, whereas on the Cu-Mn2 oxide was 78% even after 50 min. The COD reduction The higher activity and reusability of the Cu-Co2 catalyst is attributed to the high synergistic effect between CuO particles and Cu–Co2 spinel, promoting phenol degradation.
Sheik Mohideen Badhusha M; Kavitha Balasubramani; Rajarajan M; Tharmaraj P; Suganthi Ayyadurai
Abstract
ZnO, single-doped (Co-ZnO, Cu-ZnO), and co-doped ZnO ((Co, Cu)/ZnO) were effectively synthesized by the citrate gel combustion technique. The samples were characterized by UV-visible diffuse reflectance spectroscopy (UV-vis-DRS), Fourier transforms infrared spectroscopy (FT-IR), X-ray powder diffraction ...
Read More
ZnO, single-doped (Co-ZnO, Cu-ZnO), and co-doped ZnO ((Co, Cu)/ZnO) were effectively synthesized by the citrate gel combustion technique. The samples were characterized by UV-visible diffuse reflectance spectroscopy (UV-vis-DRS), Fourier transforms infrared spectroscopy (FT-IR), X-ray powder diffraction (XRD), Scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDX) and photoluminescence spectroscopy (PL). The average particle size was 30.33 nm as calculated from XRD patterns for (Co, Cu)/ZnO. UV-Vis absorption spectrum indicates that the co-doped ZnO exhibits increased visible light absorption compared to the undoped one. The photoluminescence spectroscopy shows that the separation efficiency of photo-induced electrons and hole is enhanced by the co-doping strategy. (Co, Cu)/ZnO nanoparticles demonstrated a strong visible light response and high photocatalytic activity for Rhodamine B (RhB) degradation under irradiation by visible light (400-500 nm). The visible-light photocatalytic activity of the prepared (Co, Cu)/ZnO may come about because of the incorporation of Co, Cu atoms in ZnO, photo-induced electron-hole pairs and extended the spectral response to the visible region. The antibacterial and antifungal activities of ZnO, Co-ZnO, Cu-ZnO, and (Co, Cu)/ZnO were studied respectively with Staphylococcus aureus (Gram-positive), Escherichia coli (Gram-negative) ( bacterial strain) and Aspergillus flavus, Candida albicans (fungal strain). The (Co, Cu)/ZnO enhanced the antimicrobial activity.
Reza Jazini Zadeh; Mohammad Sayadi; Mohammad Reza Rezaei
Abstract
The present study was performed on the adsorption of 2,4-Dichlorophenoxyacetic acid from aqueous solutions by amine-modified magnetic nanoparticles. The adsorbent was synthesized by the co-precipitation method. The adsorbent properties of Fe3O4@SiO2-NH2 were investigated using XRD, FTIR, TGA, VSM and ...
Read More
The present study was performed on the adsorption of 2,4-Dichlorophenoxyacetic acid from aqueous solutions by amine-modified magnetic nanoparticles. The adsorbent was synthesized by the co-precipitation method. The adsorbent properties of Fe3O4@SiO2-NH2 were investigated using XRD, FTIR, TGA, VSM and TEM. Adsorbent efficacy was studied by investigating the effect of pH, initial concentration of pollutants, and adsorbent dose, and times. Finally, kinetics equations and isotherms models were used to describe the data. The results showed that the highest removal percentage was observed at pH 6 and the initial concentration 20 mg/l of 2,4-Dichlorophenoxyacetic acid. Adsorption capacity was increased by 65.3% bypassing the time from the beginning of the process to 60 min. The results from the study of isotherms and adsorption kinetics presented that the sorption procedure follows the pseudo-second-order kinetics and the Langmuir isotherm with R2> 99. The maximum adsorption capacity of Fe3O4@SiO2-NH2 is 116.3 mg/g. Besides, thermodynamic studies have shown that the adsorption process in the present study is endothermic and spontaneous. The experiments showed that Fe3O4@SiO2-NH2 synthesized nanoparticles could be an excellent method to remove 2,4- Dichlorophenoxyacetic acid contaminants from the aqueous solutions due to the high efficiency, simplicity, and lack of secondary contamination in the solution.
Hamid Kazemi Hakki; Pouya Shekari; Ahmad Najafidoust; Nosrat Dezhvan; Masoume Seddighi Rad
Abstract
Nowadays, not only the lack of water sources but also water pollution by industrial wastewater has become a major challenge. One of the pollutants of water resources, which threatens water resources, is phenol that enters the environment through wastewater from various industries such as petrochemicals, ...
Read More
Nowadays, not only the lack of water sources but also water pollution by industrial wastewater has become a major challenge. One of the pollutants of water resources, which threatens water resources, is phenol that enters the environment through wastewater from various industries such as petrochemicals, refineries, pharmaceuticals, etc. This compound, due to its toxicity, high stability and solubility in water caused many problems and it is very important to remove it from industrial wastewater. Therefore, the main purpose of this study is to remove phenol from wastewater by using catalytic oxidation. Fe-ZSM-5 catalyst was synthesized by precipitation method and its characteristics were determined by XRD, FTIR, SEM and BET analyses. The results of XRD analysis showed that the iron ions are in the MFI structure and the catalyst has a good crystallinity phase, so that it retains its MFI structure. BET analysis showed the specific surface area of the synthesized catalyst is 293g/m2. The catalytic activity of Fe-ZSM-5 was investigated in degradation of phenol and the results showed that the mentioned catalyst had the highest removal percentage (85.82%) in 90 min at pH = 4.25 and T= 70 °C. The effect of various parameters such as ultraviolet radiation, initial pollutant concentration, catalyst loading and H2O2 concentration on the catalytic activity were also investigated.
Ashkan Zolriasatein; Sepideh Abdolahzadeh; Negar Motakef kazemi
Abstract
Nowadays, the water shortage has become a severe issue all over the world, especially in some arid and undeveloped areas. Water is a renewable natural resource that can produce freshwater globally. Interestingly, some animals in nature can collect water from fog, which can be the inspiration to develop ...
Read More
Nowadays, the water shortage has become a severe issue all over the world, especially in some arid and undeveloped areas. Water is a renewable natural resource that can produce freshwater globally. Interestingly, some animals in nature can collect water from fog, which can be the inspiration to develop novel and functional water-collecting materials. A superhydrophobic surface that utilizes no energy has been synthesized from a water-collecting mechanism of the Stenocara beetles back structures. Firstly, this paper reviews the preparation methods of superhydrophobic surface coatings and then it examines patterns inspired by nature. In this paper, hydrophobic nanocoating was manufactured based on polyurethane (PUR) modified by nano-silica. Silica nanoparticles were synthesized via the sol-gel method. Then, silica nanoparticles were dispersed in polyurethane (PU) coatings. In this article, many factors of collecting moisture are evaluated, for example, temperature, pattern placement angle, pattern shape, types of surface patterns, and ambient humidity. Finally, are introduced the optimum situation for absorbing water from the fog.
Ashok V Borhade; Yogita R Shelke; Vivek D Bobade; Dipak R. Tope; Jyoti A. Agashe
Abstract
Water pollution is one of the serious main global concerns that affect humans and numerous people die due to various diseases caused by contaminated water because of the toxic and carcinogenic nature of dyes in effluents. It is essential to develop an efficient and effective method for wastewater ...
Read More
Water pollution is one of the serious main global concerns that affect humans and numerous people die due to various diseases caused by contaminated water because of the toxic and carcinogenic nature of dyes in effluents. It is essential to develop an efficient and effective method for wastewater treatment using a highly active and reusable catalyst. Herein we report heterogeneous catalyst MgZrO3@Fe2O3@ZnO nanoparticles by sol-gel approach. They were characterized by UV-visible diffused reflectance spectroscopy (UV-DRS), X-ray diffraction (XRD), Scanning electron microscopy (SEM), Energy dispersive x-ray analysis (EDAX), and High-resolution transmission electron microscopy (HRTEM) and selected area diffraction. This characterization confirmed the structure of MgZrO3@Fe2O3@ZnO and also confirmed excellent photocatalytic activity for the decolorization of Nigrosin dye under ambient conditions. The 96±0.5 % degradation was observed within 60 min using 20 ppm Nigrosin dye solution with 0.2 g of MgZrO3@Fe2O3@ZnO core-shell nanoparticles. A mechanistic approach for photodegradation of dye was established by Liquid chromatography-mass spectrometry (LCMS) with the identification of numerous smaller fragment molecule
Amir Hossein Baghaie; Raheleh Mirzaee
Abstract
Remediation of soils polluted with heavy metals or petroleum hydrocarbons is one of the environmental problems. This research aimed to evaluate the effect of carbon nanotubes, zeolite and AMF on triticale Ni concentration in a soil co-contaminated with diesel fuel and Ni. Treatments consisted of applying ...
Read More
Remediation of soils polluted with heavy metals or petroleum hydrocarbons is one of the environmental problems. This research aimed to evaluate the effect of carbon nanotubes, zeolite and AMF on triticale Ni concentration in a soil co-contaminated with diesel fuel and Ni. Treatments consisted of applying multi walled carbon nanotubes (MWCNs) and zeolite at the rates of 0 , 1 and 2 % (W/W) in a Ni-polluted soil (0, 75 and 150 mg Ni/kg soil) which was naturally polluted with diesel fuel under cultivation of triticale plant inoculated with AMF. After 70 days, plants were harvested and soil and plant Ni concentration was measured using atomic absorption spectroscopy (AAS). Soil microbial respiration and degradation of diesel fuel were also measured. Applying 2 % (W/W) zeolite and MWCNs significantly increased the diesel fuel degradation in soil by 12.3% and 14.5 %, respectively, while the plant Ni concentration was decreased by 8.9 % and 13.1%, respectively. Increasing soil pollution with Ni from 0 to 75 mg/kg soil significantly decreased the degradation of diesel fuel in the soil under cultivation of plant inoculated with AMF by 14.4%. In addition, the soil microbial respiration was also decreased by 11.8%. The results of this study showed that application of zeolite and MWCNs had significant effect on increasing diesel fuel degradation in heavy metal polluted soil that is a positive point in environmental studies.
Ashok V. Borhade; Dipak R. Tope; Jyoti A. Agashe; Sachin S. Kushare
Abstract
In the present work, the sol-gel derived powders of the chemical form FeCr2O4@ZnO@MgO Core-shell, has been synthesized and used as a photocatalyst. The synthesized core-shell nanoparticles characterized by various analytical techniques including FTIR, XRD, SEM-EDAX, and HR-TEM-SEAD. The successful performance ...
Read More
In the present work, the sol-gel derived powders of the chemical form FeCr2O4@ZnO@MgO Core-shell, has been synthesized and used as a photocatalyst. The synthesized core-shell nanoparticles characterized by various analytical techniques including FTIR, XRD, SEM-EDAX, and HR-TEM-SEAD. The successful performance of synthesized core-shell photocatalyst FeCr2O4@ZnO@MgO has been also demonstrated for the complete mineralization of Orange G dye. The effect of various operational parameters used in dye degradation such as concentration of dye, light intensity, amount of photocatalyst, effect of light and effect of electrolyte has been studied on the rate of reaction. TEM analysis clearly shows two layers of ZnO and MgO on FeCr2O4. The highest degradation rate was found with concentration of Orange G dye 10 ppm, 0.8 g of FeCr2O4@ZnO@MgO and time 50 min. The recyclability of the photocatalyst, FeCr2O4@ZnO@MgO was performed up to four runs. The degradation mechanism has been established by using LC-MS analysis and it was used to track the numerous intermediate products formed during the course of Orange G dye degradation.
Vikas Vikas; Pawan S. Rana
Abstract
The study involved the preparation of CoFe2O4 -CNT nanocomposites through the dispersion of CNT and ferrites into Xylene. Cobalt ferrites were synthesized using a chemical co-precipitation method. The main objective was to investigate the structural, optical and photocatalytic properties of the synthesized ...
Read More
The study involved the preparation of CoFe2O4 -CNT nanocomposites through the dispersion of CNT and ferrites into Xylene. Cobalt ferrites were synthesized using a chemical co-precipitation method. The main objective was to investigate the structural, optical and photocatalytic properties of the synthesized nanocomposites using Rose Bengal dye as a model pollutant. Structural analysis was conducted using Transmission Electron Microscopy (TEM) and XRD, confirming the formation of a Single-phase spinal structure of ferrites and nanocomposites with an average particle size of around 25 nm. UV-Vis. spectroscopy and Photoluminescence (PL) were used to study the optical properties of the samples, indicating that the bandgap of the sample is within the visible range, making it suitable for visible light photocatalysis. The band gap shifted to a slightly lower side after the formation of the nanocomposite, and the PL revealed that recombination time increased after the formation of nanocomposites compared to pure CoFe2O4. FTIR spectroscopy confirmed the formation of the spinal structure of ferrites and identified various bonds present in the sample. The photocatalytic activity of the sample showed a significant increase in the dye degradation capacity of ferrites. This increase in photocatalytic activity was consistent with earlier results obtained by PL spectroscopy, which confirmed the formation of a Z-scheme visible light photocatalyst.

