Lydia Rohmawati; Lytha Rizqika Lailia; Nugrahani Primary Putri; Munasir Nasir; Darminto Darminto
Abstract
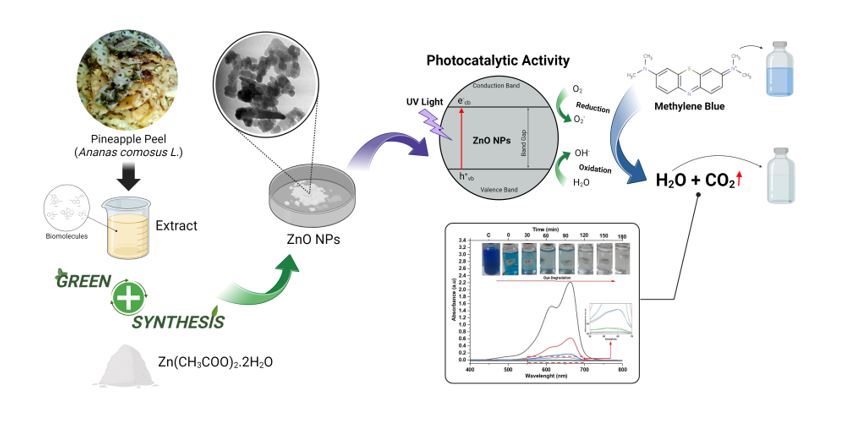
ZnO nanoparticles can be used as a photocatalyst in waste treatment because they have good photodegradation. Synthesis of ZnO nanoparticles using green synthesis method from pineapple peel extract, whose results were characterized by XRD (Xray Diffraction), FTIR (Fourier Transform Infra-red), FESEM (Field ...
Read More
ZnO nanoparticles can be used as a photocatalyst in waste treatment because they have good photodegradation. Synthesis of ZnO nanoparticles using green synthesis method from pineapple peel extract, whose results were characterized by XRD (Xray Diffraction), FTIR (Fourier Transform Infra-red), FESEM (Field Emission-Scanning Electron Microscopy), TEM (Transmission Electron Microscopy), Raman spectroscopy, photoluminescence, and a photocatalytic activity assay. This research showed that ZnO nanoparticles had wurtzite phase, alcohol functional groups, and phenol O-H, C=C alkenes, C-O, C-N, and Zn-O. ZnO nanoparticles had a particle size of 20.04 nm, a spherical shape, and a band gap energy of 3.28 eV. The Raman active mode E2(High) at 439.05 cm-1 confirmed the formation of pure phase wurtzite. Photoluminescence results indicated that two emission peaks at 392.07 nm and 595.07 nm were associated with defects such as oxygen and zinc vacancies. The results of the photocatalytic effectiveness test showed the highest percent degradation value of 99.86% at 180 minutes using UV light.
Ahmad Vakili Tajareh; Hossein Ganjidoust; Bita Ayati
Abstract
In this study, TiO2/Fe3O4 and TiO2/Fe3O4/MWCNT as a new magnetic nanophotocatalytic materials were synthesized. For this, TiO2 nanoparticle were fixed on an inert surface by sonochemical method. X-ray Diffraction (XRD), scanning electron microscopy (SEM), UV-Vis diffuse reflectance spectroscopy (DRS), ...
Read More
In this study, TiO2/Fe3O4 and TiO2/Fe3O4/MWCNT as a new magnetic nanophotocatalytic materials were synthesized. For this, TiO2 nanoparticle were fixed on an inert surface by sonochemical method. X-ray Diffraction (XRD), scanning electron microscopy (SEM), UV-Vis diffuse reflectance spectroscopy (DRS), vibration sample magnetometry (VSM) were used to characterize the magnetic nanocomposites. SEM analysis indicated that TiO2 and Fe3O4 nanoparticles were adhered to MWCNT. Ability of the nanocomposites to remove organic pollutants were investigated by photodegradation of Acid Red 14, Acid Blue 19, Reactive Red 77, and Methyl Orange dyes simulated conditions similar to sunlight. Furthermore, the removal efficiency of AR14 were investigated under direct sunlight irradiation, with an initial concentration of 50 mg/L by TiO2, TiO2/Fe3O4 and TiO2/Fe3O4/MWCNT nanocatalysts were 89.83, 21.19 and 86.27, respectively. According to the results, addition of carbon nanotubes to the TiO2/Fe3O4 magnetic nanocomposite, increased the efficiency of AR14 photodegradtion through change in energy gap visible waves and the scavenging role of carbon nanotubes. Also, The reusability of nanocomposites was assessed in five consecutive cycles of 6 hours, The results showed that after 5 cycles the degradation rate decreased only 7.79 %.
