Taher Yousefi; Ramin Yavari; D Rezaei Uchbelagh; Abbas Ariamanesh
Abstract
Al-Ce-Mn oxide samples were synthesized by the cathodic electrochemical method at current densities of 5, 15, and 35 mAcm-2. The XRD, SEM, and EDX techniques were used for the characterization of samples. The SEM images show that at high current density the one-dimensional(nanowire) structure and at ...
Read More
Al-Ce-Mn oxide samples were synthesized by the cathodic electrochemical method at current densities of 5, 15, and 35 mAcm-2. The XRD, SEM, and EDX techniques were used for the characterization of samples. The SEM images show that at high current density the one-dimensional(nanowire) structure and at low current density two-dimensional (nanosheet) structure were obtained. Moreover, the particle sizes are decreased with increasing the current density. The samples were applied for the uptake of fluorine (F-) ions from solutions. The influence of the contact time, initial fluoride concentration, and solution pH on the adsorption was investigated. The results showed more than 80 % of F- ions were uptake from solution during the three hours initial contact times and the uptake capacity has little change at pH below 6 and it has a sharp decline with increasing solution pH. The kinetic data were well fitted to the pseudo-second-order model and the equilibrium adsorption data was well described by the Langmuir isotherm model. The adsorption capacity was 48 mg/g at pH 6 and room temperature.
Mohammad Reza Jalali Sarvestani; Roya Ahmadi
Abstract
This paper investigated boron nitride nanocage performance as an adsorbent and sensing material for removal and detection of trinitroanisole by density functional theory. The calculated adsorption energies, Gibbs free energy changes (ΔGad), adsorption enthalpy changes (ΔHad) and thermodynamic ...
Read More
This paper investigated boron nitride nanocage performance as an adsorbent and sensing material for removal and detection of trinitroanisole by density functional theory. The calculated adsorption energies, Gibbs free energy changes (ΔGad), adsorption enthalpy changes (ΔHad) and thermodynamic equilibrium constants (Kth) revealed the adsorption process is experimentally feasible, spontaneous, exothermic and Irreversible. The highly negative adsorption energy values and bond lengths between B12N12 and trinitroanisole indicated the interaction between the adsorbate and the adsorbent is a chemisorption process. The N-O and C-N bond lengths and the density values showed that trinitroanisole complexes with boron nitride cage have higher explosive velocity and detonation pressure than the pure trinitroanisole without B12N12. The frontier molecular orbital parameters such as band gap, chemical hardness, electrophilicity, chemical potential and charge capacity were also studied and the findings proved B12N12 is an excellent sensing material for fabricating novel electrochemical and thermal sensors for detection of trinitroanisole.
Syed Reyaz Hasan; Zaheer Abbas; Md. Shahzad Khan
Abstract
The recent theoretical investigation has advocated the Al2O3 monolayer as a stable atomic configuration. This work deals with the interaction of NH3 and PH3 towards this monolayer configuration. Structural and electronic investigation suggests a strong affinity of the monolayer towards the NH3 and PH3 ...
Read More
The recent theoretical investigation has advocated the Al2O3 monolayer as a stable atomic configuration. This work deals with the interaction of NH3 and PH3 towards this monolayer configuration. Structural and electronic investigation suggests a strong affinity of the monolayer towards the NH3 and PH3 molecules. PDOS analysis reveals hybridization between the molecular orbital of NH3/PH3 and Al2O3-monolayer. The electronic energy bandgap of the Al2O3 monolayer gets reduced by 0.26eV and 0.21eV respectively, on NH3 and PH3 adsorption. In the bandstructure analysis of the Al2O3-monolayer, the energy band dispersion got flattened after the toxic molecular gas (NH3/PH3) adsorption, suggesting strong sensitivity towards the toxicants. Mulliken population analysis witnessed a robust amount of charge transferred from the toxic molecules to the Al2O3-nanosheet. A competency in electrical conductivity and energy-band gap flattening of the NH3/PH3-Al2O3 configurations is an interesting outcome of the present work. All these findings suggest strong sensitivity of the 2D-monolayer for NH3/PH3.
Sumon Chakrabarty; Md. Anas Mahmud; Mosummath Hosna Ara; Shovon Bhattacharjee
Abstract
The present study aimed to synthesize copper oxide (CuO) nanoparticles (NPs), which were used as an adsorbent for the sequestration of Iron (Fe) (III) ion from aqueous solution. The synthesized NPs were characterized with the help of X-ray diffraction (XRD) spectroscopy, Field Emission scanning electron ...
Read More
The present study aimed to synthesize copper oxide (CuO) nanoparticles (NPs), which were used as an adsorbent for the sequestration of Iron (Fe) (III) ion from aqueous solution. The synthesized NPs were characterized with the help of X-ray diffraction (XRD) spectroscopy, Field Emission scanning electron microscopy (FESEM), and Energy-dispersive X-ray spectroscopy (EDS). The SEM and XRD analyses indicated the average size of CuO NPs were ~25 nm with a rod-like shape. Based on the batch experiments the maximum adsorption observed at pH 9 with removal efficiency 98.38%, initial metal ion concentration of 10 ppm, and contact time 60 min. This study also revealed that adsorption capacity increases when the concentration of adsorbents decreases. To specify the adsorption characteristics of CuO NPs, the adsorption equilibrium data were treated with Langmuir and Freundlich models, which demonstrated that the removal of Fe (III) ions was mostly favored by the physical process followed by the multilayer adsorption on the heterogeneous surface of the adsorbents. Finally, this study concludes that CuO NPs could be used as a promising material for the removal of Fe(III) ion from aqueous Solution.
Noorallah Delijeh; Taher Yousefi; Hasan Aghayan; Ramin Yavari; Mehdi Parvini; Hossein Ghasemi mobtaker
Abstract
In the current work synthesis and modification of graphene oxide with Nickel Hexa Ferrocyanide (NiHCF) nanoparticles has been reported. The Graphene oxide- Nickel Hexa Ferrocyanide (GO-NiHCF) was used as an adsorbent to remove Cesium (Cs) ions from a simulated solution. The obtained product was characterized ...
Read More
In the current work synthesis and modification of graphene oxide with Nickel Hexa Ferrocyanide (NiHCF) nanoparticles has been reported. The Graphene oxide- Nickel Hexa Ferrocyanide (GO-NiHCF) was used as an adsorbent to remove Cesium (Cs) ions from a simulated solution. The obtained product was characterized with XRD, SEM, TGA, FTIR, and BET techniques. The SEM images and XRD pattern confirms the successful immobilization of Nickel Hexa Ferrocyanide on graphene oxide sheet. The cesium removal ability of GO-NiHCF was evaluated in batch mode. Effect of various parameters such as pH, initial concentration, contact time, and interferences ions were studied. The results cleared that the maximum adsorption for Cs removal was 240 mg g-1. Equilibrium modeling studies suggest that the data are reasonably and relatively fitted well to the Langmuir adsorption isotherm. Kinetic studies show that sorption process is fairly rapid and the kinetic data are fitted well to the pseudo-second order rate model. This composite offers strong potential in the field of elimination of Cs that requires rapid and complete decontamination.
Alireza Behzadi; Amirhosein Yazdanbakhsh
Abstract
Recently, there has been growing concern about the presence of pharmaceutical compounds and particularly antibiotics as emerging contaminants. This study employed high specific surface area organic aerogels to remove Tetracycline antibiotics. For this purpose, resorcinol formaldehyde aerogel (RF) was ...
Read More
Recently, there has been growing concern about the presence of pharmaceutical compounds and particularly antibiotics as emerging contaminants. This study employed high specific surface area organic aerogels to remove Tetracycline antibiotics. For this purpose, resorcinol formaldehyde aerogel (RF) was synthesized via the sol-gel process and dried under ambient drying conditions. The synthesized RF aerogel was modified by incorporating 1 wt.% graphene with 1 wt.% m-phenylenediamine during the synthesis process to prepare RF-G1/PmPDA1. Eventually, the performance of the synthesized samples as adsorbents was evaluated under various parameters such as the effects of pH values (2-12), adsorbent dose (4-10 mg), and adsorbent with antibiotics contact time (3-24 h). FTIR, FESEM, BET, CHN, and EDS tests were conducted to characterize the samples. Afterward, the adsorption rate of Tetracycline antibiotics was measured using UV-Vis. The BET test results revealed that the modification of the RF aerogel sample also increased the specific surface area from 96 to 308 m2/g. The results also discovered that the removal rate of Tetracycline antibiotics for the RF aerogel and RF-G1/PmPDA1 was obtained to be 65.2% and 93.3% at optimal pH of 4 and 4, respectively.
Sedigheh Daroumi; Mohammad Ali Aroon; Ramin Yavari; taher yousefi; Hossein Ghasemi mobtaker
Abstract
In this study, the flat sheet membranes including the neat polyethersulfone (PES) and the mixed matrix membranes (MMMs) containing 20 wt. % polyethersulfone (PES) and various amounts of Fe3O4 nanoparticles were prepared using wet phase inversion and conventional casting methods. Manganese ion rejection ...
Read More
In this study, the flat sheet membranes including the neat polyethersulfone (PES) and the mixed matrix membranes (MMMs) containing 20 wt. % polyethersulfone (PES) and various amounts of Fe3O4 nanoparticles were prepared using wet phase inversion and conventional casting methods. Manganese ion rejection and permeate flux as a performance evaluation of the prepared membranes was studied and compared. The characteristics of the fabricated membranes and the synthesized nanoparticles were fulfilled by transmission electron microscopy, field emission scanning electron microscopy and contact angle measurement. The operational parameters such as polymer concentration, pressure, pH, manganese ion concentration and time for manganese ion rejection and permeability were firstly optimized on the neat PES membrane. In the next steps, the performance of the fabricated MMMs containing various amount of Fe3O4 nanoparticles and PES (20%wt.) was evaluated and compared under these optimized conditions. Under the optimal conditions obtained for the rejection of manganese ions by neat PES, the fabricated MMMs had better performance than the neat PES membrane. Also, the results showed that the best performance of the prepared MMMs with the manganese rejection percentage of 89.3% and permeate flux of 28.7 L.m-2.h was found to belong to the PES membranes containing 0.1 wt.% of Fe3O4.

Pantea Arjmandi; Fariba Hargalani
Abstract
A simple and eco-friendly method for synthesis nanoparticles is using a green chemistry technique. Also, the utilization of green nanoparticles for the treatment of industrial wastewater could be an outstanding plan to confront environmental pollutions. The novelty of this study was to use leaf extract ...
Read More
A simple and eco-friendly method for synthesis nanoparticles is using a green chemistry technique. Also, the utilization of green nanoparticles for the treatment of industrial wastewater could be an outstanding plan to confront environmental pollutions. The novelty of this study was to use leaf extract of Stevia Rebaudiana Bertoni for green synthesized TiO₂ NPs and assessing its functioning for the photocatalytic treatment of Naphthol from real sample wastewater in a self-designed photoreactor. The amount of nano-adsorbent changes was studied under different conditions such as the amount of naphthol concentration, pH, and time period of degradation. Results: The results of the XRD showed that the Anatase and Rutile phase of TiO₂ conformed to cards no.JCPDS21-1272 and no.JCPDS21-1276 respectively. The EDX analysis illustrated the existence of TiO₂ with a weight percentage of 50.17 wt.% for Ti and 49/83 for O. The size of the particles in the SEM photo was found to be about 17nm. The removal of naphthol content was measured by the UV-Vis method. The optimum pH for naphthol removal by TiO₂ is pH = 9, the optimal contact time is 20 min, and the optimal concentration of Naphthol is 3 mg/L. Comparing the Freundlich and Langmuir adsorption isotherm models revealed that the absorption model in this study is in complete conformity with the Freundlich adsorption model. This study affirms that the green synthesis of Stevia leaf extracted is a modern beneficial procedure for the preparation of TiO₂ nanoparticles. This method is straightforward, cost-effective, eco-friendly, and rapid.
Narasimha Raghavendra; Leena V Hublikar; S.M. Patil; Pritam Bhat
Abstract
Microwave assisted biosynthesis of nanoparticles has been a cost effective, environmentally benign, and alternative to the chemical method. In this context, we report eco-friendly and robust nanoparticles synthesized using the bio-waste (Banana leaves) extract material through a microwave method. The ...
Read More
Microwave assisted biosynthesis of nanoparticles has been a cost effective, environmentally benign, and alternative to the chemical method. In this context, we report eco-friendly and robust nanoparticles synthesized using the bio-waste (Banana leaves) extract material through a microwave method. The newly synthesized Banana Leaves extract -Silver Nanoparticles (BL-AgNPs) is confirmed by using the UV-Visible, FT-IR spectroscopy and Scanning Electron Microscopy (SEM) techniques. UV-Vis spectrum shows the widening of the band around 476 nm, which confirms the polydispersed nature of BL-AgNPs. FT-IR spectroscopy explores that, hydroxyl and carbonyl groups in the Banana Leaves extract play vital role in the reduction of silver ions and also attach with AgNPs. The phytochemical studies reveal that, the polyphenols and alkaloids present in the BL extract act as reducing and stabilizing agent, which is responsible for the reduction of Ag+ (silver ions) to Ag (BL-AgNPs) and stabilization of BL-AgNPs. This clearly confirms the formation of silver nanoparticles (AgNPs). SEM results revealed that, bead shape of BL-AgNPs with particle size of 80 to 100 nm. In conclusion, BL-AgNPs exhibits promising anticancer activity against lung cancer and breast cancer cell line by endorsing inhibition of cell migration and proliferation on low concentration.
Armin Ehsani Amoli; Mojtaba Masoomi; Mazyar Sharifzadeh baei; Fatemeh Babei; Ghasem Firouzzade pasha
Abstract
In this work, hydrothermal technique and precursor materials obtained from the wastes of filtration unit of gas pressure reduction station were used to create ZnO-Fe2O3 nanocomposite. FT-IR, FE-SEM, XRD and TEM analyzes were used to investigate the properties of the produced nanocomposite. XRD analysis ...
Read More
In this work, hydrothermal technique and precursor materials obtained from the wastes of filtration unit of gas pressure reduction station were used to create ZnO-Fe2O3 nanocomposite. FT-IR, FE-SEM, XRD and TEM analyzes were used to investigate the properties of the produced nanocomposite. XRD analysis showed the structure of ZnO and Fe2O3 without impurities. The crystal size of ZnO-Fe2O3 nanocomposite was determined to be about 53 nm. FE-SEM images showed a nanocomposite pattern with an approximate diameter of 50 nm. Finally, visual decomposition of anionic and cationic dyes under visible light was used to study the photocatalytic activity of ZnO-Fe2O3 nanocomposite. By exposing a metal halide lamp to light and darkness for 60 minutes and 150 minutes, respectively, it was possible to study the photocatalytic activity of the synthesized nanocomposite in removing anionic and cationic dyes from aqueous medium. In the photocatalytic degradation of anionic and cationic dyes, the following factors were considered as essential variables: pH, initial dye concentration, nanocomposite content and exposure time. In this study, the degradation percentage of anionic and cationic dyes of ZnO-Fe2O3 nanocomposite with a ratio of 0.75:1 was 99.89 and 99.9%, respectively. The amount of band gap was calculated by Tack plot method and electrical conductivity was calculated using electrochemical impedance spectroscopy, which reduced the band gap. And the resistance increases. Due to the acceleration of charge transfer at the heterogeneous junction surface and the suppression of electron/hole pairs from recombination, the ZnO-Fe2O3 nanocomposite significantly increased the visible light current response.
Fatemeh Shahverdi; Aboulfazl Barati; Mansour Bayat
Abstract
This research work aims to investigate the sorption characteristic of synthesized Poly (vinyl alcohol)/Chitosan nanofiber mats modified with aluminum-cerium spinel oxide (CeAlO3) nanoparticles for methylene blue (MB) removal from aqueous solutions. The sorption is carried out by a batch technique. The ...
Read More
This research work aims to investigate the sorption characteristic of synthesized Poly (vinyl alcohol)/Chitosan nanofiber mats modified with aluminum-cerium spinel oxide (CeAlO3) nanoparticles for methylene blue (MB) removal from aqueous solutions. The sorption is carried out by a batch technique. The structural characterization of this nanocomposite was performed by Fourier transform infrared spectroscopy (FTIR) and X-ray diffraction analysis (XRD). Scanning electron microscopy (SEM) results showed uniform net and improved nanofibers with diameters ranging about 420 and 450 nm, respectively. The optimum conditions of MB removal onto modified PVA/CS nanofibers were found to be: pH 10, contact time 45 min, and 0.01 g of adsorbent in 400 ml in aqueous solution. Furthermore, the experimental adsorption data were in excellent agreement with the pseudo-second-order kinetics. The experimental results showed that there is a good correlation between the obtained data and the adsorption isotherm in the concentration range studied (400-600 mg/l). The results revealed that the maximum adsorption capacity of MB was 817.81 and 714.61 mg/g onto improved and net nanofibers, respectively.
Sumathi Harohally Paramesh; Veerendra Shetty Ananthpur; Nagaraju Rajendraprasad
Abstract
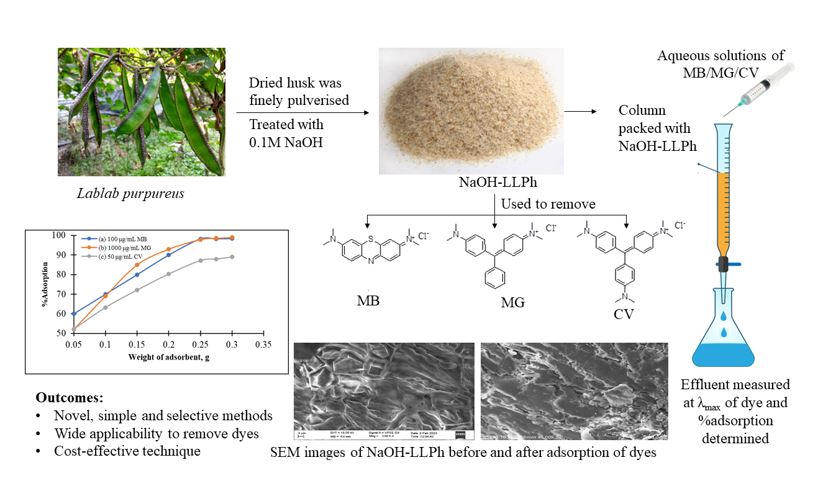
Novel results in this study showcase the utilization of sunlight-dried, ground Lablab purpureus husk (LLPh), treated with water and alkali, as a highly efficient bio-adsorbent for the removal of cationic dyes from aqueous solutions. Methylene blue (MB), malachite green (MG), and crystal violet (CV) were ...
Read More
Novel results in this study showcase the utilization of sunlight-dried, ground Lablab purpureus husk (LLPh), treated with water and alkali, as a highly efficient bio-adsorbent for the removal of cationic dyes from aqueous solutions. Methylene blue (MB), malachite green (MG), and crystal violet (CV) were effectively adsorbed onto NaOH activated LLPh (NaOH-LLPh) as bio-adsorbent. Employing the Chromatrap method within a column, successfully removed these dyes, while the surface morphology of the bio-adsorbent was elucidated through scanning electron microscopy (SEM) analysis. FTIR spectrometric data revealed valuable insights into the extent of adsorption. The impact of factors including adsorbate concentration, adsorbent dose, pH, contact time, and flow rate on the adsorption process was systematically studied and optimized. Up to 1000 µg/mL of MB and MG, 50 µg/mL of CV were found to be effectively removed by adsorption at pH 4-5, 3 and 2, respectively, at the flow rate of 1 mL/min. The results of kinetic studies and adsorption isotherms of above-mentioned dyes indicates that, all the three dyes follow the pseudo-second order kinetics. The adsorption of MB and MG are well fitted with the Langmuir isotherm model. The other dye CV suits with the Freundlich isotherm model. Based on the results, NaOH-LLPh, as an inexpensive and eco-friendly adsorbent, is suitable for the removal of cationic organic dyes from aqueous samples.
Seyedeh Maedeh Hashemi Orimi; Maryam Khavarpour; Sohrab Kazemi
Abstract
In the present study, adsorption behavior of mesoporous molecularly imprinted polymers for bisphenol A was investigated. Molecularly imprinted nanopolymers were synthesized by precipitation polymerization using bisphenol A as a template molecule. Two molecular ratios of templet: functional monomer: cross-linker ...
Read More
In the present study, adsorption behavior of mesoporous molecularly imprinted polymers for bisphenol A was investigated. Molecularly imprinted nanopolymers were synthesized by precipitation polymerization using bisphenol A as a template molecule. Two molecular ratios of templet: functional monomer: cross-linker (1:6:30 (MIP-6) and 1:4:20 (MIP-4)) was considered for experiments. Ethylene Glycol Dimethacrylate (EGDMA) as a Crosslinker, metacrylic acid (MAA) as a functional monomer and 2, 2´-azobisisobutyronitrile (AIBN) as an initiator were used for the synthesis of polymers. In addition, Langmuir and Freundlich adsorption isotherms and pseudo-first-order and pseudo-second-order kinetic models were studied for adsorption mechanism. Results showed that porous polymers with average pore diameter of 13 to 17 nm and specific surface area of 326 to 439 (cm3/g) were obtained. The maximum adsorption capacity was 400.1 μmol/g for MIP-6. SEM analysis showed that the synthesized polymer particles were spherical. The highest adsorption efficiency of bisphenol A achieved by MIP-6 was 71%.
Muthuswami Ruby Rajan; Raja Rohini
Abstract
Zinc is essential for aquatic biota including fishes at a lower concentration, but when it reaches higher concentration it becomes toxic. The objectives of the present work were related to the impact of different quantities of zinc oxide nanoparticles on the growth and hematology of Mrigal Cirrhinus ...
Read More
Zinc is essential for aquatic biota including fishes at a lower concentration, but when it reaches higher concentration it becomes toxic. The objectives of the present work were related to the impact of different quantities of zinc oxide nanoparticles on the growth and hematology of Mrigal Cirrhinus mrigala. The zinc oxide nanoparticles were synthesized by chemical precipitation method and characterized using by UV-VIS, SEM, EDAX, FTIR and XRD. Different quantity of zinc oxide nanoparticles such as 0, 5,10,15,20 and 25mg/100g were prepared by using a fish meal, groundnut oil cake, wheat flour, and tapioca flour. Feed utilization and hematological parameters of Mrigal were estimated after 21 days of feeding. UV-visible adsorption spectra show that the peak absorbance of ZnO nanoparticles was observed 500 nm. SEM shows that nanoparticles formed are clustered because of the adhesive nature of flower-shaped appearance. EDAX shows that the zinc oxide nanoparticles and the peaks are located between 1.0Kev and 8.5Kev. The FTIR spectrum of zinc oxide nanoparticles was analyzed in the range of 400-4000cm-1 and spectral bands were observed. The XRD results were viewed as the crystalline nature and average size of zinc oxide nanoparticles. Survival rate indicated that all Mrigal were healthy during the period of 21 days except in feed II,IV, and V. The feed utilization and growth parameters are higher in feed IV. Hematological parameters such as hemoglobin, RBC, Hematocrit, MCV, MCH, MCHC of Mrigal progressively increased and WBC and platelets decreased with increase in the quantity of Zinc Oxide nanoparticles.
Femina Kanjirathamthadathil Saidu; Alex Joseph; Eldhose Varghese; George Vazhathara Thomas
Abstract
Metal nanoparticles incorporated conducting polymer nanocomposites have outstanding properties and potential applications in various fields and significant research has been carried out over the last two decades for the development of efficient methods for their synthesis. The current study describes ...
Read More
Metal nanoparticles incorporated conducting polymer nanocomposites have outstanding properties and potential applications in various fields and significant research has been carried out over the last two decades for the development of efficient methods for their synthesis. The current study describes a microwave-assisted, rapid, and environmentally friendly method for depositing silver nanoparticles (AgNPs) over poly(1-naphthylamine) (PNA) using clammy cherry (Cordia Obliqua willd) extract as a reductant to create silver/nanocomposites (Gr-Ag/PNA) with varying silver contents. Thermal stability and charge transfer kinetics of PNA was significantly improved upon introducing AgNPs, as evidenced by the thermogravimetric analysis and electrochemical investigations, respectively. All prepared Gr-Ag/PNA nanocomposites could show improved catalytic activity towards the borohydride-aided reduction of 4-nitrophenol (4-NP) and the pseudo-first-order rate constants showed a direct relationship with the percent of silver incorporated over PNA. Additionally, for the first time, the Gr-Ag/PNA modified carbon paste electrode (Gr-Ag/PNA/CPE) was utilized to validate its usefulness and applicability in the electrocatalytic reduction of 4-NP. A low-cost enzymeless voltammetric 4-NP sensor based on Gr-Ag/PNA/CPE was fabricated and it showed excellent selectivity for 4-NP, as well as a strong linear response over a wide range of 4-NP concentrations (30-1000 𝜇M) and a detection limit of 6.25 𝜇M.
kimia yadolahi; Arezoo Ghadi; Seyed Abolhasan Alavi
Abstract
The aim of this project is the production of The Descurainia Sophia adsorbent in nano dimensions using a super-grinding disk mill. The top-down method was used in the process of nanogel preparation. After complete drying of the stems, the stems were reduced to smaller sizes by an electric mill, and by ...
Read More
The aim of this project is the production of The Descurainia Sophia adsorbent in nano dimensions using a super-grinding disk mill. The top-down method was used in the process of nanogel preparation. After complete drying of the stems, the stems were reduced to smaller sizes by an electric mill, and by using the available sieve to standard number 60, adsorbent granulation was performed. The adsorbent was used to remove copper from aqueous solution. The tests and their optimization results were based on the design of experiments in three levels of variables using Taguchi method. According to the experiment results, pH, contact time, and adsorption mass are the main factors and the most influential effect on the removal of copper from the aqueous solution is the pH parameter, which has a lower p-value. As the pH increases from 5 to 9, the efficiency of copper metal removal increases. The concentration of hydrogen ions is the most important parameter affecting the adsorption process. The effect of temperature on adsorption efficiency has been investigated in the range of 20-50°C. Finally, the highest efficiency of copper cation removal was at 30°C and 8.89%. The results showed that the pH of the solution had the most effect on the copper removal efficiency and in the playing environment, the effect of copper removal was more than the acid and neutral conditions of the solution. Also, the adsorbent mass and contact time have the most effect on copper removal after the pH parameter in the Taguchi method.
Sadegh Roushenas; Maryam Female Nikzad; Ali Asghar Ghoreyshi; mohsen ghorbani
Abstract
In this study, pectin/γ-Fe2O3/gl nanocomposite was synthesized using a single-step chemical precipitation method and used as an eco-friendly adsorbent to remove Cd2+ and Pb2+ from aqueous solution. The nanocomposite was characterized by FE-SEM, EDX, FTIR, XRD, VSM, and TEM analyses. The effect ...
Read More
In this study, pectin/γ-Fe2O3/gl nanocomposite was synthesized using a single-step chemical precipitation method and used as an eco-friendly adsorbent to remove Cd2+ and Pb2+ from aqueous solution. The nanocomposite was characterized by FE-SEM, EDX, FTIR, XRD, VSM, and TEM analyses. The effect of various parameters such as solution pH (2 to 5), contact time (0 to 60 min), initial ion concentration (10 to 200 mg. L-1), and adsorbent dosage (0.1 to 0.4 g.L-1) on the removal efficiency was investigated. The maximum adsorption capacity of Cd (II) in the conditions (pH: 5, dose of adsorbent: 0.2 g.L-1, and contact time:40 min, initial concentration: 50 mg.L-1 ) and Pb (II) in the conditions (pH: 4.5, dose of adsorbent: 0.1g.L-1, and contact time:30min, initial concentration: 50 mg.L-1 ) was 470 and 325 mg. g-1, respectively. The adsorption kinetics was studied using several kinetic models including Langmuir isotherm, Freundlich isotherm, Sips isotherm, and Temkin isotherm. Results indicated that the adsorption mechanism could be well represented by the pseudo-second-order model. The equilibrium data of Cd (II) and Pb (II) adsorption were reasonably described by the Sips and Langmuir isothermal models. The positive value of delta H and negative values of delta G exhibit the endothermic and spontaneous nature of the adsorption process.
Masoud Samandari; Afshin Tagva Manesh; Seyed Ali Hosseini; Sakineh Mansouri
Abstract
The catalytic wet peroxide oxidation (CWPO) of phenol from wastewater using Mg-Al LDH and calcined LDH at 500 ºC was investigated. The LDH was synthesized by co-precipitation and characterized by XRD, FTIR, SEM, EDS and BET. XRD result showed that during calcination of LDH at 500 ºC, LDH decomposed ...
Read More
The catalytic wet peroxide oxidation (CWPO) of phenol from wastewater using Mg-Al LDH and calcined LDH at 500 ºC was investigated. The LDH was synthesized by co-precipitation and characterized by XRD, FTIR, SEM, EDS and BET. XRD result showed that during calcination of LDH at 500 ºC, LDH decomposed to the mixed oxide. The SEM images approved Mg-Al LDH comprised of flakes and the calcined LDH comprised of spherical nanoparticles. BET results indicated the specific surface area of 100.2 and 86.3 m2.g-1 for pure LDH and calcined LDH, respectively. The process was optimized by one factor at a time method and considering four process factors i.e. reaction temperature, peroxide dosage, initial phenol concentration, and reaction time. The optimum conditions resulted at initial phenol concentration of 100 ppm, reaction temperature of 60 ºC, with peroxide volume of 3 mL and time on stream of 45 min over calcined LDH with maximum 85% removal of phenol. On the pure LDH, the maximum phenol removal (79%) resulted at peroxide volume of 2.5 mL at 55 min. The study concluded that the calcined Mg-Al LDH due to synergistic effect of MgO and Mg-Al mixed oxide showed higher catalytic activity despite a relatively low surface area.
Mohamed Naous; Ahmed Halfadji
Abstract
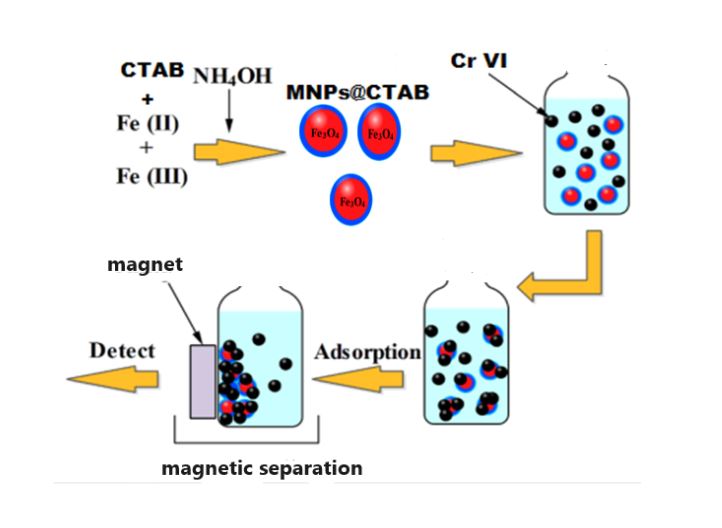
In this study, maghemite and magnetite nanoparticles were functionalized with cetyl trimethyl ammonium bromide(CTAB) surfactants, in order to obtain effective chromium removal from wastewater. X-ray diffractometry (XRD), transmission electron microscopy (TEM), and Fourier-transform infrared spectrophotometry ...
Read More
In this study, maghemite and magnetite nanoparticles were functionalized with cetyl trimethyl ammonium bromide(CTAB) surfactants, in order to obtain effective chromium removal from wastewater. X-ray diffractometry (XRD), transmission electron microscopy (TEM), and Fourier-transform infrared spectrophotometry (FTIR) were used to characterize the functionalized nanoparticles. Various parameters, including pH, initial chromium concentration, added salt, and adsorbent dose, were evaluated in batch experiments to evaluate chromium removal efficiency. Adsorbent dose and chromium ions show a synergistic relationship with pH and the chemical and electrostatic interactions between cationic surfactant and negatively charged Cr(VI) ions. In both types of functionalized nanoparticles, Cr(VI) was efficiently removed at low pH values with CTAB@MNPs, but the pH increased negatively impacted the removal process. Additionally, Fe3O4@CTAB mainly adsorbs chromium chemically, reducing Cr(VI) to Cr(III), with less impact from competitive ions compared with γ-Fe2O3@CTAB.At pH = 2, adsorbent dose = 5 g/L, and initial chromium concentration = 1 mg/L, maghemite@CTAB achieved a high chromium removal efficiency of 95%. In contrast, magnetite@CTAB achieved a chromium removal efficiency of 95.77% in 7 minutes and 30 seconds at pH = 4, adsorbent dose = 12 g/L, and initial chromium concentration = 98 mg/L. Notably, magnetite outperformed maghemite by a factor of 100 in chromium elimination, which can be attributed to the presence of two adsorption mechanisms, chemical and physisorption, in magnetite nanoparticles, whereas maghemite only had physisorption.
Balasubramani Kavitha; Sharumathi Selvam; Sivakumar Subburam
Abstract
The purpose of this study was to determine the adsorptive characteristics of a MnWO4/ZnS nanocomposite for removing Amaranth dye from aqueous solution. A simple chemical precipitation approach was used to make the MnWO4/ZnS nanocomposite. The crystal structure, morphology, and pore size of the resulting ...
Read More
The purpose of this study was to determine the adsorptive characteristics of a MnWO4/ZnS nanocomposite for removing Amaranth dye from aqueous solution. A simple chemical precipitation approach was used to make the MnWO4/ZnS nanocomposite. The crystal structure, morphology, and pore size of the resulting nanocomposites were evaluated by UV-vis-DRS, FT-IR, XRD, SEM, EDAX and BET. In a laboratory batch adsorption experiment, the effect of operational parameters such as adsorbent dose, starting dye concentration, agitation speed, contact time, and temperature was investigated to optimise the conditions for maximum amaranth removal. To reduce the number of trials and the associated costs, an artificial neural network (ANN) was used to forecast dye removal effectiveness. For amaranth dye, a contact time of 180 minutes, an adsorbent dosage of 0.35 g/l, and an initial dye concentration of 10 M resulted in a 96 percent dye removal. Different models were used to fit the equilibrium isotherm data. Langmuir and Temkin models have high R2 and are in good agreement with the experimental data (0.9966 and 0.9927). T and film diffusion may be involved in the sorption process, according to the kinetic analysis. When the experimental data was compared to the dye adsorption efficiency predicted by the artificial neural network model, it was discovered that this model can accurately predict the behaviour of the amaranth dye adsorption process on MnWO4/ZnS under various conditions.
Susarla Sastry
Abstract
Nanotechnology is getting an incredible drive due to the potential of manipulating metals into their nano size particles. The synthesis and characterization of nano particles using green technology have many applications. The wet chemical techniques used presently in the synthesis of nano particles are ...
Read More
Nanotechnology is getting an incredible drive due to the potential of manipulating metals into their nano size particles. The synthesis and characterization of nano particles using green technology have many applications. The wet chemical techniques used presently in the synthesis of nano particles are deleterious along with flammable conditions. Silver nanoparticles have the capability of killing microbes in an effective manner. This paper explains about the green technology and pollution free methodology for synthesizing silver particles at nano scale using 1mM silver nitrate solution from the extracts of Carica papaya, Emblica officianalis, Azadirachta indica and Cocos nucifera. When the silver nanoparticles are synthesized the solution turns to brownish yellow colour. The tools used in the characterisation of silver nano particles are Ultra Violet - Visible absorption Spectroscopy and Field Emission Scanning Electron Microscopy. The solutions with silver nanoparticles showed the maximum absorption at 450 nm with Ultra Violet - Visible spectroscopy. It is found that C. Papaya and E. oficianalis showed the maximum absorbance of 0.578 and 0.59 respectively at 450 nm. The average range of the produced silver nano particles are analysed to be 5 – 70 nm with FESEM and the shape is examined to be spherical.
Harshal Dabhane; Suresh Kushinath Ghotekar; Pawanwan Jagannath Tambade; Shreyas Pansambal; H C Ananda Murthy; Rajeshwari Oza; Vijay Medhane
Abstract
Nowadays, green syntheses have received crucial attention as a reliable, developing and eco-benevolent protocol for synthesizing a broad range of nanomaterials (NMs) including metal/metal oxides NMs, bio-inspired materials and hybrid/composite NMs. As such, biogenic synthesis is regarded as a significant ...
Read More
Nowadays, green syntheses have received crucial attention as a reliable, developing and eco-benevolent protocol for synthesizing a broad range of nanomaterials (NMs) including metal/metal oxides NMs, bio-inspired materials and hybrid/composite NMs. As such, biogenic synthesis is regarded as a significant tool to mitigate the destructive impacts associated with the conventional approaches of synthesis for NMs generally utilized in industry and laboratory. In this review, we summed up the general protocols and mechanisms of green synthesis routes, especially for silver (Ag), silver oxide (Ag2O), cadmium (Cd), copper (Cu), copper ferrite (CuFe2O4), palladium (Pd), aceprophyline, cellulose and graphene nanomaterials/nanoparticles using cow urine. Importantly, we explored the main role of biological constituents which is existed in cow urine. These essential biomolecules act as reducing/stabilizing agents in solvent systems. The stability, phase formation and surface morphology of NMs using characterization techniques are also discussed. Finally, we covered the eclectic applications of such synthesized NMs in terms of anti-asthma, antimicrobial, antituberculosis, antioxidant, anticancer activity, catalytic activity and removal of pollutants dyes.
Dhanraj S Shirsath
Abstract
Magnetic nano adsorbent is cost-effective and easily synthesized in the laboratory by chemical Co-precipitation method that provides not only high adsorption capacity but also rapid adsorption rate. The magnetic nano adsorbents were synthesized by Ferric and Ferrous ions precursor solution in the presence ...
Read More
Magnetic nano adsorbent is cost-effective and easily synthesized in the laboratory by chemical Co-precipitation method that provides not only high adsorption capacity but also rapid adsorption rate. The magnetic nano adsorbents were synthesized by Ferric and Ferrous ions precursor solution in the presence of ammonium hydroxide. In the present investigation, a magnetic nano adsorbent has been employed for the removal of Pb (II) from an aqueous solution by batch adsorption technique along with photocatalysis. The different parametric study also carried out such as initial concentration of Pb (II), adsorbent dose, contact time, and Solution pH. The Pb (II) was fast adsorption and the equilibrium was achieved within 45 minutes. The amount Pb (II) adsorbed increases as the temperature increase. The optimal pH for Pb (II) was around 5.4 and for the removal of Pb (II) ions was up to 96.00%. The employed adsorbents were characterized by SEM, X-ray diffraction (XRD), Vibrating spinning magnetometer (VSM), and FTIR. The Kinetic of adsorption study was examined for the pseudo-first-order model and pseudo-second-order models. This Photocatalytic adsorption study obeys Pseudo second-order kinetic. The reusability and regeneration of magnetic nano adsorbents were studied and were recycled up to 87.00 %.
Rejani Padmavathiamma; Rani Kunjiraman Pillai
Abstract
Heavy metals are known to be toxic for living organisms even if they are present at low levels. Water pollution by heavy metals from industries is a dangerous environmental problem. Due to the ease, flexibility, and cost-effectiveness of the remediation process, adsorption has been widely implemented ...
Read More
Heavy metals are known to be toxic for living organisms even if they are present at low levels. Water pollution by heavy metals from industries is a dangerous environmental problem. Due to the ease, flexibility, and cost-effectiveness of the remediation process, adsorption has been widely implemented in heavy metal wastewater treatment. In the present study, nanostructured manganese oxide was used for the removal of the heavy metal ions from aqueous solutions by a batch adsorption method and have been modelled using classical Langmuir and Freundlich adsorption isotherms. we have successfully synthesized an efficient adsorbent through a cost-effective and eco-friendly method. Bio synthesis is widely applied for the synthesis of nano materials . Various techniques such as XRD, FTIR, SEM EDX ,TEM and UV–VIS spectroscopy have been used to characterize the nanometal oxide. The obtained nano manganese oxide rods have very good adsorption efficiency due to the presence of some functionalities associated with the oxide material.
Mahidhar Prasad Tiwari; Poonam Tiwari
Abstract
The present paper was depicted to analyze the water quality status in single terms and zooplanktons of the Bhagirathi river. The water quality parameters were analyzed in lab.such as pH, Total Dissolved Solid (TDS), Total Hardness (TH), Chloride, Dissolved Oxygen, Calcium , Biochemical Oxygen Demand ...
Read More
The present paper was depicted to analyze the water quality status in single terms and zooplanktons of the Bhagirathi river. The water quality parameters were analyzed in lab.such as pH, Total Dissolved Solid (TDS), Total Hardness (TH), Chloride, Dissolved Oxygen, Calcium , Biochemical Oxygen Demand (BOD and Chloride. In the I site of the first area , the value of WQI in monsoon 55.79, winter 31.73 and summer 45.66, In the site 2, the value of WQI in monsoon 61.46 ,winter 38.17and summer 46.42 , In the site 3 ,the value of WQI in monsoon 61.36 , winter 32.91 and summer 47.55 were observed. Water quality index in the all study site is good in winter and summer, but poor in monsoon season. These fresh water systems also support various forms of aquatic life. The zooplankton of our study site mainly comprised of Protozoa, Rotifer, Copepoda and Cladocera. A total of 11 genera of Zooplanktons were observed during the course of study. The maximum number of Zooplanktons genera was during winter and minimum during rainy season. The most remarkable aspect of River Bhagirathi at these sites seems to be the tremendous potential to heal the health of entire river ecology that get damaged due to insufficient water released from the hydro-electric power project after dam wall into the main river channel .Immediate steps need to be taken in order to restore the ecological balance – mainly in main river channels after the dam wall of hydro-electric power projects.



