Sathiya Sheela D; P Viswanathan; K Kalimuthu; A Vanitha
Abstract
The development of biologically enthused green synthesis of silver nanoparticles (SNPs) has concerned significant global awareness about medical science and disease treatment. This paper discusses the green synthesis of SNPs using organic green sources; here we report a facile bottom-up ‘green’ ...
Read More
The development of biologically enthused green synthesis of silver nanoparticles (SNPs) has concerned significant global awareness about medical science and disease treatment. This paper discusses the green synthesis of SNPs using organic green sources; here we report a facile bottom-up ‘green’ route for the synthesis of SNPs using aqueous leaves extract of Pogostemon speciosus (Benth.) and evaluate its in-vitro anti-inflammatory, antibacterial and photocatalytic activities. The nanoparticles were investigated for the preparation of denaturation particles with PSLASNPs and the evaluation of anti-inflammatory activity with Protein denaturation and HRBC stabilization assays. Later, these PSLASNPs were studied for their potential role in antibacterial activity by well diffusion method, and Photocatalytic activity on degradation of dyes was demonstrated by using dyes Crystal violet, Coomassie blue, and Congo red. At 1000 µg/ml, the PSLASNPs have the greatest prevention of protein denaturation (71.92±1.37%), whilst the stabilization of the HRBC membrane exhibited significant anti-inflammatory action (64.39±1.61 %). The PSLASNPs showed the best antibacterial activity at the concentration of 10 µg/ml against Bacillus subtilis (8.2 mm), followed by Pseudomonas stuberia (6.2 mm) and Escherichia coli (6.4 mm), Staphylococcus aureus (5.3 mm), Staphylococcus gallinarium (4.5 mm) respectively at the same concentrations. Crystal violet, Coomassie blue, and Congo red were used for Photocatalytic activity on the breakdown of dyes. After 35 minutes, the degradation process was determined to be complete by the transformation of the reaction mixture's color to colorless. As a result, the PSLASNPs have anti-inflammatory, antibacterial, and photocatalytic activities.
Ubaithulla Baig A; Vadamalar R; Vinodhini A; Fairose S; Gomathiyalini A; Jabena Begum N; Shaista Jabeen
Abstract
Silver and zinc oxide are well known for both antimicrobial and pro-healing properties. ZnO is a biocompatible and bio-safe material that possesses photo-oxidizing and photocatalysis impacts on chemical and biological species. ZnO nanomaterials can interact chemically as well as physically to exhibit ...
Read More
Silver and zinc oxide are well known for both antimicrobial and pro-healing properties. ZnO is a biocompatible and bio-safe material that possesses photo-oxidizing and photocatalysis impacts on chemical and biological species. ZnO nanomaterials can interact chemically as well as physically to exhibit antibacterial activities. Chemical interactions of the ZnO nanomaterials with bacterial cells lead to the photo-induced production of reactive oxygenated species (ROS), formation of H2O2, and release of Zn2+ ions. In contrast, physical interaction can show biocidal effects through cell envelope rupturing, cellular internalization or mechanical damage. Here, we present a green method using Tridax Procumbens leaf extract to synthesize Ag doped ZnO nanoparticles (NPs) to explore the synergistic antibacterial properties of Ag and ZnO nanoparticles against certain gram positive and gram negative bacterial strains. The newly synthesized Ag doped ZnO NPs were characterized by X-ray diffraction (XRD) to study the crystalline structure, composition and purity. Transmission electron microscopy (TEM), Scanning electron microscopy (SEM) and Dynamic Light Scattering (DLS) technique was used to study particle size, shape, and morphology. The XRD and UV studies confirmed the ZnO phase. The absorbance peak around 618 cm-1 - 749 cm-1 in the FTIR spectrum referred presence of silver. The surface morphological studies also supported the FTIR result. The synthesized sample exhibited enhanced antibacterial activity irrespective of all tested microorganisms than the standard antibiotic used. The maximum size distribution of particle is found to be around 60 nm from the DLS technique.
Neena Anna Kurien; K. V. Divya; Jessyamma Kurian; K. E. Abraham
Abstract
In this work we are suggesting a method to reduce the amount of metal nitrates used in industrial and agricultural applications with the help of photocatalytic effect. Also, the work discusses how the residual amount of metal nitrates in the soil can be made useful through the same effect. Though metal ...
Read More
In this work we are suggesting a method to reduce the amount of metal nitrates used in industrial and agricultural applications with the help of photocatalytic effect. Also, the work discusses how the residual amount of metal nitrates in the soil can be made useful through the same effect. Though metal nitrates like ferric nitrate and nickel nitrate shows characteristic absorption in the UV region, what we observed is an enhancement in this UV absorption when we treated this metal nitrates with Titania Tenorite nanocomposite due to its photocatalytic action. This absorbance enhancement property is an indication of increase in concentration of the metal nitrates in the solution with light irradiation. The increase in M–OH bonds owing to the action of the nanocomposite in the presence of light is the reason by which metal nitrates absorption increases. In precise a tiny amount of metal nitrates is needed for any practical use as it can automatically increase its concentration in presence of the nanocomposite by photocatalytic reaction. This will reduce the disposal of unwanted amount of metal nitrates into the surrounding especially water bodies. Also, the residue amount in the soil can act as UV absorbers. Thus, the combination of the metal nitrates with the nanocomposite can be made used for environmental remediation where the metal nitrates are used in large quantities.
Savita Sihag; Sheetal .; Jitender Pal; Monika Yadav
Abstract
The environmental concern occurs due to the extreme use of synthetic materials that have been fortified to develop innovative, multifunctional, and sustainable materials using copious lignocellulosic biomass. In this present study, work was done on the extraction of nanocellulose from wheat straw, and ...
Read More
The environmental concern occurs due to the extreme use of synthetic materials that have been fortified to develop innovative, multifunctional, and sustainable materials using copious lignocellulosic biomass. In this present study, work was done on the extraction of nanocellulose from wheat straw, and found that wheat straw is an admirable source of cellulose. Chemical processes were used to isolate the cellulose and remove unwanted lignin and hemicellulose from wheat straw followed by sonication, cryo-crushing, and magnetic stirring to achieve nanocellulose. The observed amount of cellulose (36.1%), hemicellulose (30.3%), lignin (17%), and ash content (9.2%) of raw wheat straw. Structural, morphological, and thermal characterization were estimated from FTIR, XRD, FESEM, TEM, DSC, TGA, and AFM for the identification and characterization of extracted cellulose from wheat straw. FTIR showed that the peaks at wavelength 1430.50 cm-1 and 1638.41 cm-1 both show that cellulose is present in the extracted nanocellulose. Extracted nanocellulose was crystalline and had a 68.96% Crystallinity Index. Morphological analysis, FESEM showed that the untreated wheat straw has an irregular porous structure but the extracted nanocellulose has a regular shape having straight fibers connected. TEM analysis showed that the extracted nanocellulose has a spherical shape structure connected, showing the regular shape, the obtained spherical shape regulates the nanocellulose for further applications. Thermal degradation was observed using TGA which shows that the nanocellulose decomposition was observed around 3600C. AFM determination shows a bell-shaped structure on a smooth surface with a particle height of 3.2 nm and the mean roughness of 110.4 nm was obtained from the extracted nanocellulose. Extracted nanocellulose has a particle size of 58.77 nm.
Zahra Mirkazehi; Mohammad Reza Rezaei; Mohammad Sayadi
Abstract
Landfill leachate is the fluid percolating through the landfill and is one of the most important environmental challenges that lead to the contamination of water and soil resources. In this study, magnetic graphene oxide nanoparticles with WO3 (GO-Fe3O4/WO3) were synthesized through the hydrothermal ...
Read More
Landfill leachate is the fluid percolating through the landfill and is one of the most important environmental challenges that lead to the contamination of water and soil resources. In this study, magnetic graphene oxide nanoparticles with WO3 (GO-Fe3O4/WO3) were synthesized through the hydrothermal method to eliminate chemical oxygen demand (COD) from leachate. The obtained products were characterized using X-ray diffraction (XRD), Fourier Transform Infrared Spectroscopy (FTIR), Scanning Electron Microscopy (SEM), Thermogravimetric analysis (TGA), and Vibrating sample magnetometer (VSM) analysis. The influence of various operating parameters, such as initial solution pH, adsorbent dosage, contact time, and temperature, on COD removal was studied. Additionally, kinetic, isotherm, and thermodynamic studies were conducted to evaluate the adsorption capacity of the adsorbent. The results revealed that the maximum adsorption capacity of GO-Fe3O4/WO3 was 2500 mg/g adsorbent at pH 4, a contact time of 90 minutes, an adsorbent dosage of 25 mg g-1, and a temperature below 298 K, respectively. According to the adsorption kinetic fitting results, the experimental adsorption data were well described by the pseudo-second order kinetic with an R2 value of 0.97, and the Freundlich isotherm equation with an R2 value of 0.99. The thermodynamic results indicated that the adsorption was spontaneous and exothermic for COD adsorption. In general, the adsorption process of the synthesized GO-Fe3O4/WO3 nanocomposite revealed that it is highly effective for landfill leachate treatment and has great practical value in leachate treatment.

Najmeh Atoub; Ahmad Amiri; Alireza Badiei; Jahan B. Ghasemi
Abstract
In this study, thiol-functionalized nanostructure silica type MCM-41 was successfully prepared via the facile one-pot hydrothermal method with low amounts of the directing agent. The mesoporous silica indicated a remarkable adsorption behavior toward Pb(II) ions without any interference of the competing ...
Read More
In this study, thiol-functionalized nanostructure silica type MCM-41 was successfully prepared via the facile one-pot hydrothermal method with low amounts of the directing agent. The mesoporous silica indicated a remarkable adsorption behavior toward Pb(II) ions without any interference of the competing ions. The main experimental variables affecting removal efficiency of the adsorbent were examined, and the optimized conditions were achieved as to be 6, 50 mg, and 30 min for solution pH, the adsorbent dosage, and contact time, respectively. The adsorbent was triumphantly used for the removal of Pb (II) ion from real water samples with a notable removal efficiency as 95%. The concentrations of the competitive ions in the solution were about 10 to 100 times more than Pb (II) ions. The results show that other ions had no interfering effect on the removal efficiency of Pb (II) ions. It means that SH-SiO2 has excellent selectivity for Pb (II) ions and is an appropriate candidate for removing Pb (II) ions from the real samples.
Aref Shokri; Safoora Krimi
Abstract
In this study, the TiO2 nanoparticles were supported on Y-type zeolite as a new photocatalyst and used to degrade Acid Red 18 in aqueous media. The nano photocatalyst was synthesized by coprecipitation procedure and characterized by Fourier transfer infrared (FTIR), field emission scanning electron microscopy ...
Read More
In this study, the TiO2 nanoparticles were supported on Y-type zeolite as a new photocatalyst and used to degrade Acid Red 18 in aqueous media. The nano photocatalyst was synthesized by coprecipitation procedure and characterized by Fourier transfer infrared (FTIR), field emission scanning electron microscopy (FE-SEM), and X-ray powder diffraction (XRD). The central composite design (CCD) was employed for experimental design. The effect of operative variables including contact time, photocatalyst dosage and pH were investigated. The ANOVA (analysis of variance) studies displays the second-order regression model and a high determination coefficient value (R2 = 0.9953, R2pred = 0.9642, R2adj = 0.9910) for the destruction of AR18 was obtained. The contour plots were applied to study the shares of each variable and their interactions on the degradation of AR18. The optimal circumstances predicted by the model were as the following: the catalyst concentration at 0.88g/L, pH at 6.5, and contact time in 125 min. In this situation, the predicted and actual dye removal were 98.5% and 96.3%, respectively. The removal of COD (chemical oxygen demand) after 125 min was 53% indicating, the notable performance of photocatalyst in mineralization of AR18.
Mahdieh Chegeni; Mozhgan Mehri; Mehdi Hosseini
Abstract
The MoS2/S-doped graphitic carbon nitride (MoS2/S-g-C3N4) was synthesized by a simple method and applied for methylene blue (MB) removal as an organic pollutant. The structure of MoS2/S-doped graphitic carbon nitride was characterized using FTIR, XRD, SEM, TGA and BET techniques. The accomplishment of ...
Read More
The MoS2/S-doped graphitic carbon nitride (MoS2/S-g-C3N4) was synthesized by a simple method and applied for methylene blue (MB) removal as an organic pollutant. The structure of MoS2/S-doped graphitic carbon nitride was characterized using FTIR, XRD, SEM, TGA and BET techniques. The accomplishment of MoS2/S-doped graphitic carbon nitride as an adsorbent was investigated to removal of MB from aqueous solution. The various parameters were studied such as: pH, initial MB concentration, adsorbent dose, temperature and time. The best findings were obtained at pH=8, 8 ppm MB concentration, 0.05 g MoS2/S-g-C3N4, 30 min and 22 ˚C. The Langmuir isotherm model was adopted with the obtained data. The kinetic studies were showed that the adsorption of methylene blue can be well described by the second-order equation. Maximum adsorption was calculated as 166 mg/g. The degradation of MB was studied by MoS2/S-doped graphitic carbon nitride under Light Emmition Diode (LED). Results showed that the MoS2/S-doped graphitic carbon nitride can enhance photocatalytic activity compared to pure g-C3N4 and MoS2/g-C3N4. The findings confirmed that the MoS2/S-doped graphitic carbon nitride can be applied as an efficient, low-cost adsorbent, and photocatalyst to remove of cationic dyes such as methylene blue.
Milind S Patil; Sanjay P Shekhawat
Abstract
This paper discusses the experimental and theoretical performance of a parabolic trough receiver using a nanofluid. The main aim of this work is to analyze the performance enhancement of the parabolic trough collector system. The thermal model is developed using Engineering Equation Solver (EES). Experimental ...
Read More
This paper discusses the experimental and theoretical performance of a parabolic trough receiver using a nanofluid. The main aim of this work is to analyze the performance enhancement of the parabolic trough collector system. The thermal model is developed using Engineering Equation Solver (EES). Experimental analysis was done with a water volume flow rate of 10 L/min and water inlet temperature range from 0 to 45 OC, also the volume fraction of Al2O3 nanoparticle varied from 1% to 5%. Experimental analysis conducted using Al2O3 nanoparticle mixed with water and used as heat transfer fluid in solar parabolic trough collector. Results compared and observed that the model has very good acceptance with the experimental results. It is observed that the thermal efficiency of the collector increased by 2 to 4% and receiver heat loss decreased from 0.82% to 2.72%. The receiver water temperature increased by 15% for the range of Al2O3 nanoparticle volume fraction. This work was carried out to investigate the use of renewable energy for water heating applications on rural farms in India. Small-sized PTC is simple in construction, economical, and does not require special skills to operate. However, considering the space requirement it would be better to investigate the method to improve the performance of PTC without changing the dimensions. One way to improve the performance is with the use of nanofluids. This work’s main finding is that the Nanoparticle with a volume fraction of 4 will improve the performance. It was observed that the temperature of the water was improved by 15% and the thermal efficiency was increased by 4%.
Nandini Venkat Iyer; Jayant A Kher; Shekhar D Bhame
Abstract
The present study features the synthesis of Dodecylbenzene sulphonic acid-doped polypyrrole tungsten oxide (PPy-WO3) nanocomposites and its photocatalyic studies on Methylene Blue (MB). The nanocomposites of PPy with a very low concentration of WO3 were prepared using 0.05 to 0.3 wt.%, of WO3 nanoparticles. ...
Read More
The present study features the synthesis of Dodecylbenzene sulphonic acid-doped polypyrrole tungsten oxide (PPy-WO3) nanocomposites and its photocatalyic studies on Methylene Blue (MB). The nanocomposites of PPy with a very low concentration of WO3 were prepared using 0.05 to 0.3 wt.%, of WO3 nanoparticles. The composites were characterized by using-ray Diffraction, FTIR and FESEM for phase identification, morphological studies. The electrical conduction of the nanostructured materials at room temperature exceeded that of PPy, with the electrical conductivity increasing linearly with higher WO3 concentrations. The band gap for the nanocomposite was found to be 2.12eV. The PL spectra substantiated the adequate segregation of charge carriers photoexcited in the samples. The nanocomposites were tested as photocatalysts for the degradation of methylene blue dye (MB). A two-step mechanism has been propsed for dye removal: adsorption (in the absence of UV light) and photodegradation on the photocatalyst surface (in the presence of UV light). The maximum removal efficiency for methylene blue dye was 98.31% in UV light for 0.16 g/L of the 0.3 wt.% PPy- WO3 nanocomposite, with a dye concentration of 5 mg/L. The addition of p-benzoquinone (*O2- scavenger) contributed to a significant decrease in the photodegradation efficiency of the catalyst, i.e.,63.21%, and thus can be believed as the main active species for the degradation of the methylene blue dye.

Ashwini Prabhakar Shende; NILANJANA MITRA
Abstract
In recent years, the development in the field of nanotechnology is due to the fascinating properties of nanoparticles. In the present study, plant based bioflocculant extracted from the fruits of Okra (Abelmoschus esculentus) was purified, characterized and used for the biosynthesis of iron nanoparticles. ...
Read More
In recent years, the development in the field of nanotechnology is due to the fascinating properties of nanoparticles. In the present study, plant based bioflocculant extracted from the fruits of Okra (Abelmoschus esculentus) was purified, characterized and used for the biosynthesis of iron nanoparticles. Fourier transform infra-red (FT-IR) spectral analysis revealed the presence of hydroxyl, carboxyl and sugar derivatives in the bioflocculant. The biosynthesized Fe nanoparticles were characterized using UV-vis spectroscopy, X-ray diffraction (XRD), Fourier transform infra-red (FT-IR), Scanning electron microscopy (SEM) and Atomic force microscopy (AFM). TEM analysis was performed and the size of synthesized Fe nanoparticles was found to be 50 nm which was assessed by dynamic light scattering (DLS) analysis. Flocculation activity of bioflocculant mediated Fe nanoparticles (BFFeNPs) was tested. The effects of various parameters on Pb(II)removal using BFFeNPs were evaluated using response surface methodology (RSM) based on Box Behnken Design (BBD).The BFFeNPs exhibited high Pb (II) removal efficiency (91.45%) under optimized parameters viz. pH 6, BFFeNPs dosage 0.2 g/L, contact time 30 min and temperature 30º C. A quadratic polynomial model was fit with the actual data of R2 0.99 for metal removal. To the best of our knowledge, this is the first report on the potential use of Okra bioflocculant mediated Fe nanoparticles synthesis for the cost effective and eco-friendly removal of lead from wastewater.
Rouhollah Hedayati; Morteza Hosseini; Ghasem Najafpour; Hosein Attar
Abstract
Cobalamins are one of the most complicated cofactors produced by the microorganisms. Propionibacterium freudenreichii has to follow the anaerobic and aerobic conditions respectively during a course of batch fermentation, for the production of the biologically active form of cobalamins. Magnetite (Fe3O4) ...
Read More
Cobalamins are one of the most complicated cofactors produced by the microorganisms. Propionibacterium freudenreichii has to follow the anaerobic and aerobic conditions respectively during a course of batch fermentation, for the production of the biologically active form of cobalamins. Magnetite (Fe3O4) nanoparticles can modify gas-liquid volumetric mass transfer coefficient in the fermentation system to create more efficient aeration step. Initially, the modified production of Fe3O4 nanoparticle through coprecipitation method was investigated, and the smallest size of nanoparticles optimized to 13.86 nm via Box-Behnken design of response to surface methodology (RSM). The optimum condition was at the synthesis temperature of 60 °C, reaction duration of 10 minutes, and the medium agitation speed of 700 rpm. Synthesized nanoparticles characterized by SEM images, PXRD and EDS analysis while EDS spectrum reflects 39.33% of Fe and 51.8% of oxygen atomic distribution, which confirms Fe3O4 nanoparticles synthesis. Magnetite nanoparticle suspension added to the fermentation medium to compare the effect of nanoparticles incorporation and dimethylbenzimidazole addition on the cobalamin production via Propionibacterium freudenreichii. NPs incorporation in the fermentation broth was able to increase cyanocobalamin production by 20%, while there was no incorporation of external DMBI in the medium. Finally, by the central composite design analysis, cyanocobalamin production from Propionibacterium freudenreichii fermentation was optimized to 1.548 mg.L-1. Oily sludge (as a new carbon source) of 4 %w/v, magnetite nanoparticles suspension of 7.5 %v/v, and the fermentation temperature of 37 °C caused to CCD optimum condition.
Hassanali Rasouli; Faezeh Jafarpisheh; Mohammad Ghorbanpour
Abstract
Dyes are produced as water pollutants in the textile, plastic, and dye industries. Many efforts have been made to remove dyes from industrial wastewater. In this area, Photocatalytic performance under Vis-lights is a useful and effective method. In this study, a series of highly efficient Sn-doped TiO2 ...
Read More
Dyes are produced as water pollutants in the textile, plastic, and dye industries. Many efforts have been made to remove dyes from industrial wastewater. In this area, Photocatalytic performance under Vis-lights is a useful and effective method. In this study, a series of highly efficient Sn-doped TiO2 photocatalysts were successfully developed using a simple heat treatment process. Three concentrations of SnCl2 powder (3%, 5%, and 7 %) were used for the preparation of materials. The characterizations of resulting materials were distinguished by scanning electron microscopy (SEM), diffusion reflection spectroscopy (DRS), and X-ray diffraction spectroscopy (EDX). Also, methyl orange dye was served to indicate the photocatalytic activity of Sn-doped material under visible light irradiation. The results indicated that both doped and also pure TiO2 have a uniform size. Anatase was the only identified phase in all the products, whether doped or un-doped. The resulting Sn-doped materials have demonstrated a band gap value range of 3.14 to 2.68 eV in comparison with pure TiO2 which shows a value of 3.2 eV. The photocatalytic evaluations indicated that the samples prepared in the presence of 3, 5, and 7% SnCl2 have degradation efficiencies of 85%, 85%, and 90%, respectively within 120 min, which are much higher than that of un-doped TiO2. The enhancement in the efficiency was attributed to the gap changes in anatase by the incorporation of Sn ions into the TiO2 lattice structure. Compared to pure TiO2, which has a band gap of 3.2 eV, the band gap values for doped TiO2 ranged from 3.14 to 2.68 eV.
Jeba R; Radhika S; Padma C M; Ascar Davix X
Abstract
Pure and copper-doped Zirconium oxide nanoparticles were synthesized using a co-precipitation process and investigated the effect of doping on photocatalytic and anti-microbial activities. The prepared samples are pure tetragonal phase, as shown by the X-ray diffraction pattern, and the crystallite size ...
Read More
Pure and copper-doped Zirconium oxide nanoparticles were synthesized using a co-precipitation process and investigated the effect of doping on photocatalytic and anti-microbial activities. The prepared samples are pure tetragonal phase, as shown by the X-ray diffraction pattern, and the crystallite size decreases as the dopant concentration increases. Higher dopant concentrations resulted in needle-shaped morphology, as seen in the SEM image. The presence of Zr, Cu, and O in the sample is confirmed by EDS analysis. According to UV-VIS analysis, when the Cu content is increased, a more significant wavelength absorption band edge is formed, and the band gap reduces with the increase in dopant concentration. All samples have magnetic hysteresis loops with diamagnetic background effects, according to VSM tests. A prominent and influential peak at 485nm in the PL spectra suggests that ZrO2 nanoparticles emit blue light. The produced nanoparticles were utilized as a photocatalyst to degrade Methylene Blue (MB) dye, and the results indicate that a high dopant concentration (0.08wt percent) outperforms pure and other dopant concentrations. Copper-doped ZrO2 has moderate anti-bacterial and anti-fungal activities.
Narasimhaa Naidu Loganathan; Kabilashen Readdyi Munusamy; Veeradasan Perumal; Bothi Raja Pandian
Abstract
This paper reports a facile carbonization method of a biopolymer to synthesize reduced graphene oxide with excellent electrochemical properties for use as a supercapacitor electrode. Oil palm lignin is used as the biopolymer-based graphene precursor, and a carbon dioxide laser is used to carbonize the ...
Read More
This paper reports a facile carbonization method of a biopolymer to synthesize reduced graphene oxide with excellent electrochemical properties for use as a supercapacitor electrode. Oil palm lignin is used as the biopolymer-based graphene precursor, and a carbon dioxide laser is used to carbonize the material via lithography. Using Raman Spectroscopy, the characterization of the resultant graphene (OP-LSG) revealed D, G, and 2D peaks corresponding to multilayer graphene. Scanning Electron Microscopy of OP-LSG revealed three-dimensional particle-like fibrous and porous nanostructures with an enhanced surface area. In a three-electrode setup in ferrocyanide electrolyte, cyclic voltammetry showed the electrode coated with OP-LSG achieving a specific capacitance as high as 108.044 mF/cm² at a scan rate of 0.01 V/s. The galvanostatic charge-discharge of OP-LSG revealed energy and power density values of 15 µWh/cm² and 597 µW/cm² at a scan rate of 0.01 V/s. The OP-LSG electrode retained 97.5% of its initial capacitance after 1000 charge-discharge cycles.
Mariam Rabaa; Amani Aridi; Ghassan Younes; Ramadan Awad
Abstract
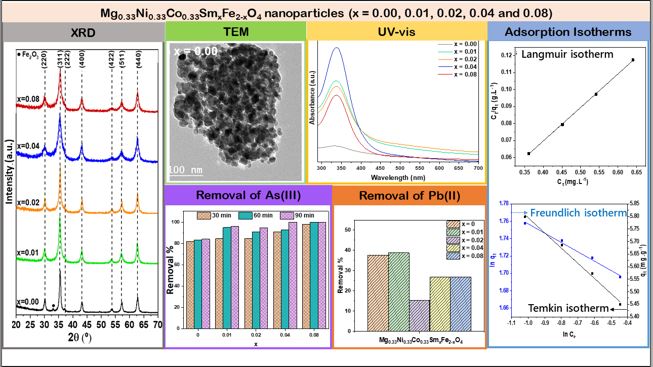
Heavy metal ions, such as As (III) and Pb (II), are harmful even at trace levels and have caused series health effects on living beings. Therefore, removing these heavy metal ions from the aqueous environment is highly desirable. In this study, Mg0.33Ni0.33Co0.33SmxFe2-xO4 nanoparticles, where x = 0.00, ...
Read More
Heavy metal ions, such as As (III) and Pb (II), are harmful even at trace levels and have caused series health effects on living beings. Therefore, removing these heavy metal ions from the aqueous environment is highly desirable. In this study, Mg0.33Ni0.33Co0.33SmxFe2-xO4 nanoparticles, where x = 0.00, 0.01, 0.02, 0.04 and 0.08, were synthesized by the co-precipitation method and characterized using X-Ray Diffraction (XRD), Transmission Electron Microscopy (TEM) and UV-Vis spectroscopy techniques in order to study the structural and optical properties. The prepared nanoparticles were applied as adsorbents for the removal of As (III) and Pb (II) from wastewater. Among the prepared samples, Mg0.33Ni0.33Co0.33SmxFe2-xO4 nanoparticles with x = 0.04 and 0.08 exhibited improved adsorption performance where As (III) was totally removed after 90 min. The experimental adsorption data of As (III) was well fitted with a second-order kinetics model and Langmuir isotherm. Furthermore, the highest removal % of Pb (II) was revealed by Mg0.33Ni0.33Co0.33SmxFe2-xO4 nanoparticles with x = 0.01. Thus, doping Mg0.33Ni0.33Co0.33Fe2O4 with Sm improved the adsorption performance of nanoparticles for the removal of As (III) more than that of Pb (II).
Mohammad Reza Mohammad Shafiee; Janan Parhizkar; Sasan Radfar
Abstract
Homogenous catalysis which the catalyst operates in the same phase as the reactants is definitely efficient in catalysis processes while it suffers from the impossibility or inconvenience of the removal of the catalyst from the reaction media. In this research, In2S3 nanoparticles were synthesized by ...
Read More
Homogenous catalysis which the catalyst operates in the same phase as the reactants is definitely efficient in catalysis processes while it suffers from the impossibility or inconvenience of the removal of the catalyst from the reaction media. In this research, In2S3 nanoparticles were synthesized by a simple precipitation method and then immobilized and stabilized in the porous structure as a substrate. The properties of pure hydrogel and In2S3 in hydrogel were characterized by FTIR, DRS, XRD, BET, BJH, FESEM, and EDX. The DRS results confirmed that the stabilization of nanoparticles in hydrogel led to redshift of bandgap. The hydrogel with In2S3 showed a more porous structure in comparison with pure hydrogel. Because of the decrease of bandgap and increase of specific surface area, In2S3 nanoparticles stabilized in hydrogel removed Rhodamine B (RhB) as a model pollutant very well. The performance of catalyst in the removal of RhB under dark condition (adsorption) and visible light irradiation (photocatalysis) was investigated and 77.7% and 95.2% of dye removal percentage were obtained in 120 min under dark and light irradiation, respectively. In conclusion, immobilization In2S3 as a high-efficiency visible light photocatalyst in hydrogel provided promising heterogeneous and reusable catalyst for water treatment
P. Koteswara Rao; B Vikram Babu; A. Rama Krishna; M. Sushma Reddi; B. Sathish Mohan; K. Anjani Devi; U. Susmitha; T. Raghava Rao
Abstract
The present study explores the green approach for the preparation of silver nanoparticles (AgNPs) through the reduction of silver nitrate by the cell-free stem and leaf aqueous extracts of Litsea glutinosa (L.glutinosa) and its potential antibacterial activity. The analytical instruments include scanning ...
Read More
The present study explores the green approach for the preparation of silver nanoparticles (AgNPs) through the reduction of silver nitrate by the cell-free stem and leaf aqueous extracts of Litsea glutinosa (L.glutinosa) and its potential antibacterial activity. The analytical instruments include scanning electron microscopy, Fourier transforms infrared spectroscopy, UV-visible spectroscopy, and X-ray diffraction spectroscopy confirmed the synthesis of smaller, uniformly spherical AgNPs (10-40 nm). The average crystalline size of prepared AgNPs produced by L. glutinosa leaf extract was found to be 19 mm. From UV-visible spectral analysis, the maximum absorbance peak appeared at 444 nm for leaf extract AgNPs different from stem extract AgNPs (422 nm), which are found to be specific for AgNPs. The L.glutinosa stem extract-assisted AgNPs have shown significant antibacterial activity against Bacillus subtilis (Gram-positive) and Escherichia coli (Gram-negative) in comparison to Gentamycin. Hence, the AgNPs obtained by green synthesis can be therapeutically explored against bacterial infections.
Buvaneswari K; Arunadevi R; Sashikala S; Kavipriya K
Abstract
Visible light active ZnWO4/ZrO2 nanocomposite was prepared via hydrothermal method. The nanocomposite was characterized by UV-visible diffuse reflectance spectroscopy (UV-vis-DRS), Fourier transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD), Scanning Electron microscopy (SEM), energy dispersive ...
Read More
Visible light active ZnWO4/ZrO2 nanocomposite was prepared via hydrothermal method. The nanocomposite was characterized by UV-visible diffuse reflectance spectroscopy (UV-vis-DRS), Fourier transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD), Scanning Electron microscopy (SEM), energy dispersive X-ray spectroscopy (EDX) and transmission electron microscopy (TEM) techniques. The XRD results showed that average particle size of ZrO2, ZnWO4 and ZnWO4/ZrO2 were found to be 29.20 nm, 23.78 nm and 20.14 nm respectively and the phase structure for ZrO2 and ZnWO4 in the composite was Rhombohedral and Monoclinic respectively. The UV–vis absorption spectra of the ZnWO4/ZrO2 nanocomposite noticeably shifted to the visible light region compared to that of the ZrO2. The prepared photocatalyst were composed of plate and spongy sphere with little agglomeration was seen from SEM result. The photocatalytic activities of the prepared nanocomposite was evaluated for the degradation of methyl orange (MO) under visible light irradiations. The effect of operational parameters such as initial dye concentration, pH, catalyst concentration and irradiation time have been investigated in detail. The photocatalytic degradation efficiency of ZnWO4/ZrO2, ZnWO4 and ZrO2 for 95%, 72% and 60 % respevtively. The high photocatalytic activity can be attributed to stronger absorption in the visible light region, a greater specific surface area, smaller crystal sizes, more surface OH groups, and to the effect of ZnWO4 doping, which resulted in a lower band gap energy.
Ashrafsadat Ghasemi; Mohmmad kia Kiani; fateme ravari
Abstract
Notwithstanding the enormous benefit of crizotinib, as anti lung cancer, severe toxicity as side effects are the main problem for this drug. In this research, the interaction of crizotinib over NH2 agent with C60 fullerene, boron-doped fullerene (C59B), and carboxylated fullerenes (C60COOH) using density ...
Read More
Notwithstanding the enormous benefit of crizotinib, as anti lung cancer, severe toxicity as side effects are the main problem for this drug. In this research, the interaction of crizotinib over NH2 agent with C60 fullerene, boron-doped fullerene (C59B), and carboxylated fullerenes (C60COOH) using density functional theory at B3LYP/6-311G(d) theoretical level in the gaseous phase and the water solvent were evaluated. Comparison of the drug-fullerenes complex in terms of structure, energy, type of interaction was performed through optimization, frequency, natural bond orbital, and atoms in molecules calculations. The results showed that the interaction of the drug with fullerenes due to the positive interaction energy and the unstable complexation could not be proper interaction between the drug and the nanoparticle. Binding between crizotinib and C59B is covalent, and the drug absorption is chemical. The interaction between crizotinib with C60COOH has been recognized as appropriate due to some properties such as higher solubility in water, relative stability, hydrogen bonding, and physical absorption of the drug. The result of this research can be counted as a promising strategy to reduce the toxicity and develop the anti lung cancer activity of crizotinib.
Pingal Sarmah; Khemnath Patir; Sonit Kumar Gogoi
Abstract
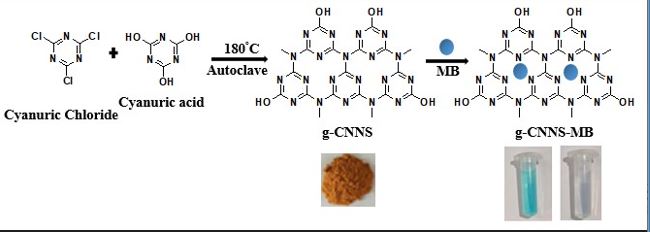
Here we report a simple and eco-friendly solvothermal synthesis of graphitic carbon nitride nanospheres (g-CNNS) at 180 oC. The synthesized g-CNNS is characterized by various analytical techniques such as FESEM, PXRD, BET isotherm, Zeta potential, EDX and FT-IR spectroscopy. The adsorption property of ...
Read More
Here we report a simple and eco-friendly solvothermal synthesis of graphitic carbon nitride nanospheres (g-CNNS) at 180 oC. The synthesized g-CNNS is characterized by various analytical techniques such as FESEM, PXRD, BET isotherm, Zeta potential, EDX and FT-IR spectroscopy. The adsorption property of g-CNNS is studied using four different dyes in aqueous medium and found that g-CNNS is an efficient material for cationic dye adsorption. A Comprehensive investigation of the kinetics, isotherms and thermodynamics of methylene blue (MB) adsorption, is carried out. The adsorption of MB on g-CNNS is well described by Langmuir isotherm model, and the experimental data fits well with pseudo-second order kinetics. The high rate of adsorption (94.92% MB removal in 120 minute at neutral pH) is attributed to electrostatic interaction between negative charged g-CNNS and cationic organic dye molecule. Additionally, g-CNNS demonstrated good reusability, retaining its efficiency for at least three cycles. Over all our findings suggests that g-CNNS has potential as an efficient adsorbent for wastewater treatment.
Kanchanmala Deshpande; Anju Thekkedath
Abstract
The key to simple and rapid detection of a large volume of samples lies in the hands of solution-based nanomaterial sensors. Quantification of mercury in the river and coastal water is analytically challenging due to the potential interference of the matrix. In this endeavor, lysine-capped gold nanoparticles ...
Read More
The key to simple and rapid detection of a large volume of samples lies in the hands of solution-based nanomaterial sensors. Quantification of mercury in the river and coastal water is analytically challenging due to the potential interference of the matrix. In this endeavor, lysine-capped gold nanoparticles (Lys-AuNPs) based colorimetric sensors are demonstrated here towards efficient detection of trace amounts of mercury ions (Hg2+) in coastal and estuarine water. The colorimetric behavior of Lys-AuNPs is related to surface plasmon resonance (SPR)During analysis, interestingly a decrease in the intensity of the original SPR peak at 530 nm was observed, with the concomitant appearance of a new peak at a longer wavelength due to agglomerated Lys- AuNPs. Developed sensors exhibit excellent performance in different environmental samples with high selectivity towards Hg2+ ions in the presence of other metal ions. For the analysis of coastal water samples, a low value of regression coefficient was observed due to the potential interference of salt in the sample. To overcome this, matrix-matching experiments were carried out. Developed Lys- AuNPs show good selectivity towards Hg2+ in matrixed matched diluted coastal water samples. With a sensitivity of 0.02 ppm, the sensor can be utilized to screen large numbers of coastal water samples for their Hg2+ content to satisfy coastal regulation norms. As a whole, this method is simple, sensitive, selective, cost-effective and can be used to screen large numbers of samples across the coastal area for monitoring Hg2+ concentration.
Amir Hossein Baghaie
Abstract
Reducing the entry of heavy metals into the food chain in plant cultivated in the areas contaminated with heavy compounds or petroleum compounds is one of the main environmental issues. This research was conducted to evaluate the role of co-inoculation of wheat with piriformospora indica and pseudomonas ...
Read More
Reducing the entry of heavy metals into the food chain in plant cultivated in the areas contaminated with heavy compounds or petroleum compounds is one of the main environmental issues. This research was conducted to evaluate the role of co-inoculation of wheat with piriformospora indica and pseudomonas putida on plant Cd concentration which has been planted in the Cd and petroleum hydrocarbon -polluted soil and treated with Zn oxide nanoparticles and agricultural steel slag. Treatments consisted of Cd-polluted soil (0, 10 and 20 mg kg-1 soil) that was amended with 0 and 2 % (W/W) Zn oxide nanoparticles and agricultural steel slag and the wheat plant that was co-inoculated with P.indica and P.putida that which was cultivated in a soil that was naturally polluted with petroleum hydrocarbon. After 90 days, plants were harvested and the Cd concentration was measured using atomic absorption spectroscopy. In addition the degradation rate of petroleum hydrocarbon in the soil was determined. Plant co-inoculation with P.indica and P.putida significantly decreased and increased the plant Cd concentration and degradation rate of petroleum hydrocarbon in the soil by 13.1 and 14.9%, respectively. In addition, using 2 % (W/W) Zn oxide nanoparticles and agricultural steel slag significantly decresed the plant Cd concentration by 18.2 and 15.4%, respectively. It can be concluded that plant co-inoculation with P.indica and P.putida had additive effect on degradation of petroleum hydrocarbon in the soil that was amended with Zn oxide nanoparticles and agricultural steel slag.
Mitra Madani Gargari; Kamran Rahnama; Maede Shahiri Tabarestani
Abstract
Among biocontrol fungi, Trichoderma species produce a wide range of bioactive compounds with antifungal activities. In this study, Di-(2-Ethylhexyl) Phthalate (DEHP) is identified via gas chromatography-mass spectrometry (GC-MS) device in Trichoderma atroviridae (1-3) secondary metabolites and its antifungal ...
Read More
Among biocontrol fungi, Trichoderma species produce a wide range of bioactive compounds with antifungal activities. In this study, Di-(2-Ethylhexyl) Phthalate (DEHP) is identified via gas chromatography-mass spectrometry (GC-MS) device in Trichoderma atroviridae (1-3) secondary metabolites and its antifungal effectiveness is confirmed. An eco-friendly approach for the extraction of DEHP is carried out by a nanoporous molecularly imprinted methacrylic acid-based network copolymer as a solid sorbent. Molecularly imprinted polymers (MIPs) are synthesized by precipitation polymerization using DEHP as a template, methacrylic acid (MAA) as a functional monomer and trimethylolpropane trimethacrylate (TRIM) as a cross-linker with molecular ratio (1: 4: 8). After the removal of DEHP, the nanoporous polymer can recognize and rebind specifically the same or structurally very similar molecules. The synthesized MIPs exhibit a suitable tendency to absorb the template with the highest binding capacity of 300 mg/g for DEHP in n-Hexane solvent as a solid phase extraction (SPE) system. The measured particle size of the MIPs with dynamic light scattering (DLS) is reported 75.38 nm. In addition, the porosity of the MIPs is evaluated by nitrogen gas adsorption/desorption using Brouneur Emmet Teller (BET) analysis. Results shows that nanoporous MIPs with an average pore diameter of 2.70 nm and a specific surface area of 309 (cm3/g) are achieved. According to the above-mentioned results, nanoporous MIPs can be considered as an acceptable candidate for separation of the antifungal bioactive compounds (natural fungicide) such as DEHP as an eco-friendly method to replace chemical pesticides.
leila Ardestani; Morteza Hosseini; Mohsen Jahanshahi; Alireza Amiri
Abstract
The bioactive compounds in extracts are prone to degradation by oxidation, heat, or light. Nanoencapsulation is one of the best techniques to keep the properties of these chemical compounds. The aim of this study was the extraction of Melissa officinalis (MO) and nanoencapsulation of the extract via ...
Read More
The bioactive compounds in extracts are prone to degradation by oxidation, heat, or light. Nanoencapsulation is one of the best techniques to keep the properties of these chemical compounds. The aim of this study was the extraction of Melissa officinalis (MO) and nanoencapsulation of the extract via chitosan as a biodegradable polymer. In this research, extraction of MO was investigated using various extraction methods and nanoencapsulation with MO extract was carried out via ionic gelation technique. The effectiveness of the extracts was evaluated by measuring the total phenolic content (TPC), antioxidant activity, and extraction efficiency of the solid contents. The highest efficiency was achieved for microwave-assisted extraction with the utmost values in each parameter. (TSC) was 22.81% and amounts of the TPC and antioxidant activity were 311.94 mg Gallic acid and 36 mg diphenyl picryl hydrazyl (DPPH) per 1g of the plant, respectively. Morphology study by field emission scanning electron microscopy (FE-SEM) indicated spherical shape nanoparticles with a diameter of 25nm. The size of the nanoparticles was evaluated by the Dynamic Light Scattering (DLS) technique for various concentrations of the used extracts in the encapsulation process. For 1.0, 3.0, and 5.0 mg /mL concentration, mean diameters were 24, 118, and 145 nm, respectively. Results indicated that microwave-assisted extraction was the best extraction method for MO and the encapsulation of MO extract could be created successfully with different particle sizes for the protection of bioactive compounds. Since MO is a beneficial herbal plant, the development of this research is recommended.



